Drug Information
General Information of This Drug
| Drug ID | DRG00018 | |||||
|---|---|---|---|---|---|---|
| Drug Name | 7-Ethyl-10-hydroxycamptothecin | |||||
| Synonyms |
7-Ethyl-10-hydroxycamptothecin; 86639-52-3; SN-38; SN 38 lactone; SN 38; 7-Ethyl-10-hydroxy-camptothecin; (S)-4,11-Diethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; 10-Hydroxy-7-ethylcamptothecin; SN38; 7-Ethyl-10-hydroxy-20(S)-camptothecin; NK 012; NK-012; NK012; CHEBI:8988; 113015-38-6; IT-141; NSC673596; 0H43101T0J; (19S)-10,19-diethyl-7,19-dihydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2,4(9),5,7,10,15(20)-heptaene-14,18-dione; (4S)-4,11-diethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione; CAMPTOTHECIN, 7-ETHYL-10-HYDROXY-; (4S)-4,11-Diethyl-4,9-dihydroxy-1H-pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione; 7-ethyl-10-hydroxy-20(s)-campthothecin; LE-SN38; Captothecin, 7-ethyl-10-hydroxy-; MFCD00871873; UNII-0H43101T0J; C22H20N2O5; 110714-48-2; 1H-PYRANO(3',4':6,7)INDOLIZINO(1,2-B)QUINOLINE-3,14(4H,12H)-DIONE, 4,11-DIETHYL-4,9-DIHYDROXY-, (4S)-; 1H-Pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione, 4,11-diethyl-4,9-dihydroxy-, (4S)-; AvaChem1025; (+)-7-ETHYL-10-HYDROXYCAMPTOTHECIN; diethyl(dihydroxy)[?]dione; SN 38- Bio-X; SCHEMBL34018; GTPL6925; SN 38 [WHO-DD]; DTXSID4040399; 10-hydroxy-7-ethyl camptothecin; 10-hydroxy-7-ethyl-camptothecin; EX-A989; HMS3413B12; HMS3652P12; HMS3677B12; BCP01386; 7-Ethyl-10-hydroxy-20(S)-CPT; BDBM50418088; s4908; AKOS015920433; AC-1357; BCP9000200; CCG-264764; CS-1579; DB05482; NSC-673596; SN-38(NK-012)?; NCGC00167831-01; NCGC00167831-05; AS-13533; BE164132; BP-24513; HY-13704; NCI60_026056; 7-Ethyl-10-hydroxy-20-(S)-camptothecine; E0748; FT-0630943; FT-0674607; FT-0674608; NS00069821; SW219948-1; S-(+)-7-ETHYL-10-HYDROXYCAMPOTHECIN; EN300-122379; NK012 , SN 38; A857464; Q-100871; Q1750127; 7-Ethyl-10-hydroxycamptothecin, >=98% (HPLC), powder; Z1541759909; (19S)-10,19-diethyl-7,19-dihydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.0^{2,11}.0^{4,9}.0^{15,20}]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaene-14,18-dione; (19S)-10,19-diethyl-7,19-dihydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaene-14,18-dione; (4S)-4,11-Diethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)dione, AldrichCPR; (4S)-4,9-Dihydroxy-4,11-diethyl-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione;SN-38; (S)-4,11-Diethyl-4,9-di-OH-1,12-dihydro-4H-2-oxa-6,12a-diaza-dibenzo[b,h]fluorene-3,13-dione; 1H-Pyrano[3',7]indolizino[1,2-b]quinoline- 3,14(4H,12H)-dione, 4,11-diethyl-4,9-dihydroxy-, (4S)-; 7-Ethyl-10-hydroxycamptothecin ((S)-4,11-Diethyl-4,9-dihydroxy-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione); H-Pyrano(3',4':6,7)indolizino(1,2-b)quinoline-3,14(4H,12H)-dione, 4,11-diethyl-4,9-dihydroxy-, (S)-; RS4
