Drug Information
General Information of This Drug
| Drug ID | DRG00065 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Irinotecan | |||||
| Synonyms |
irinotecan; 97682-44-5; (+)-Irinotecan; Camptosar; Irinophore C; Irinotecanum; Biotecan; Irinotecan lactone; Irinotecan mylan; Irinotecanum [INN-Latin]; Campto; Irinotecan Free base; CPT-11; (S)-4,11-diethyl-4-hydroxy-3,14-dioxo-3,4,12,14-tetrahydro-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl [1,4'-bipiperidine]-1'-carboxylate; HSDB 7607; Irinotecan (INN); NSC-728073; CHEBI:80630; CHEMBL481; UNII-7673326042; DTXSID1041051; 97682-44-5 (Free base); NSC728073; NSC 728073; (4S)-4,11-DIETHYL-4-HYDROXY-3,14-DIOXO-3,4,12,14-TETRAHYDRO-1H-PYRANO[3',4':6,7]INDOLIZINO[1,2-B]QUINOLIN-9-YL 1,4'-BIPIPERIDINE-1'-CARBOXYLATE; 1,4'-Bipiperidine-1'-carboxylic acid (S)-4,11-diethyl-3,4,12,14- tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl ester; IRINOTECAN [INN]; Irinotecanum (INN-Latin); Irinotecan [INN:BAN]; (1,4'-Bipiperidine)-1'-carboxylic acid, 4,11-diethyl-3,4,12-14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano(3',4':6,7)indolizino(1,2-b)quinolin-9-yl ester, (S)-; CPT-11 hydrochloride;Camptothecin 11 hydrochloride; Irrinotecan; Biotecan (TN); (1,4'-Bipiperidine)-1'-carboxylic acid, (4S)-4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano(3',4':6,7)indolizino(1,2-b)quinolin-9-yl ester; [1,4'-Bipiperidine]-1'-carboxylic acid, (4S)-4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl ester; 1,4'-bipiperidine-1'-carboxylic acid (s)-4,11-diethyl-3,4,12,14- tetrahydro-4-hydroxy-3,14-dioxo-1h-pyrano(3',4':6,7)indolizino(1,2-b)quinolin-9-yl ester; Irinotecan?; MFCD00866307; (4S)-4,11-diethyl-4-hydroxy-3,14-dioxo-3,4,12,14-tetrahydro-1H-pyrano(3',4':6,7)indolizino(1,2-b)quinolin-9-yl (1,4'-bipiperidine)-1'-carboxylate; (4S)-4,11-diethyl-4-hydroxy-3,14-dioxo-3,4,12,14-tetrahydro-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl [1,4'-bipiperidine]-1'-carboxylate; IRINOTECAN [MI]; IRINOTECAN [HSDB]; IRINOTECAN [VANDF]; SCHEMBL4034; IRINOTECAN [WHO-DD]; IRINOTECAN; CPT-11; BSPBio_002346; GTPL6823; DTXCID9021051; AMY4227; L01XX19; 1u65; BCP02860; BDBM50128267; s1198; AKOS015894969; AB07527; AC-7469; BCP9000793; CS-1138; DB00762; NCGC00178697-02; NCGC00178697-05; (4S)-4,11-Diethyl-4-hydroxy-3,14-dioxo-4,12-dihydro-1H-pyrano[3,4-f]quinolino[2,3-a]indolizin-9-yl 4-piperidylpiperidinecarboxylate; AS-14323; HY-16562; NCI60_005051; NS00004943; D08086; EN300-708800; AB00698464-07; AB00698464-09; AB00698464-10; AB00698464-11; AB00698464_12; AB00698464_13; AB00698464_14; A845740; Q412197; BRD-K08547377-003-02-4; (diethyl-hydroxy-dioxo-[?]yl) 4-(1-piperidyl)piperidine-1-carboxylate; (+)-7-ethyl-10-hydroxycamptothecine 10-(1,4'-bipiperidine)-1'-carboxylate; 2-methoxy-5-[2-(3-sulfophenyl)-5-(4-sulfophenyl)pyrylium-4-yl]benzenesulfonic acid; (+)-(4S)-4,11-diethyl-4-hydroxy-9-((4-piperidino-piperidino)carbonyloxy)-1H-pyrano(3',4':6,7)indolizino(1,2-b)quinol-3,14,(4H,12H)-dione; (1,4'-bipiperidine)-1'-carboxylic acid (S)-4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano(3',4':6,7)indolizino(1,2-b)quinolin-9-yl ester; (19S)-10,19-Diethyl-19-hydroxy-14,18-dioxo-17-oxa-3,13-diazapentacyclo[11.8.0.0[2,11].0[4,9].0[15,20]]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaen-7-yl 