Drug Information
General Information of This Drug
| Drug ID | DRG00223 | |||||
|---|---|---|---|---|---|---|
| Drug Name | 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid | |||||
| Synonyms |
4-(4-oxo-3,4-dihydroquinazolin-2-yl)butanoic acid; 95494-51-2; 4-(4-oxo-3H-quinazolin-2-yl)butanoic acid; MFCD05273570; 4-(4-Oxo-3,4-dihydro-quinazolin-2-yl)-butyric acid; CHEMBL3397958; 4-(4-oxo-1,4-dihydroquinazolin-2-yl)butanoic acid; NSC99368; 2-Quinazolinebutanoic acid, 1,4-dihydro-4-oxo-; SCHEMBL15575028; DTXSID40295034; IWUFDMAFCDCNMQ-UHFFFAOYSA-N; ALBB-000352; BBL030359; BDBM50285381; NSC-99368; STK502242; STK693864; STL521284; AKOS000303290; AKOS005605738; AKOS022667064; VS-09761; 4-(4-hydroxyquinazolin-2-yl)butanoic acid; CS-0221602; EN300-09566; F72192; A859035; SR-01000353583; 4-(4-oxo-3,4-dihydroquinazolin-2-yl)butanoicacid; J-513565; SR-01000353583-1; Z56983044; 4-(4-oxo-3,4-dihydroquinazolin-2-yl)butanoic acid, AldrichCPR
Click to Show/Hide
|
|||||
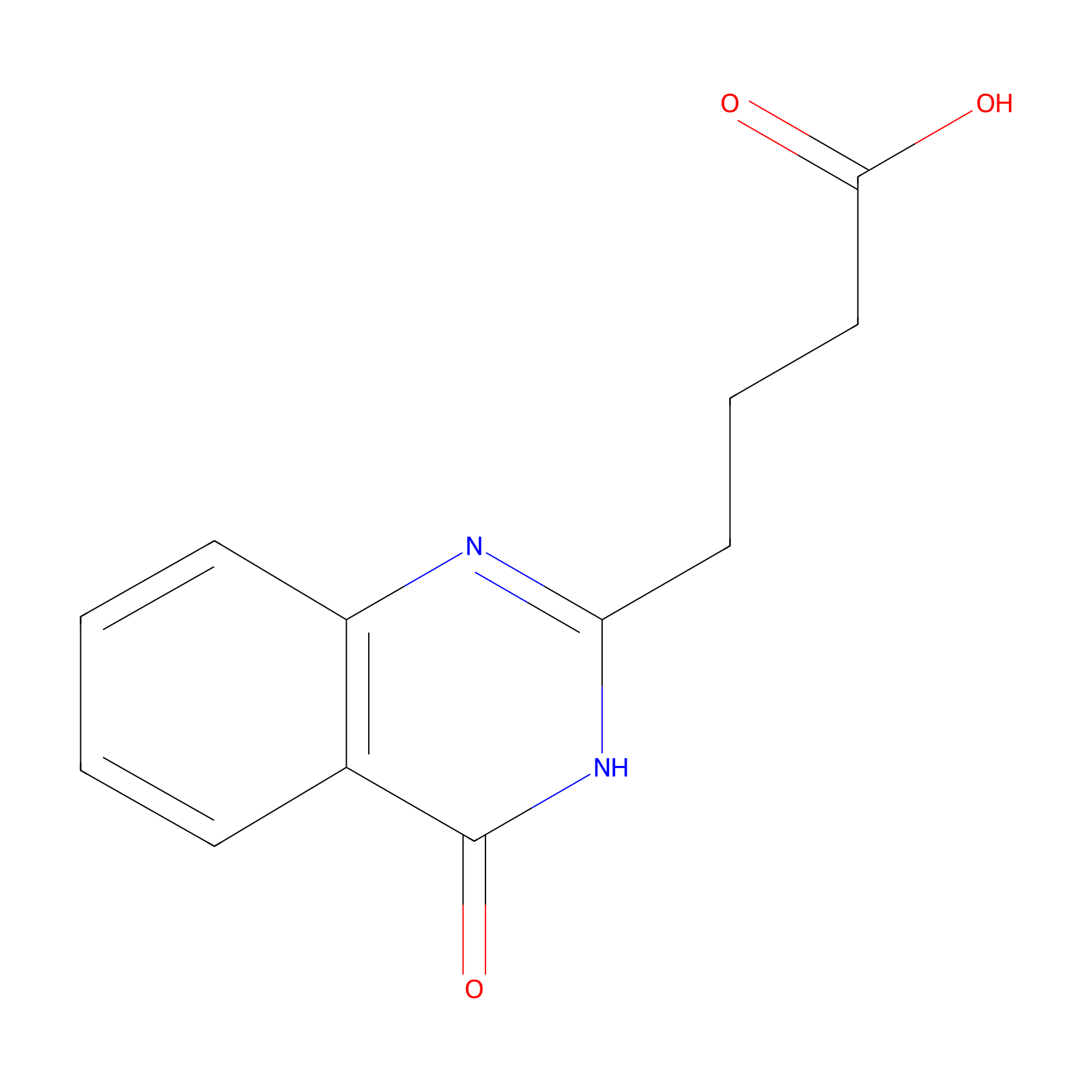
| Structure |
|
|||||
| Formula |
C12H12N2O3
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 232.23 | ||||
| Lipid-water partition coefficient (xlogp) | 0.5 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 4 | |||||
| Rotatable Bond Count (rotbonds) | 4 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C1=CC=C2C(=C1)C(=O)NC(=N2)CCCC(=O)O
|
|||||
| InChI |
InChI=1S/C12H12N2O3/c15-11(16)7-3-6-10-13-9-5-2-1-4-8(9)12(17)14-10/h1-2,4-5H,3,6-7H2,(H,15,16)(H,13,14,17)
|
|||||
| InChIKey |
IWUFDMAFCDCNMQ-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
4-(4-oxo-3H-quinazolin-2-yl)butanoic acid
|
|||||
Full Information of The Activity Data of The PDC(s) Related to This Drug
GDGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 13 ± 0.21 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 13 ± 0.34 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 14 ± 0.18 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 14 ± 0.48 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
GDGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 13 ± 0.44 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 14 ± 0.14 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 15 ± 0.24 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 16 ± 0.45 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
GTGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 13 ± 0.28 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 14 ± 0.36 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 14 ± 0.39 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 16 ± 0.59 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
GTGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 15 ± 0.24 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 16 ± 0.46 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 16 ± 0.49 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 17 ± 0.22 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
GDGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 7 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 21 ± 0.45 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 22 ± 0.38 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 23 ± 0.22 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 25 ± 0.42 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
GDGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 5 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 23 ± 0.47 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 23 ± 0.37 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 26 ± 0.36 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 27 ± 0.61 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
GWGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 24 ± 0.36 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 26 ± 0.31 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 27 ± 0.29 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 27 ± 0.37 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
GYGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 24 ± 0.56 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 25 ± 0.25 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 26 ± 0.40 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 27 ± 0.36 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
GYGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 26 ± 0.12 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 28 ± 0.42 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 28 ± 0.36 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 29 ± 0.29 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
GWGVP - 3,4-Dihydro-4-oxo-2-quinazolinebutanoic acid conjugate 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Klebsiella pneumoniae</i> infection | ||||
| Efficacy Data | Zone of inhibition | 28 ± 0.33 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Klebsiella pneumoniae infection | Klebsiella pneumoniae | 573 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Fusarium moniliforme</i> infection | ||||
| Efficacy Data | Zone of inhibition | 29 ± 0.30 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Fusarium moniliforme infection | Fusarium moniliforme | 117187 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Escherichia coli</i> infection | ||||
| Efficacy Data | Zone of inhibition | 30 ± 0.39 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Escherichia coli infection | Escherichia coli | 562 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | <i>Aspergillus niger</i> infection | ||||
| Efficacy Data | Zone of inhibition | 30 ± 0.33 mm | |||
| Administration Time | 24 h | ||||
| Administration Dosage | 30 μg/mL | ||||
| MOA of PDC |
We designed novel low charge, high hydrophobic pentapeptides with varying amino acids in the 2nd position and conjugated them to quinazolinone analogues to produce antimicrobial candidates. Preliminary structure-activity relationship revealed that constructs containing tryptophan, tyrosine, and phenylalanine amino acids were better antimicrobial agents than the standards used. Most of the candidates showed selective inhibitory power towards Gram-negative microorganisms. Most of the compounds could be considered as anti-Fusarium monoliforme since slightly superior results were observed for this species compared to A. niger. The alkyl chain length of the heterocyclic unit was found to be crucial for good activity. Finally, the presence of C-terminal polar group (COOH) seems to be favorable for antimicrobial results. Generating such hybrid compounds can be a promising approach to develop good desired biological activities.
Click to Show/Hide
|
||||
| Description |
The activity results showed that most of the compounds produced significant effects on the growth of the tested bacterial and fungal strains. The structure- antimicrobial activity relationship of the compounds revealed that quinazolinone precursors (I) and (II) conjugated with GWGVP ((XV), (XX), (XXV), and (XXX)), GYGVP ((XVI), (XXI), (XXVI), and (XXXI)), and GFGVP ((XXVII), (XXII), (XXVII), and (XXXII)) showed potent activities compared to conjugates of GDGVP ((XXVIII), (XXIII), (XXVIII), and (XXXIII)) and GTGVP ((XIX), XXIV), (XXIX), and (XXXIV)) and respective standard drugs. This could be due to aromaticity of the amino acids Trp, Tyr, and Phe, which are considered to play an important role in antimicrobial effects. It can be suggested that the presence of more hydrophobic units along with other non-polar amino acids provide amphipathicity to the compounds, which may help them in binding to the bacterial cell membranes followed by disruption. This becomes more evident with the loss of activity when the three aromatic and hydrophobic amino acids are substituted by more polar and charged Asp and Thr units. Among the above three hybrid analogues, tryptophan conjugates exhibited high potency, which may be due to the large aromaticity, hydrophobicity, light instability, and the ability to stabilize the amphiphilic structure necessary for antimicrobial activity. Further debenzylation of these compounds ((XXV), (XXVI), (XXVII), (XXX), (XXXI), and (XXXII)) resulted in more polar structures, which showed slight superior results than the benzylated analogues (XV), (XVI), (XVII), (XX), (XXI), and (XXII). This increased activity could be due to their higher polarity, which helps the molecules to establish a larger network for penetration through the cell membranes of microbes. Further, the increase in chain length of quinazolinones decreased the activity. Overall, the antimicrobial activity decreases in the order of X = Trp > Tyr > Phe > Thr > Asp.
Click to Show/Hide
|
||||
| In Vitro Model | Aspergillus niger infection | Aspergillus niger | 5061 | ||
References
