Drug Information
General Information of This Drug
| Drug ID | DRG00030 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Gambogic acid | |||||
| Synonyms |
gambogic acid; 2752-65-0; (-)-Gambogic Acid; Gambogic-acid; R-gambogic acid; beta-Guttiferin; Cambogic acid; B''-Guttiferin; 8N585K83U2; .beta.-Guttiferin; GAMBOGICACID; (Z)-4-((1S,3aR,5S,11R,14aS)-8-hydroxy-2,2,11-trimethyl-13-(3-methylbut-2-en-1-yl)-11-(4-methylpent-3-en-1-yl)-4,7-dioxo-1,2,5,7-tetrahydro-11H-1,5-methanofuro[3,2-g]pyrano[3,2-b]xanthen-3a(4H)-yl)-2-methylbut-2-enoic acid; CHEBI:67521; UNII-8N585K83U2; GAMBOGIC ACID [MI]; CHEMBL555017; CHEMBL5305342; SCHEMBL16160279; GEZHEQNLKAOMCA-RRZNCOCZSA-N; DTXSID101029723; GLXC-02792; BCP21539; HY-N0087; BDBM50366237; MFCD16878985; s2448; AKOS024463359; CCG-270284; CS-1456; AC-34804; BP-22199; Gambogic acid, >=95% (HPLC), powder; H10129; Q5519727; (2Z)-2-METHYL-4-((1R,3AS,5S,11R,14AS)-3A,4,5,7-TETRAHYDRO-8-HYDROXY-3,3,11-TRIMETHYL-13-(3-METHYL-2-BUTEN-1-YL)-11-(4-METHYL-3-PENTEN-1-YL)-7,15-DIOXO-1,5-METHANO-1H,3H,11H-FURO(3,4-G)PYRANO(3,2-B)XANTHEN-1-YL)-2-BUTENOIC ACID; (Z)-4-[(1S,2S,8R,17S,19R)-12-hydroxy-8,21,21-trimethyl-5-(3-methylbut-2-enyl)-8-(4-methylpent-3-enyl)-14,18-dioxo-3,7,20-trioxahexacyclo[15.4.1.02,15.02,19.04,13.06,11]docosa-4(13),5,9,11,15-pentaen-19-yl]-2-methylbut-2-enoic acid; 1,5-Methano-1H,3H,11H-furo(3,4-g)pyrano(3,2-b)xanthene-1-crotonic acid, 3a,4,5,7-tetrahydro-8-hydroxy-alpha,3,3,11-tetramethyl-13-(3-methyl-2-butenyl)-11-(4-methyl-3-pentenyl)-7,15-dioxo-, (Z)-; 2-BUTENOIC ACID, 2-METHYL-4-((1R,3AS,5S,11R,14AS)-3A,4,5,7-TETRAHYDRO-8-HYDROXY-3,3,11-TRIMETHYL-13-(3-METHYL-2-BUTEN-1-YL)-11-(4-METHYL-3-PENTEN-1-YL)-7,15-DIOXO-1,5-METHANO-1H,3H,11H-FURO(3,4-G)PYRANO(3,2-B)XANTHEN-1-YL)-, (2Z)-
Click to Show/Hide
|
|||||
| Structure |
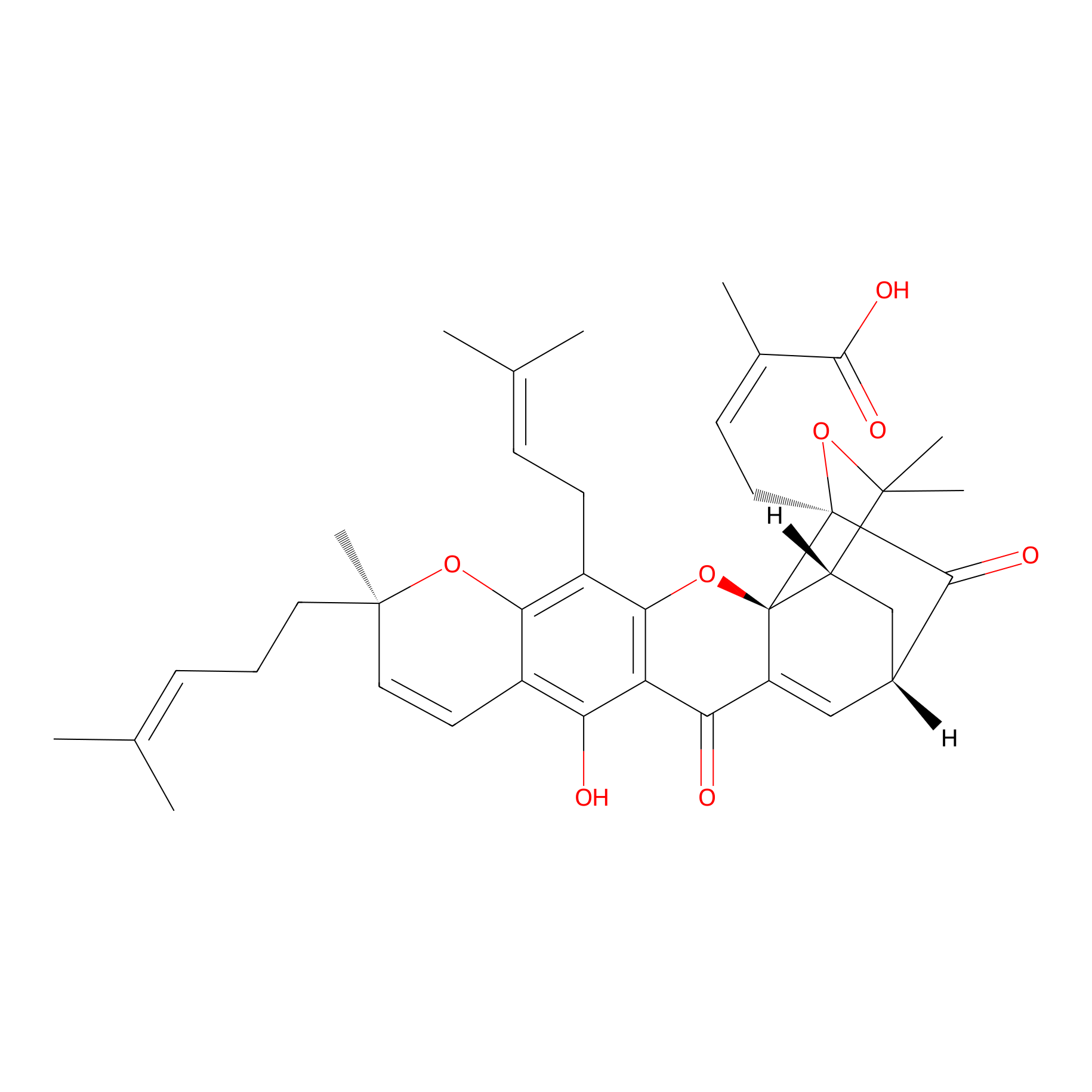
|
|||||
| Formula |
C38H44O8
|
|||||
| #Ro5 Violations (Lipinski): 2 | Molecular Weight (mw) | 628.7 | ||||
| Lipid-water partition coefficient (xlogp) | 7.3 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 8 | |||||
| Rotatable Bond Count (rotbonds) | 8 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC(=CCCC1(C=CC2=C(C3=C(C(=C2O1)CC=C(C)C)OC45C6CC(C=C4C3=O)C(=O)C5(OC6(C)C)CC=C(C)C(=O)O)O)C)C
|
