Linker Information
General Information of This Linker
| Linker ID |
LIN00046
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
3-(3-((2-(carboxyamino)ethyl)disulfaneyl)propanamido)propanoic acid
|
|||||
| Linker Type |
GSH concentration-sensitive linkers
|
|||||
| Structure |
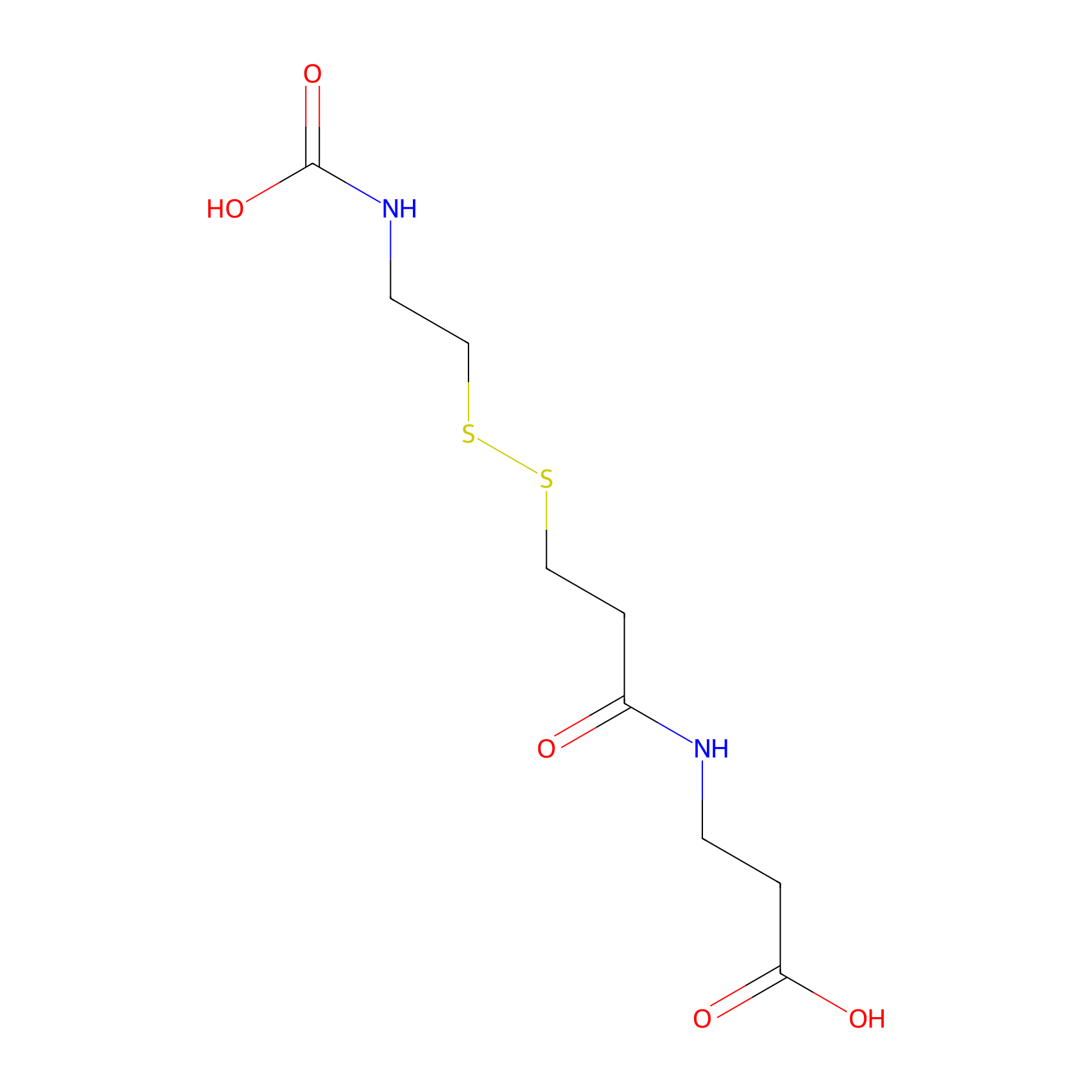
|
|||||
| Formula |
C9H16N2O5S2
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 296.37 | ||||
| Lipid-water partition coefficient (xlogp) | 0.6164 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 4 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | |||||
| Rotatable Bond Count (rotbonds) | 10 | |||||
| Canonical smiles |
O=C(O)CCNC(=O)CCSSCCNC(=O)O
|
|||||
| InChI |
InChI=1S/C9H16N2O5S2/c12-7(10-3-1-8(13)14)2-5-17-18-6-4-11-9(15)16/h11H,1-6H2,(H,10,12)(H,13,14)(H,15,16)
|
|||||
| InChIKey |
MTVICSBYAXGEPU-UHFFFAOYSA-N
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
PDC-CPT2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
80%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
Antiproliferative results showed that PTX1 inhibited cell proliferation by 18.7%. The anti-proliferative activity of CPT1 was diminished by 1.9-fold as compared to CPT whereas the activity of CPT2 was comparable to CPT, since CPT2 reduced the cell viability to 61%.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
References