Click to Show/Hide
|
|||||
| Target(s) | DNA topoisomerase 1 (TOP1) | Target Info | ||||
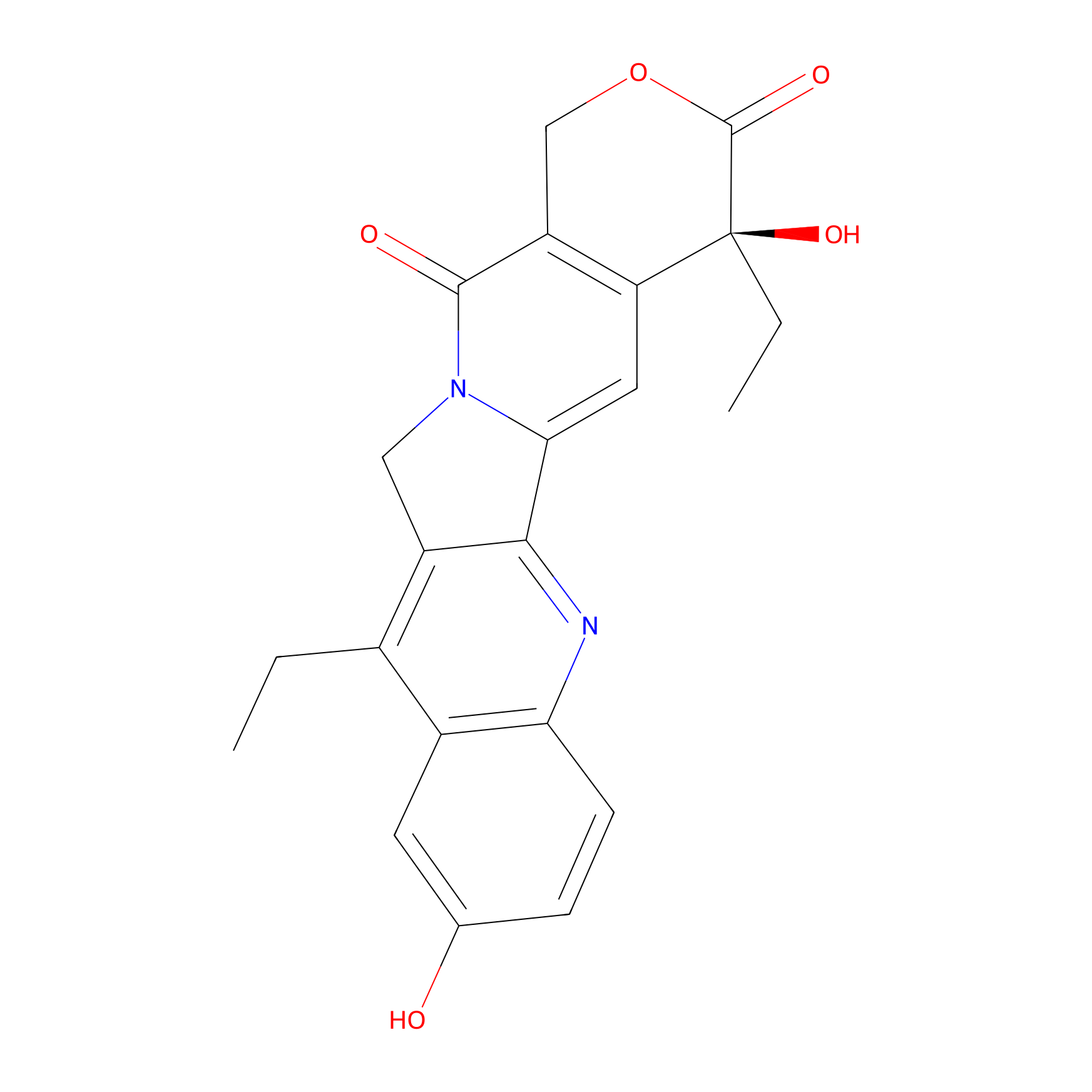
| Structure |
|
|||||
| Formula |
C22H20N2O5
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 392.4 | ||||
| Lipid-water partition coefficient (xlogp) | 1.4 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | |||||
| Rotatable Bond Count (rotbonds) | 2 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CCC1=C2CN3C(=CC4=C(C3=O)COC(=O)C4(CC)O)C2=NC5=C1C=C(C=C5)O
|
|||||
| InChI |
InChI=1S/C22H20N2O5/c1-3-12-13-7-11(25)5-6-17(13)23-19-14(12)9-24-18(19)8-16-15(20(24)26)10-29-21(27)22(16,28)4-2/h5-8,25,28H,3-4,9-10H2,1-2H3/t22-/m0/s1
|
|||||
| InChIKey |
FJHBVJOVLFPMQE-QFIPXVFZSA-N
|
|||||
| IUPAC Name |
(19S)-10,19-diethyl-7,19-dihydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2,4(9),5,7,10,15(20)-heptaene-14,18-dione
|
|||||
The activity data of This Drug
| Standard Type | Value | Administration times | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | 26.41 nM | 72 h | U-87MG cell | CVCL_0022 | [1] | |
| Half Maximal Growth Inhibition (GI50) | 3 nM | N.A. | MCF-7 cell | CVCL_0031 | [2] | |
| Half Maximal Growth Inhibition (GI50) | 23 nM | N.A. | HCT 116 cell | CVCL_0291 | [2] | |
| Half Maximal Growth Inhibition (GI50) | 110 nM | N.A. | MKN-28 cell | CVCL_1416 | [2] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.43 nM | N.A. | PC-6 cell | CVCL_C002 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.55 nM | N.A. | HCT 116 cell | CVCL_0291 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.78 nM | N.A. | HCT 116 cell | CVCL_0291 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.8 nM | N.A. | QG-56 cell | CVCL_6943 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.3 nM | N.A. | NCI-H460 cell | CVCL_0459 | [3] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | N.A. | DU145 cell | CVCL_0105 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.9 nM | N.A. | HT29 cell | CVCL_A8EZ | [6] | |
| Half Maximal Inhibitory Concentration (IC50) | 6 nM | N.A. | NCI-H128 cell | CVCL_1460 | [7] | |