|||||
| InChI |
InChI=1S/C38H44O8/c1-20(2)10-9-15-36(8)16-14-24-29(39)28-30(40)26-18-23-19-27-35(6,7)46-37(33(23)41,17-13-22(5)34(42)43)38(26,27)45-32(28)25(31(24)44-36)12-11-21(3)4/h10-11,13-14,16,18,23,27,39H,9,12,15,17,19H2,1-8H3,(H,42,43)/b22-13-/t23-,27+,36-,37+,38-/m1/s1
|
|||||
| InChIKey |
GEZHEQNLKAOMCA-RRZNCOCZSA-N
|
|||||
| IUPAC Name |
(Z)-4-[(1S,2S,8R,17S,19R)-12-hydroxy-8,21,21-trimethyl-5-(3-methylbut-2-enyl)-8-(4-methylpent-3-enyl)-14,18-dioxo-3,7,20-trioxahexacyclo[15.4.1.02,15.02,19.04,13.06,11]docosa-4(13),5,9,11,15-pentaen-19-yl]-2-methylbut-2-enoic acid
|
|||||
The activity data of This Drug
| Standard Type | Value | Administration times | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | 4.95 µM | 24 h | Bladder cancer cell | N.A. | [1] | |
Full Information of The Activity Data of The PDC(s) Related to This Drug
GA-TAT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bladder cancer | ||||
| Efficacy Data | Inhibition rate | 46.4% ± 4.86% | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 1.0 µM | ||||
| MOA of PDC |
These findings suggest that GA-TAT-induced EJ cell apoptosis is mediated via ROS production. Our study demonstrated that GA-TAT enhanced the pro-apoptotic effect via increasing caspase-3 and caspase-9 processing and activities and decreasing the Bcl-2/Bax ratio, which were regulated by intracellular ROS.
|
||||
| Description |
However, after treatment with GA-TAT, the percentage of apoptotic cells was greatly increased to 46.4%±4.86%.
|
||||
| In Vitro Model | Bladder carcinoma | EJ-1 cell | CVCL_2893 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Bladder cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.24 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| MOA of PDC |
These findings suggest that GA-TAT-induced EJ cell apoptosis is mediated via ROS production. Our study demonstrated that GA-TAT enhanced the pro-apoptotic effect via increasing caspase-3 and caspase-9 processing and activities and decreasing the Bcl-2/Bax ratio, which were regulated by intracellular ROS.
|
||||
| Description |
The 50% inhibitory concentration (IC50) of GA-TAT at 24 h was 1.24 uM
|
||||
| In Vitro Model | Bladder cancer | Bladder cancer cell | Homo sapiens | ||
References
