Drug Information
General Information of This Drug
| Drug ID | DRG00021 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Exatecan | |||||
| Synonyms |
Exatecan; 171335-80-1; Exatecan [INN]; DX-8951; DX-8951f; OC71PP0F89; (1S,9S)-1-Amino-9-ethyl-5-fluoro-1,2,3,9,12,15-hexahydro-9-hydroxy-4-methyl-10H,13H-benzo(de)pyrano(3',4':6,7)indolizino(1,2-b)quinoline-10,13-dione; Dx 8951; (10S,23S)-23-amino-10-ethyl-18-fluoro-10-hydroxy-19-methyl-8-oxa-4,15-diazahexacyclo[14.7.1.02,14.04,13.06,11.020,24]tetracosa-1,6(11),12,14,16,18,20(24)-heptaene-5,9-dione; (1S,9S)-1-amino-9-ethyl-5-fluoro-9-hydroxy-4-methyl-2,3,12,15-tetrahydrobenzo[de]pyrano[3',4':6,7]indolizino[1,2-b]quinoline-10,13(1H,9H)-dione; C24H22FN3O4; UNII-OC71PP0F89; exatecan-mesylate; (1s,9s)-1-amino-9-ethyl-5-fluoro-1,2,3,9,12,15-hexahydro-9-hydroxy-4-methyl-10h,13h-benzo[de]pyrano[3',4':6,7]indolizino[1,2-b]quinoline-10,13-dione; EXATECAN [MI]; EXATECAN [WHO-DD]; SCHEMBL2512959; CHEMBL1614650; DTXSID60169061; CHEBI:135709; GLXC-27628; EX-A2683; NSC829066; AKOS005146469; AT33978; BCP9000674; DB12185; NSC-829066; 10H,13H-Benzo(de)pyrano(3',4':6,7)indolizino(1,2-b)quinoline-10,13-dione, 1-amino-9-ethyl-5-fluoro-1,2,3,9,12,15-hexahydro-9-hydroxy-4-methyl-, (1S,9S)-; 10H,13H-Benzo(de)pyrano(3',4':6,7)indolizino(1,2-b)quinoline-10,13-dione, 1-amino-9-ethyl-5-fluoro-1,2,3,9,12,15-hexahydro-9-hydroxy-4-methyl-, (1S-trans)-; AC-32495; BP-27995; BP-27996; HY-13631; TS-07806; DB-064817; NS00069222; J-521361; Q5419343
Click to Show/Hide
|
|||||
| Target(s) | DNA topoisomerase 1 (TOP1) | Target Info | ||||
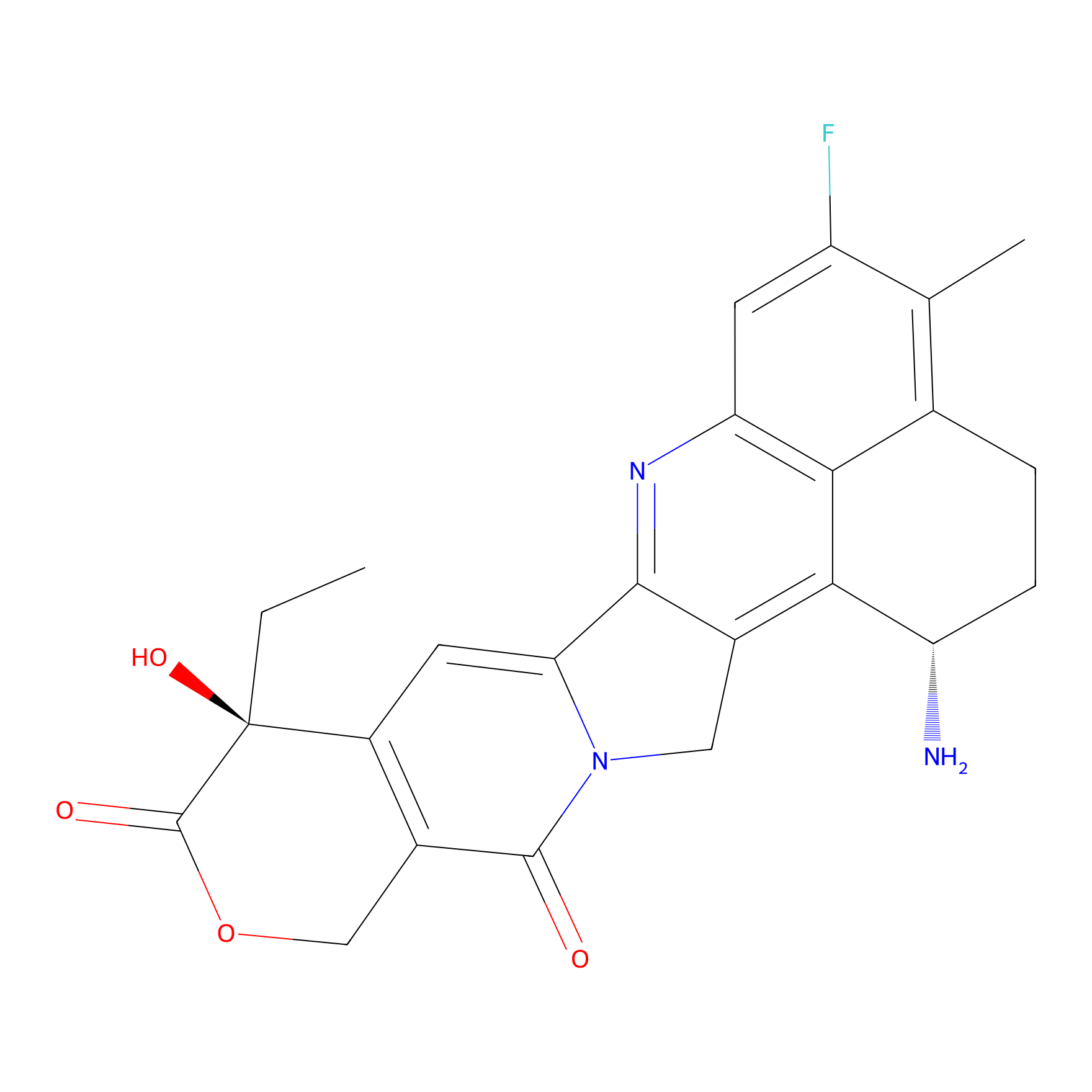
| Structure |
|
|||||
| Formula |
C24H22FN3O4
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 435.4 | ||||
| Lipid-water partition coefficient (xlogp) | 0.4 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 7 | |||||
| Rotatable Bond Count (rotbonds) | 1 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CCC1(C2=C(COC1=O)C(=O)N3CC4=C5C(CCC6=C5C(=CC(=C6C)F)N=C4C3=C2)N)O