4-(piperidin-1-yl)piperidine-1-carboxylate; (19S)-10,19-diethyl-19-hydroxy-14,18-dioxo-17-oxa-3,13-diazapentacyclo[11.8.0.0^{2,11}.0^{4,9}.0^{15,20}]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaen-7-yl [1,4'-bipiperidine]-1'-carboxylate; (19S)-10,19-diethyl-19-hydroxy-14,18-dioxo-17-oxa-3,13-diazapentacyclo[11.8.0.0^{2,11}.0^{4,9}.0^{15,20}]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaen-7-yl 4-(piperidin-1-yl)piperidine-1-carboxylate; (4S)-4,11-DIETHYL-4-HYDROXY-3,14-DIOXO-3,4,12,14-TETRAHYDRO-1H-PYRANO[3'',4'':6,7]INDOLIZINO[1,2-B]QUINOLIN-9-YL 1,4''-BIPIPERIDINE-1''-CARBOXYLATE; (S)-4,11-diethyl-4-hydroxy-3,14-dioxo-3,4,12,14-tetrahydro-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl[1,4'-bipiperidine]-1'-carboxylate; [(19S)-10,19-diethyl-19-hydroxy-14,18-dioxo-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2,4(9),5,7,10,15(20)-heptaen-7-yl] 4-piperidin-1-ylpiperidine-1-carboxylate; [1,4'']bipiperidinyl-1''-carboxylic acid (S)-4,11-diethyl-4-hydroxy-3,13-dioxo-3,4,12,13-tetrahydro-1H-2-oxa-6,12a-diaza-dibenzo[b,h]fluoren-9-yl ester; [1,4'']Bipiperidinyl-1''-carboxylic acid 4,11-diethyl-4-hydroxy-3,13-dioxo-3,4,12,13-tetrahydro-1H-2-oxa-6,12a-diaza-dibenzo[b,h]fluoren-9-yl ester
Click to Show/Hide
|
|||||
| Target(s) | DNA topoisomerase 1 (TOP1) | Target Info | ||||
| Structure |
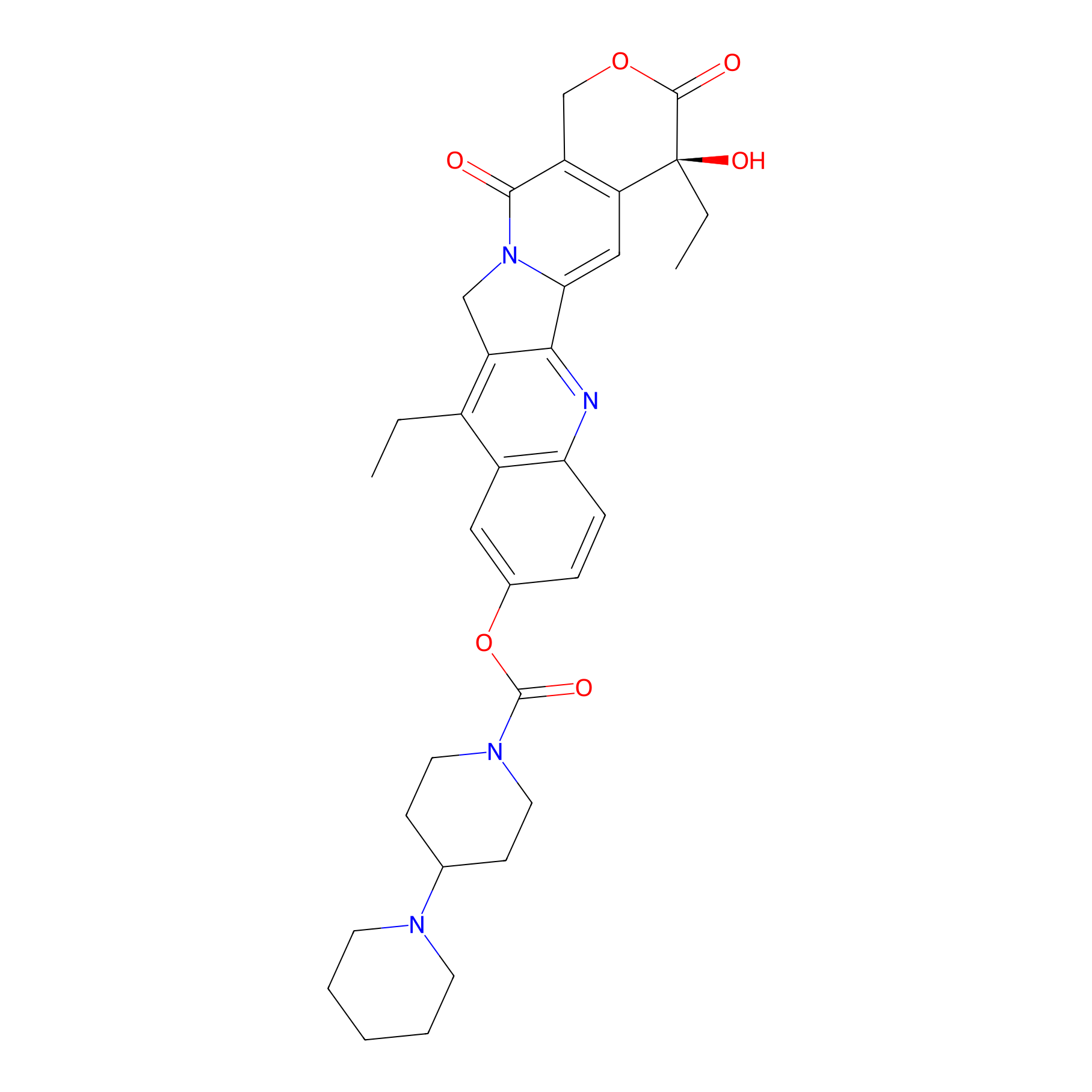
|
|||||
| Formula |
C33H38N4O6
|
|||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 586.7 | ||||
| Lipid-water partition coefficient (xlogp) | 3 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 8 | |||||
| Rotatable Bond Count (rotbonds) | 5 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CCC1=C2CN3C(=CC4=C(C3=O)COC(=O)C4(CC)O)C2=NC5=C1C=C(C=C5)OC(=O)N6CCC(CC6)N7CCCCC7
|
|||||
| InChI |
InChI=1S/C33H38N4O6/c1-3-22-23-16-21(43-32(40)36-14-10-20(11-15-36)35-12-6-5-7-13-35)8-9-27(23)34-29-24(22)18-37-28(29)17-26-25(30(37)38)19-42-31(39)33(26,41)4-2/h8-9,16-17,20,41H,3-7,10-15,18-19H2,1-2H3/t33-/m0/s1
|
|||||
| InChIKey |
UWKQSNNFCGGAFS-XIFFEERXSA-N
|
|||||
| IUPAC Name |
[(19S)-10,19-diethyl-19-hydroxy-14,18-dioxo-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2,4(9),5,7,10,15(20)-heptaen-7-yl] 4-piperidin-1-ylpiperidine-1-carboxylate
|
|||||
The activity data of This Drug
| Standard Type | Value | Disease Model | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Growth Inhibition (GI50) | >1000 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [1] | |
| Half Maximal Growth Inhibition (GI50) | 7.1 uM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [2] | |
| Half Maximal Growth Inhibition (GI50) | 9.17 uM | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | [3] | |
| Half Maximal Growth Inhibition (GI50) | >10 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [3] | |
| Half Maximal Infective Dose (ID50) | >500 nM | High grade ovarian serous adenocarcinoma | NCI-ADR-RES cell | CVCL_1452 | [4] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | Prostate carcinoma | PC-3 cell | CVCL_0035 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | Amelanotic melanoma | A-375 cell | CVCL_0132 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 6.9 nM | Colon adenocarcinoma | T84 cell | CVCL_0555 | [6] | |
