Drug Information
General Information of This Drug
| Drug ID | DRG00046 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Hydroxycamptothecin | |||||
| Synonyms |
SCHEMBL24513666
Click to Show/Hide
|
|||||
| Target(s) | DNA topoisomerase 1 (TOP1) | Target Info | ||||
| Structure |
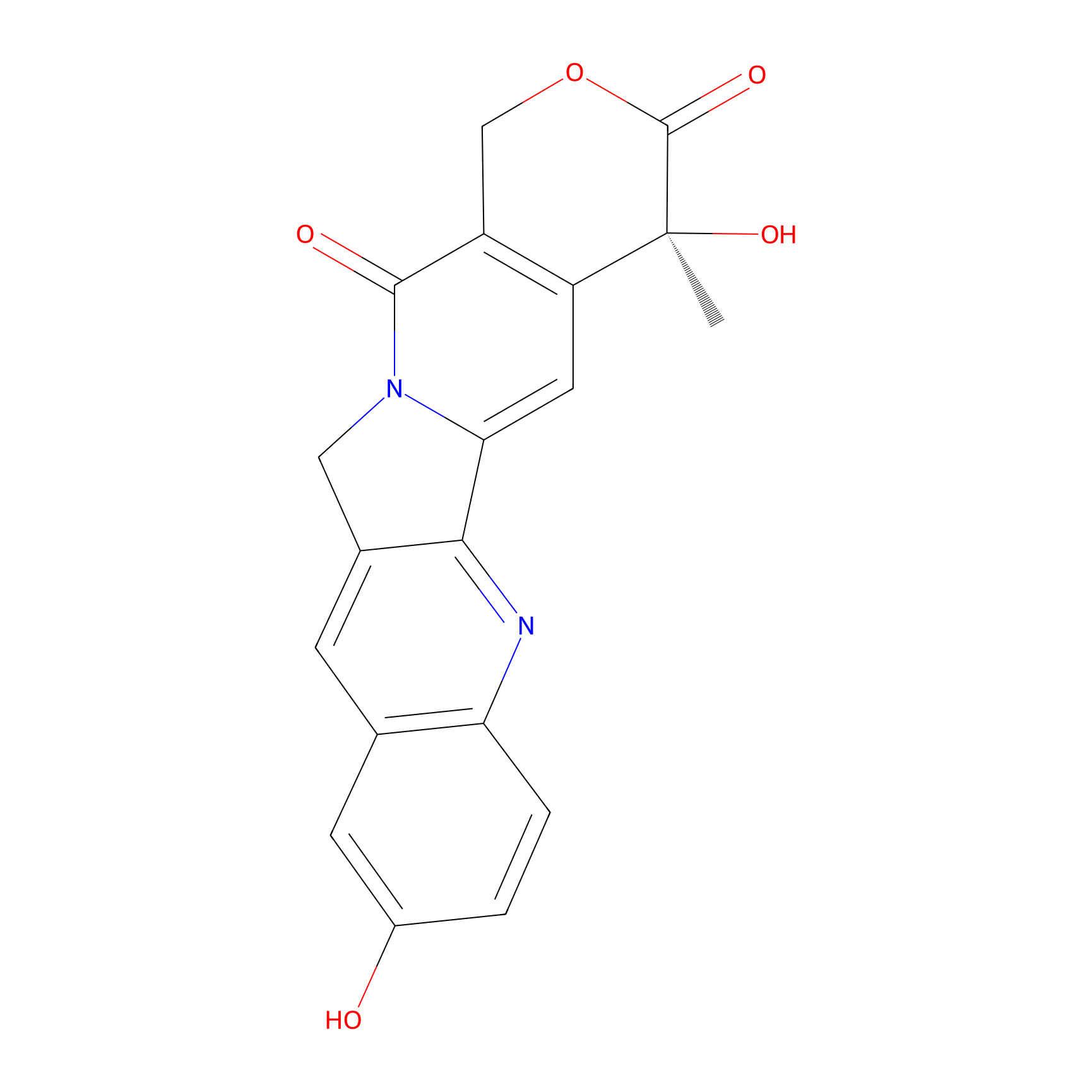
|
|||||
| Formula |
C19H14N2O5
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 350.3 | ||||
| Lipid-water partition coefficient (xlogp) | 0.1 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | |||||
| Rotatable Bond Count (rotbonds) | 0 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC1(C2=C(COC1=O)C(=O)N3CC4=C(C3=C2)N=C5C=CC(=CC5=C4)O)O
|
|||||
| InChI |
InChI=1S/C19H14N2O5/c1-19(25)13-6-15-16-10(4-9-5-11(22)2-3-14(9)20-16)7-21(15)17(23)12(13)8-26-18(19)24/h2-6,22,25H,7-8H2,1H3/t19-/m0/s1
|
|||||
| InChIKey |
INELCMUAACUTMU-IBGZPJMESA-N
|
|||||
| IUPAC Name |
(19S)-7,19-dihydroxy-19-methyl-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaene-14,18-dione
|
|||||
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
HCPTMDP26-PEG [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.27 µM | |||
| Description |
Peptides and drug-peptide conjugates were incubated with HepG2 cells for 48 hours followed by a CCK8 cytotoxicity assay. Despite the large population of the positive charges, supramolecular peptides alone demonstrated negligible cytotoxicity (Fig. S8, ESI). The drug conjugate, HCPT-MDP26-PEG showed a dose-dependent cytotoxicity with an IC50of 1.27 uM, comparable to the IC50of free HCPT at ˜1.3 uM (Fig. 3a). At a high drug dosage, the drug conjugate is much more potent than free HCPT.
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
References
