Drug Information
General Information of This Drug
| Drug ID | DRG00008 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Dexamethasone | |||||
| Synonyms |
dexamethasone; 50-02-2; Decadron; Maxidex; Dexamethazone; Hexadecadrol; Dexasone; Hexadrol; Aeroseb-Dex; Fluormethylprednisolone; Desametasone; Superprednol; Visumetazone; Cortisumman; Decaderm; Decaspray; Dexacortal; Dexacortin; Gammacorten; Millicorten; Oradexon; Auxiron; Calonat; Dexason; Dexone; Dectancyl; Deltafluorene; Desamethasone; Desameton; Fortecortin; Mediamethasone; Mymethasone; Decalix; Dekacort; Dexacort; Dexameth; Dinormon; Loverine; Luxazone; Azium; Dexa-Scheroson; Prednisolone F; Dexa-sine; Isopto-Dex; Aeroseb-D; Anaflogistico; Aphtasolon; Decacortin; Decasone; Dergramin; Desadrene; Deseronil; Dexadeltone; Dexafarma; Dexalona; Dexapolcort; Dexinolon; Dexinoral; Dextelan; Policort; Spoloven; Corsone; Deronil; Dexapos; Mexidex; Sunia Sol D; Bisu DS; Dexa Mamallet; Dexa-Cortidelt; Dexa-Cortisyl; Prednisolon F; Lokalison F; Ocu-trol; Dex-ide; SK-Dexamethasone; Dexamethasonum; Dexaprol; Fluormone; Decagel; Dexone 4; Hexadrol Elixir; Pet Derm III; Dexamethasone Intensol; Hl-dex; Methylfluorprednisolone; Dexone 0.5; Dexone 1.5; Dexametasona; Fluorocort; Decaject; Decameth; Dexone 0.75; Dexpak; Dxms; Dexamethasone alcohol; Hexadrol Tablets; Desametasone [DCIT]; Decacort; Dexamonozon; Dextenza; Turbinaire; Ozurdex; 9alpha-Fluoro-16alpha-methylprednisolone; Dexametasona [INN-Spanish]; Dexamethasonum [INN-Latin]; Dezone; Decadron Tablets, Elixir; Azium (Veterinary); Dexametasone; Dexamethansone; Adexone; Osurdex; Posurdex; Dexycu; DexaSite; OTO-104; Corson; 16alpha-Methyl-9alpha-fluoro-1-dehydrocortisol; 16alpha-Methyl-9alpha-fluoroprednisolone; (3H)-Dexamethasone; Azimycin (Veterinary); delta1-9alpha-Fluoro-16alpha-methylcortisol; MK 125; NSC 34521; 1-Dehydro-16alpha-methyl-9alpha-fluorohydrocortisone; Naquasone (Veterinary); Tresaderm (Veterinary); CCRIS 7067; Diodex; DTXSID3020384; HSDB 3053; ISV-305; (11beta,16alpha)-9-Fluoro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione; Zema-Pak; 16-alpha-Methyl-9-alpha-fluoroprednisolone; 9-alpha-Fluoro-16-alpha-methylprednisolone; UNII-7S5I7G3JQL; 16-alpha-Methyl-9-alpha-fluoro-delta1-hydrocortisone; EINECS 200-003-9; 7S5I7G3JQL; MFCD00064136; NSC-34521; 16alpha-Methyl-9alpha-fluoro-delta(sup 1)-hydrocortisone; DXM [Steroid]; 16-alpha-Methyl-9-alpha-fluoro-delta(sup 1)-hydrocortisone; HEMADY; 16-alpha-Methyl-9-alpha-fluoro-1-dehydrocortisol; Dexamethasone (GMP); Dexamethasone, topical; CHEBI:41879; AI3-50934; delta(sup 1)-9-alpha-Fluoro-16-alpha-methylcortisol; 9A-FLUORO-16BETA-METHYLPREDNISOLONE; Prednisolone, 9alpha-fluoro-16alpha-methyl-; DTXCID10384; MLS001332507; 16.alpha.-Methyl-9.alpha.-fluoroprednisolone; Dexamethasone [USP:INN:BAN:JAN]; DXM (Steroid); 9-Fluoro-11beta,17,21-trihydroxy-16alpha-methylpregna-1,4-diene-3,20-dione; [3H]-dexamethasone; DEXA; [3H]dexamethasone; CIPRODEX COMPONENT DEXAMETHASONE; Decaject-L.A.; MAXITROL COMPONENT DEXAMETHASONE; TOBRADEX COMPONENT DEXAMETHASONE; (1R,2S,10S,11S,13R,14R,15S,17S)-1-fluoro-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,13,15-trimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-3,6-dien-5-one; DEXACIDIN COMPONENT DEXAMETHASONE; DEXASPORIN COMPONENT DEXAMETHASONE; Aphthasolone; TOBRADEX ST COMPONENT DEXAMETHASONE; Dexa-Mamallet; 9.alpha.