| Half Maximal Inhibitory Concentration (IC50) | 9 nM | Prostate carcinoma | LNCaP cell | CVCL_0395 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 15 nM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 27 nM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 32 nM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [7] | |
| Half Maximal Inhibitory Concentration (IC50) | 200 nM | Prostate carcinoma | DU145 cell | CVCL_0105 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 220 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 250 nM | Lung small cell carcinoma | H69AR cell | CVCL_3513 | [5] | |
| Half Maximal Inhibitory Concentration (IC50) | 570 nM | T acute lymphoblastic leukemia | RPMI-8402 cell | CVCL_1667 | [8] | |
| Half Maximal Inhibitory Concentration (IC50) | 800 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [9] | |
| Half Maximal Inhibitory Concentration (IC50) | 900 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [10] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.2 uM | Lymphoblastic leukemia | L1210 cell | CVCL_0382 | [11] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.5 uM | Pancreatic ductal adenocarcinoma | Capan-1 cell | CVCL_0237 | [12] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.557 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [13] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.9 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [10] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.55 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [14] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.15 uM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [15] | |
| Half Maximal Inhibitory Concentration (IC50) | 6.3 uM | Amelanotic melanoma | MDA-MB-435 cell | CVCL_0417 | [16] | |
| Half Maximal Inhibitory Concentration (IC50) | 6.4 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [17] | |
| Half Maximal Inhibitory Concentration (IC50) | 7.99 uM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [18] | |
| Half Maximal Inhibitory Concentration (IC50) | 8.3 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [19] | |
| Half Maximal Inhibitory Concentration (IC50) | 8.31 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [18] | |
| Half Maximal Inhibitory Concentration (IC50) | 8.8 uM | Colon adenocarcinoma | LoVo cell | CVCL_0399 | [17] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.5 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [20] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.7 uM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [21] | |
| Half Maximal Inhibitory Concentration (IC50) | >10 uM | Mantle cell lymphoma | JeKo-1 cell | CVCL_1865 | [12] | |
| Half Maximal Inhibitory Concentration (IC50) | >10 uM | Diffuse large B-cell lymphoma | OCI-Ly3 cell | CVCL_8800 | [12] | |
| Half Maximal Inhibitory Concentration (IC50) | >10 uM | Burkitt lymphoma | DG-75 cell | CVCL_0244 | [12] | |
| Half Maximal Inhibitory Concentration (IC50) | 11.32 uM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [18] | |
| Half Maximal Inhibitory Concentration (IC50) | 12.3 uM | Colon adenocarcinoma | SW480 cell | CVCL_0546 | [22] | |
