Drug Information
General Information of This Drug
| Drug ID | DRG00037 | |||||
|---|---|---|---|---|---|---|
| Drug Name | TGX-D1 | |||||
| Synonyms |
TGX-D1; BL-05; GLXC-01756; 1308384-65-7
Click to Show/Hide
|
|||||
| Target(s) | Phosphatidylinositol 3-kinase catalytic subunit type 3 (PIK3C3) | Target Info | ||||
| Structure |
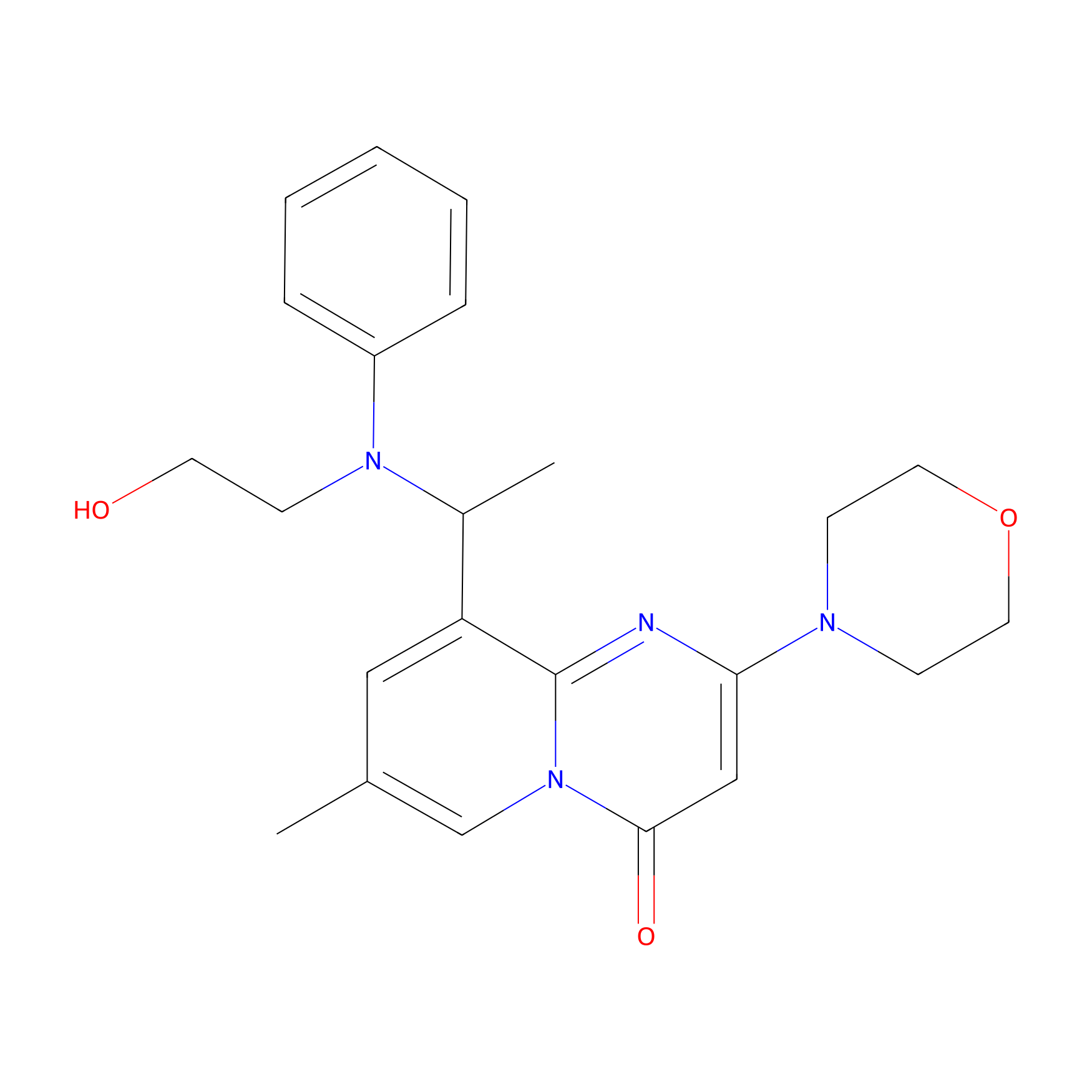
|
|||||
| Formula |
C23H28N4O3
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 408.5 | ||||
| Lipid-water partition coefficient (xlogp) | 1.3 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | |||||
| Rotatable Bond Count (rotbonds) | 6 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC1=CN2C(=O)C=C(N=C2C(=C1)C(C)N(CCO)C3=CC=CC=C3)N4CCOCC4
|
|||||
| InChI |
InChI=1S/C23H28N4O3/c1-17-14-20(18(2)26(8-11-28)19-6-4-3-5-7-19)23-24-21(15-22(29)27(23)16-17)25-9-12-30-13-10-25/h3-7,14-16,18,28H,8-13H2,1-2H3
|
|||||
| InChIKey |
VNYXTYFKPUGCCT-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
9-[1-[N-(2-hydroxyethyl)anilino]ethyl]-7-methyl-2-morpholin-4-ylpyrido[1,2-a]pyrimidin-4-one
|
|||||
The activity data of This Drug
| Standard Type | Value | Disease Model | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | 2.67 µM | Prostate carcinoma | LNCaP cell | CVCL_0395 | [1] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
KYL-TGX [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Inhinition rate | 14.93 µM | |||
| Administration Time | 72 h | ||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
| Half life period | 4 h | ||||
References