-Fluoro-16.alpha.-methylprednisolone; Dexametasona (INN-Spanish); Dexamethasonum (INN-Latin); (8S,9R,10S,11S,13S,14S,16R,17R)-9-fluoro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthren-3-one; C22H29FO5; DEXAMETHASONE (MART.); DEXAMETHASONE [MART.]; 16.alpha.-Methyl-9.alpha.-fluoro-1-dehydrocortisol; DEXAMETHASONE (USP-RS); DEXAMETHASONE [USP-RS]; 1-Dehydro-16.alpha.-methyl-9.alpha.-fluorohydrocortisone; Dexamethasone (USP:INN:BAN:JAN); DEXAMETHASONE (EP MONOGRAPH); DEXAMETHASONE [EP MONOGRAPH]; DEXAMETHASONE (USP MONOGRAPH); DEXAMETHASONE [USP MONOGRAPH]; Dexamethasone Base; Decadron (TN); (8S,9R,10S,11S,13S,14S,16R,17R)-9-fluoro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-3-one; SMR000857119; BETAMETHASONE IMPURITY A (EP IMPURITY); BETAMETHASONE IMPURITY A [EP IMPURITY]; dexamethasone (tetramethyl-rhodamine conjugated ); Prednisolone, 9.alpha.-fluoro-16.alpha.-methyl-; alin; Amplidermis; Cortidexason; Dexafluorene; Dexamecortin; Dexamethsone; Fluorodelta; Aacidexam; Baycadron; Decadrol; Dexabliss; Dexachel; Dexalocal; Dexasine; Spersadex; .delta.(sup 1)-9-.alpha.-Fluoro-16-.alpha.-methylcortisol; 16.alpha.-Methyl-9.alpha.-fluoro-.delta.(sup1)-hydrocortisone; Dxevo; NSC34521; TaperDex; Aknichthol Dexa; Alin Oftalmico; Anemul mono; Azium Solution; Dexium Tablets; Azium Powder; Dexa-Rhinosan; .gamma.corten; ZoDex; 9-Fluoro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione; Alin Depot; Baycuten N; Lokalison-F; Apo-dexamethasone; Decadron DP; Dexasone 4mg; Alba-Dex; Zonometh Solution; Dexameth-A-Vet; PHL-dexamethasone; PMS-dexamethasone; TaperDex 6-day; TaperDex 7-day; Decasone R.p.; Dexamethasone-omega; NCGC00091019-08; Dexasone 0.5mg; TaperDex 12-day; HiDex 6-day; Dexasone 0.75mg; Sandoz Dexamethasone; Dexamethasone1.5 mg; Pregna-1,4-diene-3,20-dione, 9-fluoro-11,17,21-trihydroxy-16-methyl-, (11beta,16alpha)-; Dexamethasone Solution; Dexamethasone Injection; DEXYCU KIT; Maxidex Ont 0.1%; Maxidex Sus 0.1%; Azium Boluses 10 mg; Dexamethasone 1.5 mg; Pet Derm III Tablets; Dexameth-A-Vet Injection; Spectrum5_002019; Azium Tablets 0.25 mg; DEXAMETHASONE 6-Day; Azium Oral Solution 2 mg; DEXAMETHASONE [MI]; MolMap_000018; (9R,14S)-Dexamethasone; DEXAMETHASONE [INN]; DEXAMETHASONE [JAN]; SCHEMBL3774; 16-alpha-Methyl-9-alpha-fluoro-1,4-pregnadiene-11-beta,17-alpha,21-triol-3,20-dione; 16-alpha-Methyl-9-alpha-fluoro-11-beta,17-alpha,21-trihydroxypregna-1,4-diene-3,20-dione; 4-alpha-Fluoro-16-alpha-methyl-11-beta,17,21-trihydroxypregna-1,4-diene-3,20-dione; 9-alpha-Fluoro-16-alpha-methyl-1,4-pregnadiene-11-beta,17-alpha,21-triol-3,20-dione; DEXAMETHASONE [HSDB]; BIDD:PXR0060; BSPBio_000995; DEXAMETHASONE [VANDF]; MLS001055412; MLS001332508; BIDD:ER0494; Dexamethasone Sterile Solution; sustained-release dexamethasone; DEXAMETHASONE [WHO-DD]; DEXAMETHASONE [WHO-IP]; CHEMBL384467; GTPL2768; GTPL3447; SGCUT00126; 9-Fluoro-16-methylprednisolone; Ak Dex Oph Otic Soln 0.1%; DEXAMETHASONE [EMA EPAR]; BDBM18207; Dexamethasone (anti-inflammatory); Dexamethasone (JP17/USP/INN); 1p93; DEXAMETHASONE [GREEN BOOK]; HMS1792A17; HMS1990A17; HMS2089N13; HMS2235F08; HMS3039L11; HMS3259N11; HMS3403A17; Azium Aqueous Suspension Veterinary; DEXAMETHASONE [ORANGE BOOK]; AMY28815; to_000038; Tox21_200122; Dexamethasone, >=97.0% (HPLC); HB2521; HY-14648G; IBI-10090; MK-125; s1322; Dexamethasone 1.0 mg/ml in Methanol; DEXAMETHASONUM [WHO-IP LATIN]; PMS Dexamethasone Elixir 0.5mg/5ml; AKOS005259009; AKOS015895509; 1ST2218; CCG-264887; CS-1505; DB01234; KS-1451; NC00645; alpha -Fluoro-16-alpha -methylcortisol; CAS-50-02-2; Pregna-1,4-diene-3,20-dione, 9-fluoro-11beta,17,21-trihydroxy-16alpha-methyl-; SMP1_000092; Dexamethasone, >=98% (HPLC), powder; NCGC00091019-01; NCGC00091019-02; NCGC00091019-03; NCGC00091019-04; NCGC00091019-05; NCGC00091019-06; NCGC00091019-07; NCGC00091019-23; NCGC00257676-01; (11beta,16alpha)-9-Fluoro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione labeled with tritium; (8S,9R,10S,11S,13S,14S,16R,17R)-9-Fluoro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]ph; 23495-06-9; 9alpha-fluoro-16alpha-methyl-Prednisolone; AC-11056; DEXAMETHASONE COMPONENT OF CIPRODEX; DEXAMETHASONE COMPONENT OF MAXITROL; DEXAMETHASONE COMPONENT OF TOBRADEX; HY-14648; NCI60_003067; Pregna-1,4-diene-3,20-dione, 9-fluoro-11,17,21-trihydroxy-16-methyl-, labeled with tritium, (11beta,16alpha)-; SMR001227192; DEXAMETHASONE COMPONENT OF DEXACIDIN; Fluoro-9alpha Methyl-16alpha Prednisolone; 16alpha -Methyl-9alpha -fluoroprednisolone; 9alpha -Fluoro-16alpha -methylprednisolone; DEXAMETHASONE COMPONENT OF DEXASPORIN; Dexamethasone, tested according to Ph.Eur.; CS-0626118; DEXAMETHASONE COMPONENT OF TOBRADEX ST; NS00000344; EN300-52607; D00292; Dexamethasone, meets USP testing specifications; Prednisolone, 9alpha -fluoro-16alpha -methyl-; 9.alpha.-Fluoro-16.alpha.-methyl-1,20-dione; AB00918428-05; AB00918428-08; AB00918428-09; AB00918428_10; 16alpha -Methyl-9alpha -fluoro-1-dehydrocortisol; 16Alpha-methyl-9alpha-fluoro-delta1-hydrocortisone; Dexamethasone, VETRANAL(TM), analytical standard; Q422252; 1-Dehydro-16alpha-methyl-0alpha-fluorohydrocortisone; delta(Sup1)-9-alpha-fluoro-16-alpha-methylcortisol; Q-200939; BRD-K38775274-001-02-3; BRD-K38775274-001-06-4; DEXAMETHASONE ACETATE IMPURITY A [EP IMPURITY]; 1-Dehydro-16alpha -methyl-9alpha -fluorohydrocortisone; 9-Fluoro-11alpha -methylpregna-1,4-diene-3,20-dione; Dexamethasone, British Pharmacopoeia (BP) Assay Standard; Z756391748; Pregna-1,4-diene-3,20-dione, 9-fluoro-11alpha -methyl-; Dexamethasone, European Pharmacopoeia (EP) Reference Standard; WLN: L E5 B666 OV KU MUTJ A1 BF CQ E1 FV1Q FQ G1; Dexamethasone, powder, BioReagent, suitable for cell culture, >=97%; Dexamethasone, United States Pharmacopeia (USP) Reference Standard; 16-.alpha.-Methyl-9-.alpha.-fluoro-1,17-.alpha.,21-triol-3,20-dione; 4.alpha.-Fluoro-16.alpha.