| Half Maximal Inhibitory Concentration (IC50) | 16.53 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [23] | |
| Half Maximal Inhibitory Concentration (IC50) | 17.403 uM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [24] | |
| Half Maximal Inhibitory Concentration (IC50) | 18 uM | Breast carcinoma | ZR-75-30 cell | CVCL_1661 | [25] | |
| Half Maximal Inhibitory Concentration (IC50) | 18.81 uM | Lung squamous cell carcinoma | NCI-H2170 cell | CVCL_1535 | [15] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 uM | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | [26] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 uM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [26] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
BGC-0222 [Phase 1]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values | 9.21% | |||
| Administration Time | 32 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 2 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values | 9.87% | |||
| Administration Time | 29 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values | 16.90% | |||
| Administration Time | 25 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 4 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values | 27.60% | |||
| Administration Time | 22 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 5 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values | 57.00% | |||
| Administration Time | 18 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values | 88.80% | |||
| Administration Time | 15 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 7 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor increment rates values | 100% | |||
| Administration Time | 12 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 8 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 90% | |||
| Administration Time | 32 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 9 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Glioblastoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 94% | |||
| Administration Time | 40 days | ||||
| Administration Dosage | 60 mg/kg, QW3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse U-87MG cells xenograft model. | ||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Small cell lung cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 95% | |||
| Administration Time | 30 days | ||||
| Administration Dosage | 20 mg/kg, QW3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse NCI-H446 cells xenograft model. | ||||
| In Vitro Model | Lung small cell carcinoma | NCI-H446 cell | CVCL_1562 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 98% | |||
| Administration Time | 31 days | ||||
| Administration Dosage | 20 mg/kg, QW3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse MIA PaCa-2 cells xenograft model. | ||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 99% | |||
| Administration Time | 36 days | ||||
| Administration Dosage | 20 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
It was worth noting that, similar with that in HT-29 mice model, as shown in Fig. 3, BGC0222 also exhibited better antitumor effect than irinotecan and NKTR-102 in MIA PaCa-2(B), NCI-H446(C), U-87 MG(D) and MDA-MB-231(E) xenograft models, with lower RTV and T/C values. Moreover, in these model assays, there were also no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse MDA-MB-231 cells xenograft model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) | 0.72 | |||
