Drug Information
General Information of This Drug
| Drug ID | DRG00022 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Dizocilpine | |||||
| Synonyms |
DIZOCILPINE; 77086-21-6; MK-801; Dizocilpine [INN]; MK 801; Lopac-M-107; Lopac-M-108; MK-801 (Dizocilpine); Prestwick0_000109; (5S,10R)-5-methyl-10,11-dihydro-5H-5,10-epiminodibenzo[a,d][7]annulene; 7PY8KH681I; CHEMBL284237; CHEBI:34725; MK801; (1S,9R)-1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2,4,6,10,12,14-hexaene; Dizocilpina; Dizocilpinum; (Rac)-Dizocilpine; (Rac)-MK-801; Dizocilpinum [INN-Latin]; Dizocilpina [INN-Spanish]; UNII-7PY8KH681I; HSDB 7641; dizocilpine-(+); (+)MK-801; (-)-MK801; (+)-MK-801; (+/-)-MK801; DIZOCILPINE [MI]; Prestwick1_000109; Prestwick2_000109; Prestwick3_000109; DIZOCILPINE [HSDB]; Biomol-NT_000210; Lopac0_000872; SCHEMBL34528; BSPBio_000098; DIZOCILPINE [WHO-DD]; SPBio_002037; BPBio1_000108; BPBio1_001272; GTPL2403; 10,11-Dihydro-5-methyl-5H-dibenzo[a,d]cyclohepten-5,10-imine.(MK-801); DTXSID3048447; (5S,10R)-(+)-5-methyl-10,11-dihydro-5Hdibenzo[a,d]cyclohepten-5,10-imine; (5S,10S)-(+)-5-methyl-10,11-dihydro-5Hdibenzo[a,d]cyclohepten-5,10-imine; 1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene; 1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10,12,14-hexaene; 1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2,4,6,10(15),11,13-hexaene; CHEBI:132408; LBOJYSIDWZQNJS-CVEARBPZSA-N; HMS3267C15; (+)-1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene; (+/-) MK-8011-methyl-(9R,1R)-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene; (+/-)-1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene; (-)-1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene; (1S,9R)-1-methyl-16-azatetracyclo[7.6.1.0^{2,7}.0^{10,15}]hexadeca-2(7),3,5,10(15),11,13-hexaene; (1S,9R)-1-methyl-16-azatetracyclo[7.6.1.0^{2,7}.0^{10,15}]hexadeca-2,4,6,10(15),11,13-hexaene; (Dizocilpine)1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene; (MK-801)1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene; 1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene(MK-801); CDA08621; EX-A1701; BDBM50030386; BDBM50344263; HY-15084B; PDSP1_000178; PDSP1_001281; PDSP2_000177; PDSP2_001265; AKOS026673945; AT27821; CCG-204954; SDCCGSBI-0050847.P002; NCGC00015627-01; NCGC00015627-02; NCGC00016935-04; NCGC00016935-05; NCGC00016935-06; NCGC00016935-19; NCGC00024876-02; AC-35306; TS-09284; CS-0020032; NS00076277; SW197022-4; Q4386371; BRD-K58930050-001-01-1; BRD-K58930050-050-03-4; (+)-10,11-DIHYDRO-5-METHYL-5H-DIBENZO(A,D)CYCLOHEPTEN-5,10-IMINE; 