-methyl-11.beta.-17,4-diene-3,20-dione; Pregna-1,20-dione, 9-fluoro-11.beta.,17,21-trihydroxy-16.alpha.-methyl-; 16.alpha.-Methyl-9.alpha.-fluoro-11.beta.-17.alpha.-21-trihydroxypregna-1,20-dione; 9-Fluoro-11.beta.,21-trihydroxy-16.alpha.-methylpregna-1,4-diene-3,20-dione; 9-fluoro-16alpha-methyl-11beta,17,21-trihydroxypregna-1,4-diene-3,20-dione; 9.alpha.-Fluoro-11.beta.-17.alpha.- 21-trihydroxy-16.alpha.-methylpregna-1,20-dione; 9alpha-Fluoro-16alpha-methyl-11beta,17alpha,21-trihydroxy-1,4-pregnadiene-3,20-dione; Dexamethasone for peak identification, European Pharmacopoeia (EP) Reference Standard; Dexamethasone for system suitability, European Pharmacopoeia (EP) Reference Standard; Dexamethasone, Pharmaceutical Secondary Standard; Certified Reference Material; Dexamethasone, powder, gamma-irradiated, BioXtra, suitable for cell culture, >=80% (HPLC); Pregna-1,20-dione, 9-fluoro-11,17,21-trihydroxy-16-methyl-, (11.beta.,16.alpha.)-; (11.BETA.,16.ALPHA.)-9-FLUORO-11,17,21-TRIHYDROXY-16-METHYLPREGNA-1,4-DIENE-3,20-DIONE; (11alpha,14beta,16alpha,17alpha)-9-fluoro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dion
Click to Show/Hide
|
|||||
| Target(s) | Glucocorticoid receptor (NR3C1) | Target Info | ||||
| Structure |
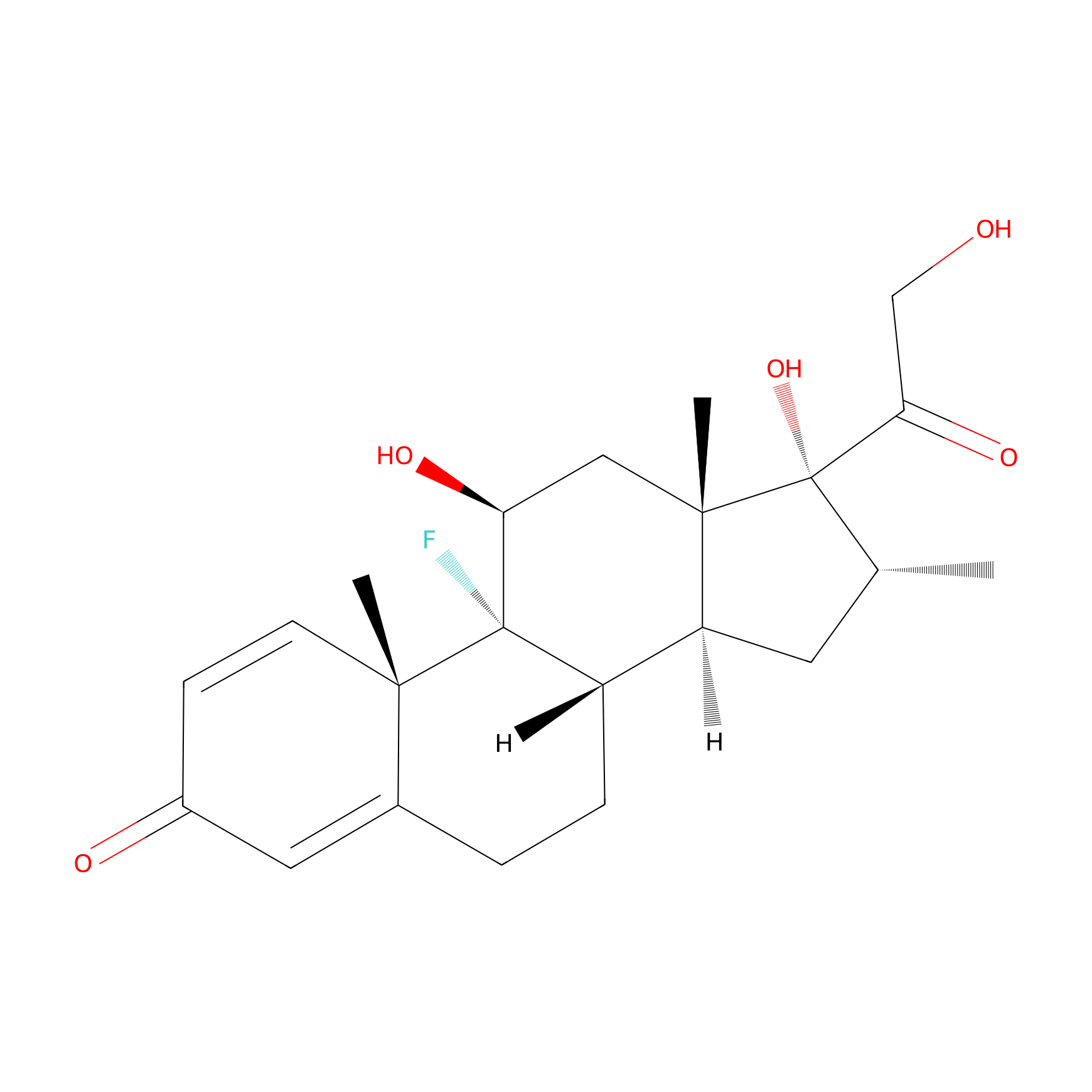
|
|||||
| Formula |
C22H29FO5
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 392.5 | ||||
| Lipid-water partition coefficient (xlogp) | 1.9 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | |||||
| Rotatable Bond Count (rotbonds) | 2 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC1CC2C3CCC4=CC(=O)C=CC4(C3(C(CC2(C1(C(=O)CO)O)C)O)F)C
|
|||||
| InChI |
InChI=1S/C22H29FO5/c1-12-8-16-15-5-4-13-9-14(25)6-7-19(13,2)21(15,23)17(26)10-20(16,3)22(12,28)18(27)11-24/h6-7,9,12,15-17,24,26,28H,4-5,8,10-11H2,1-3H3/t12-,15+,16+,17+,19+,20+,21+,22+/m1/s1
|
|||||
| InChIKey |
UREBDLICKHMUKA-CXSFZGCWSA-N
|
|||||
| IUPAC Name |
(8S,9R,10S,11S,13S,14S,16R,17R)-9-fluoro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-3-one
|
|||||
The activity data of This Drug
| Standard Type | Value | Administration times | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Minimum inhibitory concentration (MIC) | 64 µg/mL | 18 h | Klebsiella pneumoniae strain | 573 | [1] | |
| Minimum inhibitory concentration (MIC) | 64 µg/mL | 18 h | MDR Klebsiella pneumoniae strain | 573 | [1] | |
| Minimum inhibitory concentration (MIC) | 64 µg/mL | 18 h | Multiple-resistant staphylococcus aureus infection strain | 1280 | [1] | |
| Minimum inhibitory concentration (MIC) | 64 µg/mL | 18 h | Staphylococcus aureus infection strain | 1280 | [1] | |
| Minimum inhibitory concentration (MIC) | 64 µg/mL | 18 h | Pseudomonas aeruginosa strain | 287 | [1] | |
| Minimum inhibitory concentration (MIC) | 64 µg/mL | 18 h | Candida albicans fungus strains | 5476 | [1] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 91.1 uM | N.A. | J774 cell | CVCL_4692 | [2] | |
