Linker Information
General Information of This Linker
| Linker ID |
LIN00006
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
Gamma-Aminobutyric Acid
|
|||||
| Structure |
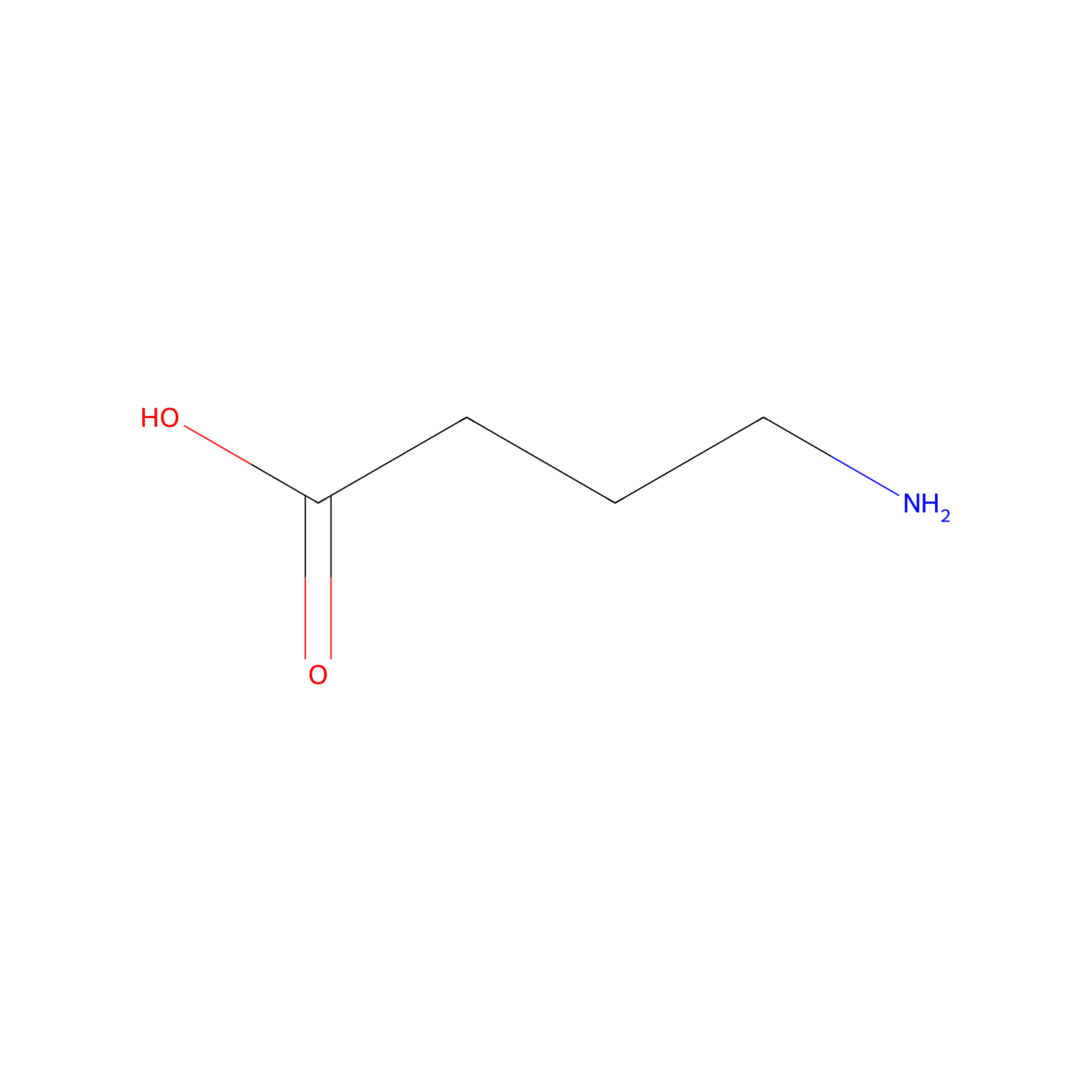
|
|||||
| Formula |
C4H9NO2
|
|||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 103.12 | ||||
| Lipid-water partition coefficient (xlogp) | -3.2 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||
| Rotatable Bond Count (rotbonds) | 3 | |||||
| Chemble ID | ||||||
| Chemble ID | ||||||
| PubChem CID | ||||||
| Canonical smiles |
C(CC(=O)O)CN
|
|||||
| InChI |
InChI=1S/C4H9NO2/c5-3-1-2-4(6)7/h1-3,5H2,(H,6,7)
|
|||||
| InChIKey |
BTCSSZJGUNDROE-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
4-aminobutanoic acid
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
Glypican-3-targeting peptide-chlorin e6 conjugates 8c [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.25 ± 0.03 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.31 ± 0.01 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.72 ± 0.12 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.88 ± 0.07 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.1 ± 0.9 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
Glypican-3-targeting peptide-chlorin e6 conjugates 8d [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.33 ± 0.01 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.34 ± 0.01 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.38 ± 0.03 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.57 ± 0.05 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.79 ± 0.01 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
Glypican-3-targeting peptide-chlorin e6 conjugates 8b [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.82 ± 0.06 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.4 ± 0.60 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
Glypican-3-targeting peptide-chlorin e6 conjugates 8a [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.7 ± 0.19 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.9 ± 0.44 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
4.7 ± 1.1 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
5.3 ± 1.0 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
9.8 ± 1.0 µM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Hepatoblastoma | L-O2 cell line | CVCL_0027 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Adult hepatocellular carcinoma | GPC3-positive Huh-7 cell | CVCL_0336 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | GPC3 negative MCF7 cell | CVCL_0031 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | GPC3-negative HeLa cell | CVCL_0030 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 200 µM | |||
| Administration Time | 24 h | ||||
| Evaluation Method | Annexin V/PI staining assay | ||||
| MOA of PDC |
Hepatocellular carcinoma (HCC) is a highly aggressive and lethal malignancy with poor prognosis, necessitating the urgent development of effective treatments. Targeted photodynamic therapy (PDT) offers a promising way to selectively eradicate tumor cells without affecting normal cells. Inspired by promising features of peptide-drug conjugates (PDCs) in targeted cancer therapy, herein a novel glypican-3 (GPC3)-targeting PDC-PDT strategy was developed for the precise PDT treatment of HCC. The GPC3-targeting photosensitizer conjugates were developed by attaching GPC3-targeting peptides to chlorin e6. Conjugate 8b demonstrated the ability to penetrate HCC cells via GPC3-mediated entry process, exhibiting remarkable tumor-targeting capacity, superior antitumor efficacy, and minimal toxicity towards normal cells. Notably, conjugate 8b achieved complete tumor elimination upon light illumination in a HepG2 xenograft model without harm to normal tissues. Overall, this innovative GPC3-targeting conjugation strategy demonstrates considerable promise for clinical applications for the treatment of HCC.