| Half Maximal Inhibitory Concentration (IC50) | 8 nM | N.A. | Capan-1 cell | CVCL_0237 | [8] | |
| Half Maximal Inhibitory Concentration (IC50) | 8 nM | N.A. | MKN45 cell | CVCL_0434 | [7] | |
| Half Maximal Inhibitory Concentration (IC50) | 8.9 nM | N.A. | L1210 cell | CVCL_0382 | [9] | |
| Half Maximal Inhibitory Concentration (IC50) | 11 nM | N.A. | A-549 cell | CVCL_0023 | [8] | |
| Half Maximal Inhibitory Concentration (IC50) | 16 nM | N.A. | A-549 cell | CVCL_0023 | [7] | |
| Half Maximal Inhibitory Concentration (IC50) | 18 nM | N.A. | A-549 cell | CVCL_0023 | [10] | |
| Half Maximal Inhibitory Concentration (IC50) | 28 nM | N.A. | A-549 cell | CVCL_0023 | [11] | |
| Half Maximal Inhibitory Concentration (IC50) | 35.3 nM | N.A. | PC-3 cell | CVCL_0035 | [12] | |
| Half Maximal Inhibitory Concentration (IC50) | 54 nM | N.A. | HT29 cell | CVCL_A8EZ | [13] | |
| Half Maximal Inhibitory Concentration (IC50) | 88 nM | N.A. | A-549 cell | CVCL_0023 | [14] | |
| Half Maximal Inhibitory Concentration (IC50) | 220 nM | N.A. | NCI-H460 cell | CVCL_0459 | [15] | |
| Half Maximal Inhibitory Concentration (IC50) | 300 nM | N.A. | A-549 cell | CVCL_0023 | [16] | |
| Half Maximal Inhibitory Concentration (IC50) | 300 nM | N.A. | HeLa cell | CVCL_0030 | [16] | |
| Half Maximal Inhibitory Concentration (IC50) | 370 nM | N.A. | MCF-7 cell | CVCL_0031 | [17] | |
| Half Maximal Inhibitory Concentration (IC50) | 480 nM | N.A. | SK-MEL-24 cell | CVCL_0599 | [18] | |
| Half Maximal Inhibitory Concentration (IC50) | 4.98 uM | N.A. | CCD-841CoN cell | CVCL_2871 | [11] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
SN38-HKD [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [19] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 43% (Day 14) | |||
| Evaluation Method | Tumor volume detection assay | ||||
| Administration Time | 4-injection regimen (on day 0, 2, 4, and 6) | ||||
| Administration Dosage | 10 mg/kg | ||||
| MOA of PDC |
N38-HKD increases infiltration, activity, and viability of CD8+T cells, and thus inhibits the growth of primary tumors and pulmonary metastasis. This study highlights the synergistic modulation of cancerous cells and TECs with integrin-targeting PDC filaments as a promising strategy for TNBC chemoimmunotherapy.
|
||||
| Description |
SN38-HKD/RGDR slowed the 4T1 tumor growth by 74%, while irinotecan and SN38-HKD only showed 28% and 43% tumor growth inhibition, respectively, which was further confirmed by its capability in reducing tumor burden.