| Half Maximal Effective Concentration (EC50) | 17 nM | N.A. | HeLa cell | CVCL_0030 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 50 nM | N.A. | THP-1 cell | CVCL_0006 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.2 uM | N.A. | J774 cell | CVCL_4692 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 22 uM | N.A. | J774.A1 cell | CVCL_0358 | [6] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
Omi-hyd-Dex [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) | 8 µg/mL | |||
| Evaluation Method | MIC assay | ||||
| Administration Time | 18 h | ||||
| MOA of PDC |
To minimize the toxic side effect of omiganan and optimize its anti-inflammatory capability, we designed an IMEs-responsive self-delivery nanosystem consisting of omiganan, an anti-inflammatory agent, and natural polysaccharide to achieve on-demand degradation and responsive drug release. Based on the cationic hydrophilic fragments on omiganan, we chose dexamethasone (Dex, a hydrophobic anti-inflammatory drug) to link with this peptide via a hydrazone bond to construct an amphiphilic conjugate (Omi-hyd-Dex). With the assistance of PLGA, this conjugate could self-assemble into nanoparticles (Omi-hyd-Dex NPs) in an aqueous solution without introducing any other hazardous materials. Then, the negatively-charged hyaluronic acid (HA, a natural ligand of ICAM-1 and CD44) was used to coat Omi-hyd-Dex NPs to form a core-shell structural formulation (Omi-hyd-Dex@HA NPs). This HA coating could not only eliminate the hemolytic activity of omiganan to reduce side effects but also act as the IMEs targeting molecule through interaction with intercellular adhesion molecule-1 (ICAM-1) on inflamed endothelial cells. After entering IMEs, the HA coating would be degraded and detached to expose the cationic surface of the Omi-hyd-Dex core and enable it to accumulate in IMEs. Meanwhile, the hydrazone bond between omiganan and Dex could be cleaved in response to the acidic condition of IMEs, thereby releasing the cationic peptide and anti-inflammatory agent that would concurrently inhibit the infection and inflammation precisely.
Click to Show/Hide
|
||||
| Description |
To verify whether Omi-hyd-Dex@HA NPs inherited the antimicrobial activity of omiganan, the MICs were tested by the standardized agar doubling-dilution method as shown in Table 1. Omi-hyd-Dex@HA NPs and free omiganan showed similar extended broad-spectrum antibacterial activity for G+ bacteria, G- bacteria and fungi pathogens, including drug-sensitive strains (S. aureus, P. aeruginosa, K.P., Candida albicans) and drug-resistant strains (MRSA, MDR-K.P.). The MICs of Omi-hyd-Dex@HA NPs showed none or only a one-fold increase compared with the free Omiganan-NHNH2 group. In addition, free Dex did not show any antimicrobial activity for the tested pathogens, indicating that Dex moieties of Omi-hyd-Dex@HA NPs did not contribute to the bacterial killing effect. In contrast, vancomycin, as a standard drug regimen for drug-resistant G+ strains, showed inefficacious for G- strains and fungus. Similar results were also observed in oxacillin-treated groups. Therefore, Omi-hyd-Dex@HA NPs retained the broad-spectrum antimicrobial activity of omiganan.