5H-Dibenzo(a,d)cyclohepten-5,10-imine, 10,11-dihydro-5-methyl-, (5S)-; 5H-DIBENZO(A,D)CYCLOHEPTEN-5,10-IMINE, 10,11-DIHYDRO-5-METHYL-, (+)-; 5H-Dibenzo[a,d]cyclohepten-5,10-imine, 10,11-dihydro-5-methyl-, (5S,10R)-; (1S)-1-methyl-16-azatetracyclo[7.6.1.0^{2,7}.0^{10,15}]hexadeca-2(7),3,5,10,12,14-hexaene; (2Z)-but-2-enedioate; (1S,9R)-1-methyl-16-azatetracyclo[7.6.1.0?,?.0??,??]hexadeca-2,4,6,10,12,14-hexaene; 1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2(7),3,5,10(15),11,13-hexaene maleate((+)-MK801); 5H-Dibenzo[a,d]cyclohepten-5,10-imine, 10,11-dihydro-5-methyl-, (5S)-, (2Z)-2-butenedioate (1:1)
Click to Show/Hide
|
|||||
| Target(s) | Glutamate receptor ionotropic, NMDA 3A (GRIN3A) | Target Info | ||||
| Structure |
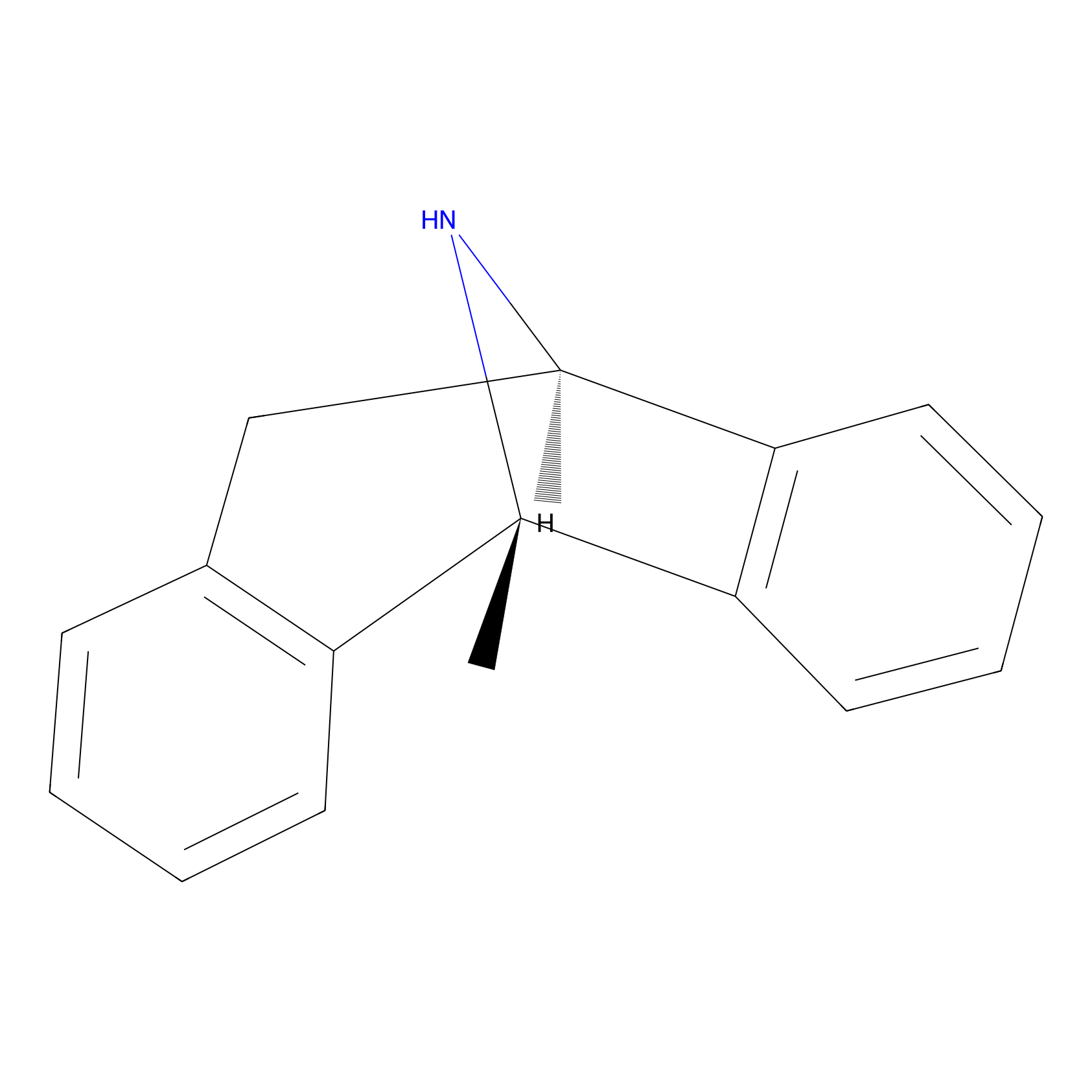
|
|||||
| Formula |
C16H15N
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 221.3 | ||||
| Lipid-water partition coefficient (xlogp) | 2.8 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 1 | |||||
| Rotatable Bond Count (rotbonds) | 0 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC12C3=CC=CC=C3CC(N1)C4=CC=CC=C24
|
|||||
| InChI |
InChI=1S/C16H15N/c1-16-13-8-4-2-6-11(13)10-15(17-16)12-7-3-5-9-14(12)16/h2-9,15,17H,10H2,1H3/t15-,16+/m1/s1
|
|||||
| InChIKey |
LBOJYSIDWZQNJS-CVEARBPZSA-N
|
|||||
| IUPAC Name |
(1S,9R)-1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]hexadeca-2,4,6,10,12,14-hexaene
|
|||||
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
GLP-1-MK-801 [Preclinical]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Obesity | ||||
| Efficacy Data | Vehicle-corrected weight loss rate | 9.50% | |||
| Evaluation Method | This study revealed a superior vehicle-corrected weight loss of 9.5% in response to the GLP-1-MK-801 infusion relative to a weight loss of 4.5% after semaglutide infusion (Fig. 4b). | ||||
| Administration Time | 1 dose | ||||
| Administration Dosage | 0.22 nmol | ||||
| MOA of PDC |
Treatment with GLP-1-inactive MK-801 produced no additional weight loss efficacy relative to GLP-1 monotherapy (Fig. 2g-i), indicating that the pronounced weight loss induced by GLP-1-MK-801 is driven by concerted and site-directed pharmacological GLP-1 receptor agonism and NMDA receptor antagonism.