| Administration Time | 29 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 14 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) | 0.82 | |||
| Administration Time | 32 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 15 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) | 0.88 | |||
| Administration Time | 25 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 16 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) | 1 | |||
| Administration Time | 12 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 17 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) | 1.15 | |||
| Administration Time | 22 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 18 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) | 1.47 | |||
| Administration Time | 15 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 19 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Relative tumor volume (RTV) | 1.56 | |||
| Administration Time | 18 days | ||||
| Administration Dosage | 40 mg/kg, Q4D3 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
BGC0222 exhibited remarkable inhibition on HT-29 tumor growth. Firstly, for BGC0222, the RTV values of days 12, 15, 18, 22, 25, 29 and 32 were found to be 1.00, 1.47, 1.56, 1.15, 0.88, 0.72, 0.82, while that of irinotecan and NKTR-102 were found to be 1.00, 1.71, 2.54, 3.13, 3.60, 4.43, 6.31 and 1.00, 1.59, 1.91, 2.14, 2.03, 2.07, 2.41, respectivel. Evidently, the RTV values of BGC0222 were much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100X300 mm3 (after day 12), indicating that the in vivo antitumor activity of BGC0222 was obviously better than that of irinotecan and NKTR-102. In addition, as shown in Tables S31, T/C values of BGC0222 for days 12, 15, 18, 22, 25, 29 and 32 were determined to be 100%, 88.8%, 57.0%, 27.6%, 16.9%, 9.87% and 9.21%, while that of irinotecan and NKTR-102 were found to be 100%, 103%, 93.1%, 75.1%, 68.8%, 60.6%, 71.1% and 100%, 96.0%, 70.0%, 51.3%, 38.7%, 28.3%, 27.2%, respectively. Clearly, the T/C values of BGC0222 were also much lower than that of irinotecan and NKTR-102 when the average tumor size reached approximately 100300 mm3, demonstrating that its' in vivo antitumor activity was better than that of irinotecan and NKTR-102. This result was consistent with that of RTV assay. These in vivo results indicated that BGC0222 exhibited higher antitumor effect than irinotecan and NKTR-102 at the same condition in the HT-29 mouse model, consistent with the results of the in vitro cytotoxicity assay. It should be important to note that no significant change in body weight and no other adverse effects were observed among the mice treated with BGC0222, indicating that BGC0222 displayed no significant toxicity to the mice within the period of treatment. It was obvious that the weight change range of the mice treated with irinotecan was bigger than that of BGC0222, impling that the toxicity of BGC0222 may be lower than that of irinotecan, consistent with that in preliminary safety evaluation.