Click to Show/Hide
|
||||
| In Vitro Model | Staphylococcus aureus infection | Staphylococcus aureus infection strain | 1280 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) | 16 µg/mL | |||
| Evaluation Method | MIC assay | ||||
| Administration Time | 18 h | ||||
| MOA of PDC |
To minimize the toxic side effect of omiganan and optimize its anti-inflammatory capability, we designed an IMEs-responsive self-delivery nanosystem consisting of omiganan, an anti-inflammatory agent, and natural polysaccharide to achieve on-demand degradation and responsive drug release. Based on the cationic hydrophilic fragments on omiganan, we chose dexamethasone (Dex, a hydrophobic anti-inflammatory drug) to link with this peptide via a hydrazone bond to construct an amphiphilic conjugate (Omi-hyd-Dex). With the assistance of PLGA, this conjugate could self-assemble into nanoparticles (Omi-hyd-Dex NPs) in an aqueous solution without introducing any other hazardous materials. Then, the negatively-charged hyaluronic acid (HA, a natural ligand of ICAM-1 and CD44) was used to coat Omi-hyd-Dex NPs to form a core-shell structural formulation (Omi-hyd-Dex@HA NPs). This HA coating could not only eliminate the hemolytic activity of omiganan to reduce side effects but also act as the IMEs targeting molecule through interaction with intercellular adhesion molecule-1 (ICAM-1) on inflamed endothelial cells. After entering IMEs, the HA coating would be degraded and detached to expose the cationic surface of the Omi-hyd-Dex core and enable it to accumulate in IMEs. Meanwhile, the hydrazone bond between omiganan and Dex could be cleaved in response to the acidic condition of IMEs, thereby releasing the cationic peptide and anti-inflammatory agent that would concurrently inhibit the infection and inflammation precisely.
Click to Show/Hide
|
||||
| Description |
To verify whether Omi-hyd-Dex@HA NPs inherited the antimicrobial activity of omiganan, the MICs were tested by the standardized agar doubling-dilution method as shown in Table 1. Omi-hyd-Dex@HA NPs and free omiganan showed similar extended broad-spectrum antibacterial activity for G+ bacteria, G- bacteria and fungi pathogens, including drug-sensitive strains (S. aureus, P. aeruginosa, K.P., Candida albicans) and drug-resistant strains (MRSA, MDR-K.P.). The MICs of Omi-hyd-Dex@HA NPs showed none or only a one-fold increase compared with the free Omiganan-NHNH2 group. In addition, free Dex did not show any antimicrobial activity for the tested pathogens, indicating that Dex moieties of Omi-hyd-Dex@HA NPs did not contribute to the bacterial killing effect. In contrast, vancomycin, as a standard drug regimen for drug-resistant G+ strains, showed inefficacious for G- strains and fungus. Similar results were also observed in oxacillin-treated groups. Therefore, Omi-hyd-Dex@HA NPs retained the broad-spectrum antimicrobial activity of omiganan.
Click to Show/Hide
|
||||
| In Vitro Model | Multiple-resistant Staphylococcus aureus infection | Multiple-resistant staphylococcus aureus infection strain | 1280 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) | 16 µg/mL | |||
| Evaluation Method | MIC assay | ||||
| Administration Time | 18 h | ||||
| MOA of PDC |
To minimize the toxic side effect of omiganan and optimize its anti-inflammatory capability, we designed an IMEs-responsive self-delivery nanosystem consisting of omiganan, an anti-inflammatory agent, and natural polysaccharide to achieve on-demand degradation and responsive drug release. Based on the cationic hydrophilic fragments on omiganan, we chose dexamethasone (Dex, a hydrophobic anti-inflammatory drug) to link with this peptide via a hydrazone bond to construct an amphiphilic conjugate (Omi-hyd-Dex). With the assistance of PLGA, this conjugate could self-assemble into nanoparticles (Omi-hyd-Dex NPs) in an aqueous solution without introducing any other hazardous materials. Then, the negatively-charged hyaluronic acid (HA, a natural ligand of ICAM-1 and CD44) was used to coat Omi-hyd-Dex NPs to form a core-shell structural formulation (Omi-hyd-Dex@HA NPs). This HA coating could not only eliminate the hemolytic activity of omiganan to reduce side effects but also act as the IMEs targeting molecule through interaction with intercellular adhesion molecule-1 (ICAM-1) on inflamed endothelial cells. After entering IMEs, the HA coating would be degraded and detached to expose the cationic surface of the Omi-hyd-Dex core and enable it to accumulate in IMEs. Meanwhile, the hydrazone bond between omiganan and Dex could be cleaved in response to the acidic condition of IMEs, thereby releasing the cationic peptide and anti-inflammatory agent that would concurrently inhibit the infection and inflammation precisely.