|
|||||
| InChI |
InChI=1S/C24H22FN3O4/c1-3-24(31)14-6-18-21-12(8-28(18)22(29)13(14)9-32-23(24)30)19-16(26)5-4-11-10(2)15(25)7-17(27-21)20(11)19/h6-7,16,31H,3-5,8-9,26H2,1-2H3/t16-,24-/m0/s1
|
|||||
| InChIKey |
ZVYVPGLRVWUPMP-FYSMJZIKSA-N
|
|||||
| IUPAC Name |
(10S,23S)-23-amino-10-ethyl-18-fluoro-10-hydroxy-19-methyl-8-oxa-4,15-diazahexacyclo[14.7.1.02,14.04,13.06,11.020,24]tetracosa-1,6(11),12,14,16,18,20(24)-heptaene-5,9-dione
|
|||||
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
CBX-12 [Phase 2]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 58.10% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | 23 days | ||||
| Administration Dosage | 5 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 76.50% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | 35 days | ||||
| Administration Dosage | 10 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 81.40% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | 23 days | ||||
| Administration Dosage | 5 mg/kg with ceralasertib 25 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 95% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | 35 days | ||||
| Administration Dosage | 10 mg/kg with ceralasertib 25 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate | 60 mm3 | |||
| Administration Time | 45 days | ||||
| Administration Dosage | 5 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate | 75 mm3 | |||
| Administration Time | 42 days | ||||
| Administration Dosage | 10 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate | 100 mm3 | |||
| Administration Time | 42 days | ||||
| Administration Dosage | 10 mg/kg with ceralasertib 25 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate | 100 mm3 | |||
| Administration Time | 45 days | ||||
| Administration Dosage | 5 mg/kg with ceralasertib 25 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
References