Click to Show/Hide
|
||||
| In Vivo Model | Female Balb/c nude mouse HT-29 cells xenograft model. | ||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.68 ± 0.04 μM | |||
| Evaluation Method | Graph Pad Prism 5.0 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
Compared with irinotecan and NKTR-102, compound BGC0222 exhibited better antiproliferative activity in all these assayed cell lines. In the antiproliferative activity assay with the HT29 cell line, compound BGC0222 displayed higher antiproliferative activity than irinotecan (IC50 = 7.64 0.19 μM) and NKTR-102 (IC50 = 2.02 0.10 μM), with IC50 value of 1.83 0.09 μM. In addition, compound BGC0222 showed better antiproliferative activity than irinotecan (IC50 = 16.2 0.22 μM) and NKTR-102 (IC50 = 4.68 0.15 μM) against MIA PaCa-2 cells line, with IC50 value of 3.95 0.16 μM, while compound BGC0222 also exhibited stronger antiproliferative activity than irinotecan (IC50 = 2.69 0.12 μM) and NKTR-102 (IC50 = 0.71 0.05 μM) in MCF-7 cells line assay, with IC50 value of 0.68 0.04 μM, respectively. Clearly, the order of in vitro antiproliferative effect against the assay cell lines was: BGC0222>NKTR-102 > irinotecan. The structure-activity relationship was then investigated. The success of NKTR-102 proved that the introducing of PEG should lead to better antiproliferative activity. In addition, by the comparison of the structure of BGC0222 with that of NKTR-102, it should be found the presence of cRGD lead to better antiproliferative effect. Therefore, based on the above observation, it could be concluded that the introduction of PEG and cRGD to irinotecan should efficiently improve the antiproliferative effect, consistent with our expectation.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.83 ± 0.09 μM | |||
| Evaluation Method | Graph Pad Prism 5.0 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
Compared with irinotecan and NKTR-102, compound BGC0222 exhibited better antiproliferative activity in all these assayed cell lines. In the antiproliferative activity assay with the HT29 cell line, compound BGC0222 displayed higher antiproliferative activity than irinotecan (IC50 = 7.64 0.19 μM) and NKTR-102 (IC50 = 2.02 0.10 μM), with IC50 value of 1.83 0.09 μM. In addition, compound BGC0222 showed better antiproliferative activity than irinotecan (IC50 = 16.2 0.22 μM) and NKTR-102 (IC50 = 4.68 0.15 μM) against MIA PaCa-2 cells line, with IC50 value of 3.95 0.16 μM, while compound BGC0222 also exhibited stronger antiproliferative activity than irinotecan (IC50 = 2.69 0.12 μM) and NKTR-102 (IC50 = 0.71 0.05 μM) in MCF-7 cells line assay, with IC50 value of 0.68 0.04 μM, respectively. Clearly, the order of in vitro antiproliferative effect against the assay cell lines was: BGC0222>NKTR-102 > irinotecan. The structure-activity relationship was then investigated. The success of NKTR-102 proved that the introducing of PEG should lead to better antiproliferative activity. In addition, by the comparison of the structure of BGC0222 with that of NKTR-102, it should be found the presence of cRGD lead to better antiproliferative effect. Therefore, based on the above observation, it could be concluded that the introduction of PEG and cRGD to irinotecan should efficiently improve the antiproliferative effect, consistent with our expectation.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [27] | ||||
| Indication | Pancreatic ductal adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 3.95 ± 0.16 μM | |||
| Evaluation Method | Graph Pad Prism 5.0 | ||||
| MOA of PDC |