Click to Show/Hide
|
||||
| Description |
Subsequently, the antiproliferative activities of the PS conjugates, including dark toxicity and phototoxicity (660 nM, light dose: 10 J/cm2), were evaluated. Dark toxicity refers to the toxicity of PS conjugates against cancer cells in the absence of light and can be regarded as a crucial measure of the safety profile, while phototoxicity is the most direct indicator of the PDT efficacy of PS. The results showed that the dark toxicity of all the compounds against the five cell lines was comparatively low, demonstrating that the PS conjugates exhibited a high safety index in the absence of light. Compound 8b exhibited the optimum tumor cell selectivity in terms of phototoxicity, with the IC50 values of 0.82 uM against HepG2 cells and 2.4 uM against Huh-7 cells, respectively, whereas its toxicity against GPC3-negative cell lines was >10 uM. Although compounds 8a and 8c-d exhibited excellent phototoxicity against HepG2 and Huh-7 cells, (IC50 range: 0.2-2.0 uM), these compounds lacked selectivity between GPC3-overexpressed and GPC3-negative cells because they also showed considerable phototoxicity against the cell lines with minimal GPC3 expression. Notably, compound 1 showed low phototoxicity and selectivity against all the tested cell lines (IC50 > 10 uM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
P6-chlorambucil [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
22.00%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
Conjugation of the drugs to P4 affected their efficacy toward A20 cells. For chlorambucil and melphalan, conjugation reduced the cytotoxic effect and this was significant for chlorambucil at 25 μM (p = 0.0013). On the other hand, conjugation significantly improved the cytotoxic effect of bendamustine at 25 (p = 0.043) and 50 μM (p = 0.048). The efficacies of all P6-conjugates were significantly lower than those of P4-conjugates at concentrations above 10 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
P6-bendamustine [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
28.00%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
Conjugation of the drugs to P4 affected their efficacy toward A20 cells. For chlorambucil and melphalan, conjugation reduced the cytotoxic effect and this was significant for chlorambucil at 25 μM (p = 0.0013). On the other hand, conjugation significantly improved the cytotoxic effect of bendamustine at 25 (p = 0.043) and 50 μM (p = 0.048). The efficacies of all P6-conjugates were significantly lower than those of P4-conjugates at concentrations above 10 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
P4-Bend-PEG-AuNP [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
30.00%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
75.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
80.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
P4-Chlor-PEG-AuNP [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
40.00%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
72.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| MOA of PDC |
In biological systems, antioxidants such as catalase, superoxide dismutase, glutathione peroxidase, and glutathione reductase are responsible for the elimination or reduction of the adverse effects of ROS, that is, they prevent or reduce ROS generation. Dietary antioxidants, such as vitamins E, A, and C, and anthocyanins and polyphenols have a role in the protection of cells against ROS damage.
Click to Show/Hide
|
||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
80.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
P4-chlorambucil [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
70.00%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
Conjugation of the drugs to P4 affected their efficacy toward A20 cells. For chlorambucil and melphalan, conjugation reduced the cytotoxic effect and this was significant for chlorambucil at 25 μM (p = 0.0013). On the other hand, conjugation significantly improved the cytotoxic effect of bendamustine at 25 (p = 0.043) and 50 μM (p = 0.048). The efficacies of all P6-conjugates were significantly lower than those of P4-conjugates at concentrations above 10 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
P4-bendamustine [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition |
70.00%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | XTT assay | ||||
| Description |
Conjugation of the drugs to P4 affected their efficacy toward A20 cells. For chlorambucil and melphalan, conjugation reduced the cytotoxic effect and this was significant for chlorambucil at 25 μM (p = 0.0013). On the other hand, conjugation significantly improved the cytotoxic effect of bendamustine at 25 (p = 0.043) and 50 μM (p = 0.048). The efficacies of all P6-conjugates were significantly lower than those of P4-conjugates at concentrations above 10 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
References