|
||||
| In Vivo Model | 4T1 tumor-bearing mice. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [19] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 40% (Day 18) | |||
| Evaluation Method | Tumor volume detection assay | ||||
| Administration Time | 4-injection regimen (on day 0, 2, 4, and 6) | ||||
| Administration Dosage | 10 mg/kg | ||||
| MOA of PDC |
N38-HKD increases infiltration, activity, and viability of CD8+T cells, and thus inhibits the growth of primary tumors and pulmonary metastasis. This study highlights the synergistic modulation of cancerous cells and TECs with integrin-targeting PDC filaments as a promising strategy for TNBC chemoimmunotherapy.
|
||||
| Description |
SN38-HKD/RGDR slowed the 4T1 tumor growth by 74%, while irinotecan and SN38-HKD only showed 28% and 43% tumor growth inhibition, respectively, which was further confirmed by its capability in reducing tumor burden.
|
||||
| In Vivo Model | EMT6 tumor-bearing mice. | ||||
| In Vitro Model | Mammary gland malignant neoplasms | EMT6 cell | CVCL_1923 | ||
SN38-HKD/RGDR [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [19] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 74% (Day 14) | |||
| Evaluation Method | Tumor volume detection assay | ||||
| Administration Time | 4-injection regimen (on day 0, 2, 4, and 6) | ||||
| Administration Dosage | 10 mg/kg | ||||
| MOA of PDC |
N38-HKD/RGDR increases infiltration, activity, and viability of CD8+ T cells, and thus inhibits the growth of primary tumors and pulmonary metastasis. This study highlights the synergistic modulation of cancerous cells and TECs with integrin-targeting PDC filaments as a promising strategy for TNBC chemoimmunotherapy.
|
||||
| Description |
SN38-HKD/RGDR slowed the 4T1 tumor growth by 74%, while irinotecan and SN38-HKD only showed 28% and 43% tumor growth inhibition, respectively, which was further confirmed by its capability in reducing tumor burden.
|
||||
| In Vivo Model | 4T1 tumor-bearing mice. | ||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [19] | ||||
| Indication | Triple-negative breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 75% (Day 18) | |||
| Evaluation Method | Tumor volume detection assay | ||||
| Administration Time | 4-injection regimen (on day 0, 2, 4, and 6) | ||||
| Administration Dosage | 10 mg/kg | ||||
| MOA of PDC |
N38-HKD/RGDR increases infiltration, activity, and viability of CD8+T cells, and thus inhibits the growth of primary tumors and pulmonary metastasis. This study highlights the synergistic modulation of cancerous cells and TECs with integrin-targeting PDC filaments as a promising strategy for TNBC chemoimmunotherapy.
|
||||
| Description |
SN38-HKD/RGDR slowed the 4T1 tumor growth by 74%, while irinotecan and SN38-HKD only showed 28% and 43% tumor growth inhibition, respectively, which was further confirmed by its capability in reducing tumor burden.
|
||||
| In Vivo Model | EMT6 tumor-bearing mice. | ||||
| In Vitro Model | Mammary gland malignant neoplasms | EMT6 cell | CVCL_1923 | ||
Cot-APTEDB-SN38 [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [20] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Anti-tumor activity | 23.90% | |||
| Administration Time | 6 days | ||||
| Administration Dosage | Equivalent to 2 mg SN38/kg | ||||
| Description |
In situ HC[cot-APTEDB-SN38/Abcot] at an SN38/kg dose-equivalent of 2 mg effectively suppressed tumor growth and showed much greater antitumor activity (49.8% inhibition) than both cot-APTEDB-SN38 alone (23.9% inhibition) and CPT-11 (10.6% inhibition).
|
||||
| In Vivo Model | EDB-positive human glioblastoma-bearing mice. | ||||
| Half life period | 2.01 h | ||||
T7-SN-38 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 70.07nM | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
In the present work, the cytotoxic drug SN-38 is coupled to the tumor-targeting T7 peptide via a cathepsin B cleavable VA peptide linker. This ensures that the drug remains covalently bound until it reaches the intended site of action, where Cat B is overexpressed to release the drug. Within this framework, our research pursuits entail the synthesis and characterization of a T7-SN-38-targeted drug conjugate using strain-promoted azide-alkyne cycloaddition (SPAAC). Our investigation extended to evaluating the cellular uptake and assessing the cytotoxicity of the drug conjugate in U87MG glioblastoma cells.
Click to Show/Hide
|
||||
| Description |
Further, IC50values of SN-38 and T7-SN-38 on U87MG cells at 72 h were determined. An estimated IC50value of 26.41nM was obtained for SN-38, which was considerably lower than an IC50value of 70.07nM obtained for the T7-SN-38 conjugate. These IC50data confirm the greater cytotoxicity of the pure drug compared to the conjugate at 72 h (p< 0.05).
|
||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
References
