Drug Information
General Information of This Drug
| Drug ID | DRG00036 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Cytolysin-dithiopyridine | |||||
| Structure |
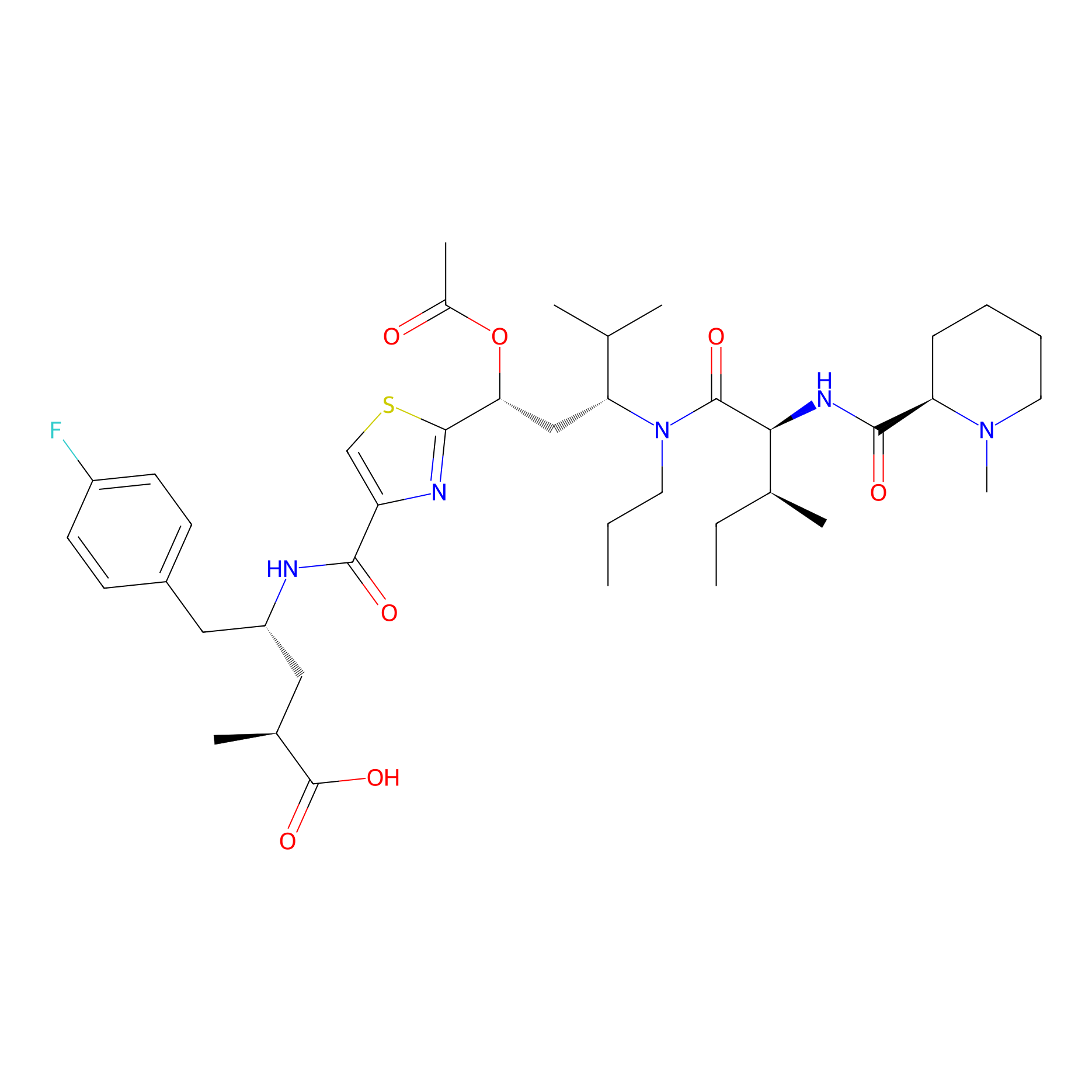
|
|||||
| Formula |
C40H60FN5O7S
|
|||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight (mw) | 774 | ||||
| Lipid-water partition coefficient (xlogp) | 4.8 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 11 | |||||
| Rotatable Bond Count (rotbonds) | 21 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CCCN(C(CC(C1=NC(=CS1)C(=O)NC(CC2=CC=C(C=C2)F)CC(C)C(=O)O)OC(=O)C)C(C)C)C(=O)C(C(C)CC)NC(=O)C3CCCCN3C
|
|||||
| InChI |
InChI=1S/C40H60FN5O7S/c1-9-18-46(39(50)35(25(5)10-2)44-37(49)32-13-11-12-19-45(32)8)33(24(3)4)22-34(53-27(7)47)38-43-31(23-54-38)36(48)42-30(20-26(6)40(51)52)21-28-14-16-29(41)17-15-28/h14-17,23-26,30,32-35H,9-13,18-22H2,1-8H3,(H,42,48)(H,44,49)(H,51,52)/t25-,26-,30+,32+,33+,34+,35-/m0/s1
|
|||||
| InChIKey |
JEGBLJXQGQRAKP-YRKZXFTRSA-N
|
|||||
| IUPAC Name |
(2S,4R)-4-[[2-[(1R,3R)-1-acetyloxy-4-methyl-3-[[(2S,3S)-3-methyl-2-[[(2R)-1-methylpiperidine-2-carbonyl]amino]pentanoyl]-propylamino]pentyl]-1,3-thiazole-4-carbonyl]amino]-5-(4-fluorophenyl)-2-methylpentanoic acid
|
|||||
Full Information of The Activity Data of The PDC(s) Related to This Drug
Cytolysin[F7,P34]-NPY bioconjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 236.1 nM | |||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 405.3 nM | |||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
References