Click to Show/Hide
|
||||
| Description |
To verify whether Omi-hyd-Dex@HA NPs inherited the antimicrobial activity of omiganan, the MICs were tested by the standardized agar doubling-dilution method as shown in Table 1. Omi-hyd-Dex@HA NPs and free omiganan showed similar extended broad-spectrum antibacterial activity for G+ bacteria, G- bacteria and fungi pathogens, including drug-sensitive strains (S. aureus, P. aeruginosa, K.P., Candida albicans) and drug-resistant strains (MRSA, MDR-K.P.). The MICs of Omi-hyd-Dex@HA NPs showed none or only a one-fold increase compared with the free Omiganan-NHNH2 group. In addition, free Dex did not show any antimicrobial activity for the tested pathogens, indicating that Dex moieties of Omi-hyd-Dex@HA NPs did not contribute to the bacterial killing effect. In contrast, vancomycin, as a standard drug regimen for drug-resistant G+ strains, showed inefficacious for G- strains and fungus. Similar results were also observed in oxacillin-treated groups. Therefore, Omi-hyd-Dex@HA NPs retained the broad-spectrum antimicrobial activity of omiganan.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae strain | 573 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) | 16 µg/mL | |||
| Evaluation Method | MIC assay | ||||
| Administration Time | 18 h | ||||
| MOA of PDC |
To minimize the toxic side effect of omiganan and optimize its anti-inflammatory capability, we designed an IMEs-responsive self-delivery nanosystem consisting of omiganan, an anti-inflammatory agent, and natural polysaccharide to achieve on-demand degradation and responsive drug release. Based on the cationic hydrophilic fragments on omiganan, we chose dexamethasone (Dex, a hydrophobic anti-inflammatory drug) to link with this peptide via a hydrazone bond to construct an amphiphilic conjugate (Omi-hyd-Dex). With the assistance of PLGA, this conjugate could self-assemble into nanoparticles (Omi-hyd-Dex NPs) in an aqueous solution without introducing any other hazardous materials. Then, the negatively-charged hyaluronic acid (HA, a natural ligand of ICAM-1 and CD44) was used to coat Omi-hyd-Dex NPs to form a core-shell structural formulation (Omi-hyd-Dex@HA NPs). This HA coating could not only eliminate the hemolytic activity of omiganan to reduce side effects but also act as the IMEs targeting molecule through interaction with intercellular adhesion molecule-1 (ICAM-1) on inflamed endothelial cells. After entering IMEs, the HA coating would be degraded and detached to expose the cationic surface of the Omi-hyd-Dex core and enable it to accumulate in IMEs. Meanwhile, the hydrazone bond between omiganan and Dex could be cleaved in response to the acidic condition of IMEs, thereby releasing the cationic peptide and anti-inflammatory agent that would concurrently inhibit the infection and inflammation precisely.
Click to Show/Hide
|
||||
| Description |
To verify whether Omi-hyd-Dex@HA NPs inherited the antimicrobial activity of omiganan, the MICs were tested by the standardized agar doubling-dilution method as shown in Table 1. Omi-hyd-Dex@HA NPs and free omiganan showed similar extended broad-spectrum antibacterial activity for G+ bacteria, G- bacteria and fungi pathogens, including drug-sensitive strains (S. aureus, P. aeruginosa, K.P., Candida albicans) and drug-resistant strains (MRSA, MDR-K.P.). The MICs of Omi-hyd-Dex@HA NPs showed none or only a one-fold increase compared with the free Omiganan-NHNH2 group. In addition, free Dex did not show any antimicrobial activity for the tested pathogens, indicating that Dex moieties of Omi-hyd-Dex@HA NPs did not contribute to the bacterial killing effect. In contrast, vancomycin, as a standard drug regimen for drug-resistant G+ strains, showed inefficacious for G- strains and fungus. Similar results were also observed in oxacillin-treated groups. Therefore, Omi-hyd-Dex@HA NPs retained the broad-spectrum antimicrobial activity of omiganan.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | MDR Klebsiella pneumoniae strain | 573 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) | 32 µg/mL | |||
| Evaluation Method | MIC assay | ||||
| Administration Time | 18 h | ||||
| MOA of PDC |
To minimize the toxic side effect of omiganan and optimize its anti-inflammatory capability, we designed an IMEs-responsive self-delivery nanosystem consisting of omiganan, an anti-inflammatory agent, and natural polysaccharide to achieve on-demand degradation and responsive drug release. Based on the cationic hydrophilic fragments on omiganan, we chose dexamethasone (Dex, a hydrophobic anti-inflammatory drug) to link with this peptide via a hydrazone bond to construct an amphiphilic conjugate (Omi-hyd-Dex). With the assistance of PLGA, this conjugate could self-assemble into nanoparticles (Omi-hyd-Dex NPs) in an aqueous solution without introducing any other hazardous materials. Then, the negatively-charged hyaluronic acid (HA, a natural ligand of ICAM-1 and CD44) was used to coat Omi-hyd-Dex NPs to form a core-shell structural formulation (Omi-hyd-Dex@HA NPs). This HA coating could not only eliminate the hemolytic activity of omiganan to reduce side effects but also act as the IMEs targeting molecule through interaction with intercellular adhesion molecule-1 (ICAM-1) on inflamed endothelial cells. After entering IMEs, the HA coating would be degraded and detached to expose the cationic surface of the Omi-hyd-Dex core and enable it to accumulate in IMEs. Meanwhile, the hydrazone bond between omiganan and Dex could be cleaved in response to the acidic condition of IMEs, thereby releasing the cationic peptide and anti-inflammatory agent that would concurrently inhibit the infection and inflammation precisely.