|
||||
| In Vivo Model | High-fat high-sucrose-fed mice | ||||
| Half life period | 1.9 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Obesity | ||||
| Efficacy Data | Vehicle-corrected weight loss rate | 15.20% | |||
| Evaluation Method | We observed a pronounced vehicle-corrected weight loss of 15.2% in the group treated with GLP-1-MK-801 compared with 3.5% in the group treated with the parent GLP-1 analogue after 9days of treatment, underscoring that the weight-lowering efficacy of the conjugate is intact in the absence of functional MC4R signalling (Fig. 3l,m). | ||||
| Administration Time | Once a day for 9 days | ||||
| Administration Dosage | 100 nmol kg-1 | ||||
| MOA of PDC |
Treatment with GLP-1-inactive MK-801 produced no additional weight loss efficacy relative to GLP-1 monotherapy (Fig. 2g-i), indicating that the pronounced weight loss induced by GLP-1-MK-801 is driven by concerted and site-directed pharmacological GLP-1 receptor agonism and NMDA receptor antagonism.
|
||||
| In Vivo Model | Mc4r-KO mice | ||||
| Half life period | 1.9 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Obesity | ||||
| Efficacy Data | Vehicle-corrected weight loss rate | 23.20% | |||
| Evaluation Method | Over a 14-day treatment period, GLP-1-MK-801 synergistically lowered body weight compared with the dose-matched monotherapies and produced a vehicle-corrected weight loss of 23.2%. | ||||
| Administration Time | Once a day for 14 days | ||||
| Administration Dosage | 100 nmol kg-1 | ||||
| MOA of PDC |
Treatment with GLP-1-inactive MK-801 produced no additional weight loss efficacy relative to GLP-1 monotherapy (Fig. 2g-i), indicating that the pronounced weight loss induced by GLP-1-MK-801 is driven by concerted and site-directed pharmacological GLP-1 receptor agonism and NMDA receptor antagonism.
|
||||
| In Vivo Model | DIO mice | ||||
| Half life period | 1.9 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Obesity | ||||
| Efficacy Data | Lean mass rate | 8% | |||
| Evaluation Method | GLP-1-MK-801 produced a vehicle-corrected reduction in body fat mass of 45%, accompanied by an 8% loss in lean mass. | ||||
| Administration Time | Once a day for 14 days | ||||
| Administration Dosage | 100 nmol kg-1 | ||||
| MOA of PDC |
Treatment with GLP-1-inactive MK-801 produced no additional weight loss efficacy relative to GLP-1 monotherapy (Fig. 2g-i), indicating that the pronounced weight loss induced by GLP-1-MK-801 is driven by concerted and site-directed pharmacological GLP-1 receptor agonism and NMDA receptor antagonism.
|
||||
| In Vivo Model | DIO mice | ||||
| Half life period | 1.9 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Obesity | ||||
| Efficacy Data | Fat mass reduction rate | 45% | |||
| Evaluation Method | GLP-1-MK-801 produced a vehicle-corrected reduction in body fat mass of 45%, accompanied by an 8% loss in lean mass. | ||||
| Administration Time | Once a day for 14 days | ||||
| Administration Dosage | 100 nmol kg-1 | ||||
| MOA of PDC |
Treatment with GLP-1-inactive MK-801 produced no additional weight loss efficacy relative to GLP-1 monotherapy (Fig. 2g-i), indicating that the pronounced weight loss induced by GLP-1-MK-801 is driven by concerted and site-directed pharmacological GLP-1 receptor agonism and NMDA receptor antagonism.
|
||||
| In Vivo Model | DIO mice | ||||
| Half life period | 1.9 h | ||||
References
