Linker Information
General Information of This Linker
| Linker ID |
LIN00021
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
GSHG
|
|||||
| Linker Type |
Enzyme-sensitive linkers
|
|||||
| Structure |
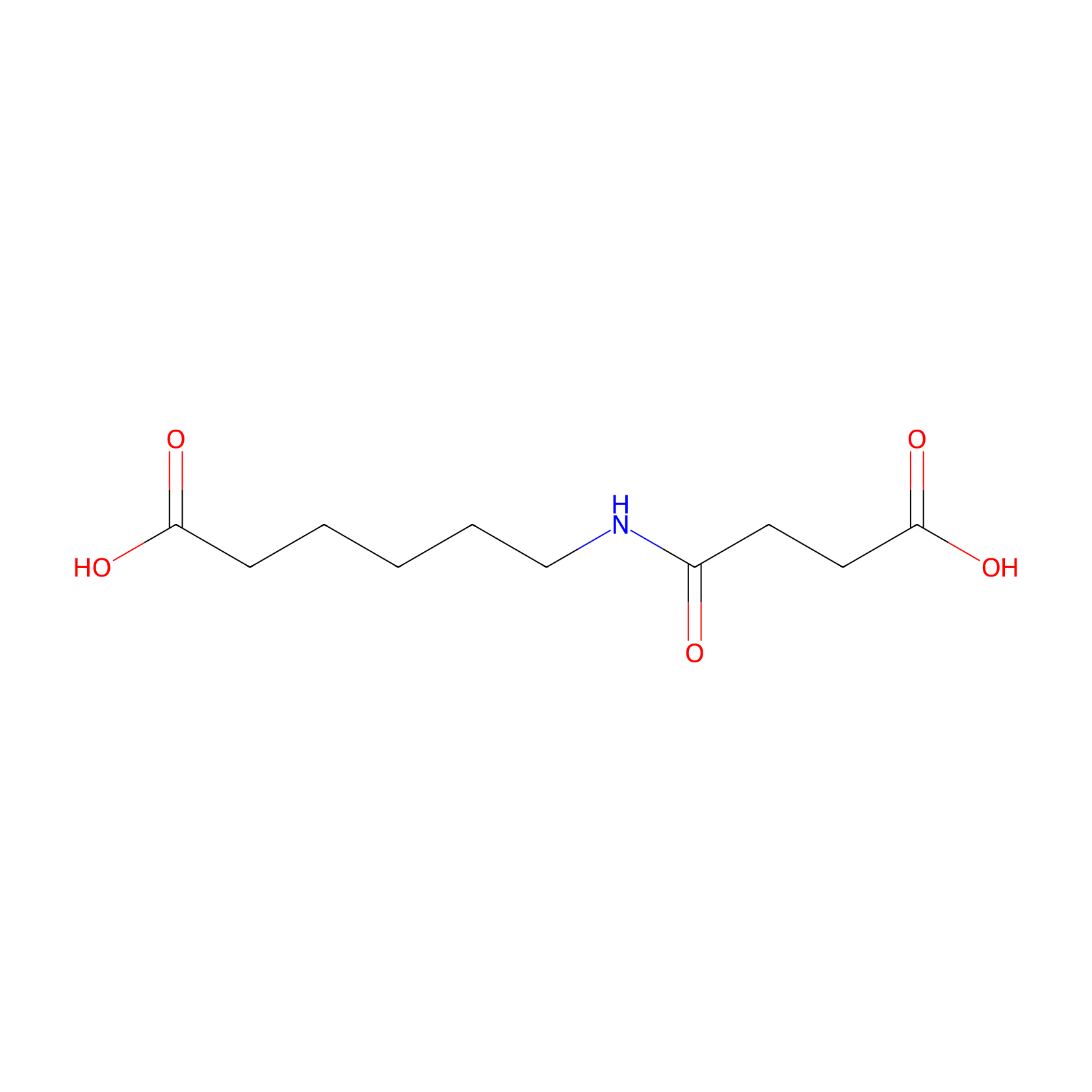
|
|||||
| Formula |
C10H17NO5
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 231.25 | ||||
| Lipid-water partition coefficient (xlogp) | -0.5 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | |||||
| Rotatable Bond Count (rotbonds) | 9 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C(CCC(=O)O)CCNC(=O)CCC(=O)O
|
|||||
| InChI |
InChI=1S/C10H17NO5/c12-8(5-6-10(15)16)11-7-3-1-2-4-9(13)14/h1-7H2,(H,11,12)(H,13,14)(H,15,16)
|
|||||
| InChIKey |
MFPUVZGABREEIK-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
6-(3-carboxypropanoylamino)hexanoic acid
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
Cq-C10-TP10 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.8 ± 0.3 µM
|
|||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| In Vivo Model | Plasmodium falciparum 3D7. | ||||
D-Lys6-GnRH-gemcitabine(GSHG) [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2.40 nM
|
|||
| Description |
The presented data show that 2G2, 2G1 and GSHG bind to GnRH-R with 95.5-, 15.2-, and 4.4-fold higher affinity, respectively, than that of the native peptide D-Lys6-GnRH (10.5 ± 0.2 nM, according to our former study [3]).
|
||||
| In Vitro Model | Normal | HEK293 cell | CVCL_0045 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
55.5 nM
|
|||
| Description |
GSHGpossesses the highest cytotoxic effect among the three conjugates, which is comparable with that of gemcitabine in the examined cell lines and especially regarding MCF-7 cells.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
684 nM
|
|||
| Description |
GSHGpossesses the highest cytotoxic effect among the three conjugates, which is comparable with that of gemcitabine in the examined cell lines and especially regarding MCF-7 cells.
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
937 nM
|
|||
| Description |
GSHGpossesses the highest cytotoxic effect among the three conjugates, which is comparable with that of gemcitabine in the examined cell lines and especially regarding MCF-7 cells.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
2387 nM
|
|||
| Description |
GSHGpossesses the highest cytotoxic effect among the three conjugates, which is comparable with that of gemcitabine in the examined cell lines and especially regarding MCF-7 cells.
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
References