As a semisynthetic analog of camptothecin (CPT), irinotecan is a well-known topoisomerase 1 (Top1) inhibitor and widely used chemotherapeutic agent. In order to increase the antitumor efficiency and low solubility and reduce the toxicity of irinotecan, the PEG-conjugated irinotecan derivative etirinotecan pegol (NKTR-102), which contains a 4-arm PEG polymer, a hydrolysable ester-based linker, and one irinotecan molecule at the end of each arm, has been designed and developed by Nektar Therapeutics. Study has demonstrated that NKTR-102 shows improved drug penetration into tumors leading to improved efficacy over irinotecan in a series of mouse models of human cancers, and it even exhibits a good result in phase II clinical trial, indicating that PEGylation of irinotecan is a feasible method to improve the antitumor activity and toxicity, though the phase III study fails to meet its prespecified response rate endpoint. However, the failure of the phase III study of NKTR-102 seems to demonstrate that the extent of passive tumor-targeting alone by enhanced permeation and retention of PEGylation is still limited. It is assumed that the further modification of NKTR-102 with active targeted moiety may lead to better efficiency and lower toxicity. The arginine-glycine-aspartic acid (RGD) peptide is a cell adhesion motif that can forwardly interact efficiently with the overexpressed integrin receptors (mainly αv3), which plays a major role in tumor-induced angiogenesis, tumor neovascularization, and tumor metastasis. Moreover, cyclic RGD (cRGD) peptide, which has been proven to be a more efficient tumor-targeting ligand in comparison with linear RGD peptide, has been widely used for the delivery of anticancer drugs to tumors. Previous work had demonstrated that the introduction of cRGD peptide to CPT scaffold may effectively improve the antitumor activity, receptor affinity (mainly αv3) and tumor cell adhesion. Herein, inspiring by the structure of NKTR-102, PEG linker was used as passive tumor targeting ligand to functionalize irinotecan (CPT derivative), while cRGD was designed as active tumor targeting moiety. It is expected that the combination of PEGylation and cRGD may lead to better enhanced permeation and retention, which may result to better efficiency and lower toxicity of irinotecan. Therefore, in the present work, a novel PEG-cRGD-conjugated irinotecan derivative was designed and synthesized. However, to the best of our knowledge, irinotecan simultaneously elaborated with cRGD and PEGylation has not been reported. The in vivo and in vitro antitumor activities, as well as the preliminary safety were also evaluated. Furthermore, integrin-binding competition between recombinant human αv3 and αv5 integrin, as well as chick chorioallantoic membrane (CAM) angiogenesis assays were carried out to evaluated the action mechanism. Finally, preliminary pharmacokinetic study of the PEG-cRGD-conjugated irinotecan derivative in the whole blood was performed.
Click to Show/Hide
|
||||
| Description |
Compared with irinotecan and NKTR-102, compound BGC0222 exhibited better antiproliferative activity in all these assayed cell lines. In the antiproliferative activity assay with the HT29 cell line, compound BGC0222 displayed higher antiproliferative activity than irinotecan (IC50 = 7.64 0.19 μM) and NKTR-102 (IC50 = 2.02 0.10 μM), with IC50 value of 1.83 0.09 μM. In addition, compound BGC0222 showed better antiproliferative activity than irinotecan (IC50 = 16.2 0.22 μM) and NKTR-102 (IC50 = 4.68 0.15 μM) against MIA PaCa-2 cells line, with IC50 value of 3.95 0.16 μM, while compound BGC0222 also exhibited stronger antiproliferative activity than irinotecan (IC50 = 2.69 0.12 μM) and NKTR-102 (IC50 = 0.71 0.05 μM) in MCF-7 cells line assay, with IC50 value of 0.68 0.04 μM, respectively. Clearly, the order of in vitro antiproliferative effect against the assay cell lines was: BGC0222>NKTR-102 > irinotecan. The structure-activity relationship was then investigated. The success of NKTR-102 proved that the introducing of PEG should lead to better antiproliferative activity. In addition, by the comparison of the structure of BGC0222 with that of NKTR-102, it should be found the presence of cRGD lead to better antiproliferative effect. Therefore, based on the above observation, it could be concluded that the introduction of PEG and cRGD to irinotecan should efficiently improve the antiproliferative effect, consistent with our expectation.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | ||
References