Click to Show/Hide
|
||||
| Description |
To verify whether Omi-hyd-Dex@HA NPs inherited the antimicrobial activity of omiganan, the MICs were tested by the standardized agar doubling-dilution method as shown in Table 1. Omi-hyd-Dex@HA NPs and free omiganan showed similar extended broad-spectrum antibacterial activity for G+ bacteria, G- bacteria and fungi pathogens, including drug-sensitive strains (S. aureus, P. aeruginosa, K.P., Candida albicans) and drug-resistant strains (MRSA, MDR-K.P.). The MICs of Omi-hyd-Dex@HA NPs showed none or only a one-fold increase compared with the free Omiganan-NHNH2 group. In addition, free Dex did not show any antimicrobial activity for the tested pathogens, indicating that Dex moieties of Omi-hyd-Dex@HA NPs did not contribute to the bacterial killing effect. In contrast, vancomycin, as a standard drug regimen for drug-resistant G+ strains, showed inefficacious for G- strains and fungus. Similar results were also observed in oxacillin-treated groups. Therefore, Omi-hyd-Dex@HA NPs retained the broad-spectrum antimicrobial activity of omiganan.
Click to Show/Hide
|
||||
| In Vitro Model | Pseudomonas aeruginosa strain infection | Pseudomonas aeruginosa strain | 287 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bacterial infection | ||||
| Efficacy Data | Minimum inhibitory concentration (MIC) | 32 µg/mL | |||
| Evaluation Method | MIC assay | ||||
| Administration Time | 18 h | ||||
| MOA of PDC |
To minimize the toxic side effect of omiganan and optimize its anti-inflammatory capability, we designed an IMEs-responsive self-delivery nanosystem consisting of omiganan, an anti-inflammatory agent, and natural polysaccharide to achieve on-demand degradation and responsive drug release. Based on the cationic hydrophilic fragments on omiganan, we chose dexamethasone (Dex, a hydrophobic anti-inflammatory drug) to link with this peptide via a hydrazone bond to construct an amphiphilic conjugate (Omi-hyd-Dex). With the assistance of PLGA, this conjugate could self-assemble into nanoparticles (Omi-hyd-Dex NPs) in an aqueous solution without introducing any other hazardous materials. Then, the negatively-charged hyaluronic acid (HA, a natural ligand of ICAM-1 and CD44) was used to coat Omi-hyd-Dex NPs to form a core-shell structural formulation (Omi-hyd-Dex@HA NPs). This HA coating could not only eliminate the hemolytic activity of omiganan to reduce side effects but also act as the IMEs targeting molecule through interaction with intercellular adhesion molecule-1 (ICAM-1) on inflamed endothelial cells. After entering IMEs, the HA coating would be degraded and detached to expose the cationic surface of the Omi-hyd-Dex core and enable it to accumulate in IMEs. Meanwhile, the hydrazone bond between omiganan and Dex could be cleaved in response to the acidic condition of IMEs, thereby releasing the cationic peptide and anti-inflammatory agent that would concurrently inhibit the infection and inflammation precisely.
Click to Show/Hide
|
||||
| Description |
To verify whether Omi-hyd-Dex@HA NPs inherited the antimicrobial activity of omiganan, the MICs were tested by the standardized agar doubling-dilution method as shown in Table 1. Omi-hyd-Dex@HA NPs and free omiganan showed similar extended broad-spectrum antibacterial activity for G+ bacteria, G- bacteria and fungi pathogens, including drug-sensitive strains (S. aureus, P. aeruginosa, K.P., Candida albicans) and drug-resistant strains (MRSA, MDR-K.P.). The MICs of Omi-hyd-Dex@HA NPs showed none or only a one-fold increase compared with the free Omiganan-NHNH2 group. In addition, free Dex did not show any antimicrobial activity for the tested pathogens, indicating that Dex moieties of Omi-hyd-Dex@HA NPs did not contribute to the bacterial killing effect. In contrast, vancomycin, as a standard drug regimen for drug-resistant G+ strains, showed inefficacious for G- strains and fungus. Similar results were also observed in oxacillin-treated groups. Therefore, Omi-hyd-Dex@HA NPs retained the broad-spectrum antimicrobial activity of omiganan.
Click to Show/Hide
|
||||
| In Vitro Model | Candida albicans infection | Candida albicans fungus strains | 5476 | ||
References
