Linker Information
General Information of This Linker
| Linker ID |
LIN00047
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
AHX
|
|||||
| Linker Type |
Enzyme-sensitive linkers
|
|||||
| Structure |
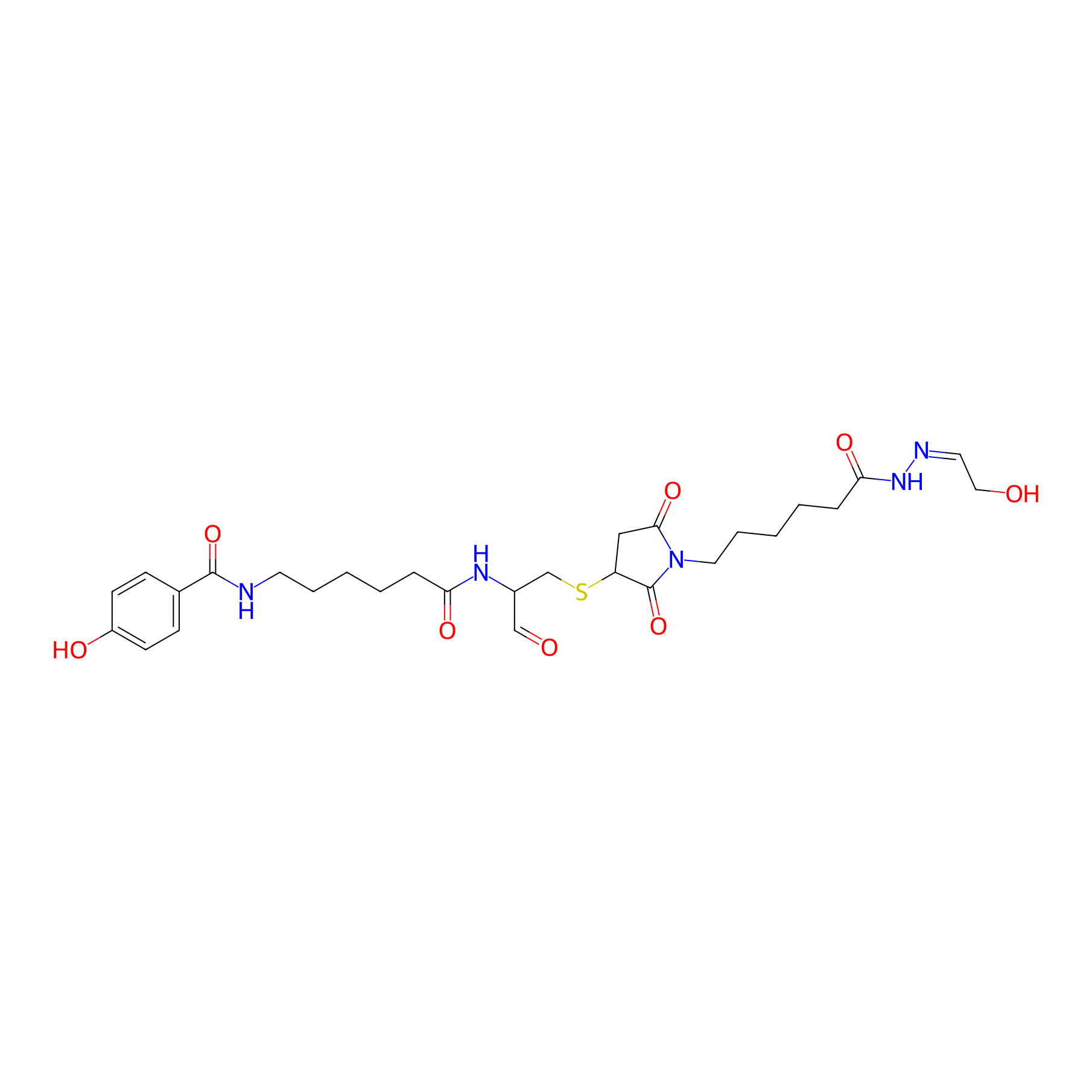
|
|||||
| Formula |
C28H39N5O8S
|
|||||
| #Ro5 Violations (Lipinski): 2 | Molecular Weight (mw) | 605.714 | ||||
| Lipid-water partition coefficient (xlogp) | 0.8815 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 5 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 10 | |||||
| Rotatable Bond Count (rotbonds) | 20 | |||||
| Canonical smiles |
O=CC(CSC1CC(=O)N(CCCCCC(=O)NN=CCO)C1=O)NC(=O)CCCCCNC(=O)c1ccc(O)cc1
|
|||||
| InChI |
InChI=1S/C28H39N5O8S/c34-16-14-30-32-25(38)8-4-2-6-15-33-26(39)17-23(28(33)41)42-19-21(18-35)31-24(37)7-3-1-5-13-29-27(40)20-9-11-22(36)12-10-20/h9-12,14,18,21,23,34,36H,1-8,13,15-17,19H2,(H,29,40)(H,31,37)(H,32,38)/b30-14-
|
|||||
| InChIKey |
ZTRDUNVKNZLLGZ-CPDSRJINSA-N
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
pHA-AHX-VAP-DOX [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.56 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The existence of the blood-brain barrier (BBB) and blood-brain tumor barrier (BBTB) greatly limits the application of chemotherapy in glioma. To address this challenge, an optimal drug delivery system must efficiently cross the BBB/BBTB and specifically deliver therapeutic drugs into glioma cells while minimizing systemic toxicity. Here we demonstrated that glucose-regulated protein 78 (GRP78) and dopamine receptor D2 were highly expressed in patient-derived glioma tissues, and dopamine receptors were highly expressed on the BBB. Subsequently, we synthesized a novel Y-shaped peptide and compared the effects of different linkers on the receptor affinity and targeting ability of the peptide. A peptide-drug conjugate (pHA-AOHX-VAP-doxorubicin conjugate, pHA-AOHX-VAP-DOX) with a better affinity for glioma cells and higher solubility was derived for glioma treatment. pHA-AOHX-VAP-DOX could cross both BBB and BBTB via dopamine receptor and GRP78 receptor, and finally target glioma cells, significantly prolonging the survival time of nude mice bearing intracranial glioma. Furthermore, pHA-AOHX-VAP-DOX significantly reduced the toxicity of DOX and increased the maximum tolerated dose (MTD). Collectively, this work paves a new avenue for overcoming multiple barriers and effectively delivering chemotherapeutic agents to glioma cells while providing key evidence to identify potential receptors for glioma-targeted drug delivery.
Click to Show/Hide
|
||||
| Description |
The coupling of peptides with chemotherapeutic drugs often increased the solubility of the drugs, so the solubility of peptide-drug conjugates was determined, and the results were shown in Fig. S9. DOX free base exhibited poor solubility, with a solubility of approximately 0.22 ± 0.03 mg/mL in PBS. However, upon formation of a peptide-drug conjugate, the hydrophilicity of the peptide significantly enhanced the solubility of DOX. The solubility of pHA-AHX-VAP-DOX and pHA-AOHX-VAP-DOX drastically increased to 7.09 ± 0.15 mg/mL and 17.29 ± 0.43 mg/mL, which was 32-fold and 78-fold higher than that of the DOX free base, respectively. The improved solubility performance was consistent with their LogP values predicted by the ALOGPS 2.1 program.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glioma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.62 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
The existence of the blood-brain barrier (BBB) and blood-brain tumor barrier (BBTB) greatly limits the application of chemotherapy in glioma. To address this challenge, an optimal drug delivery system must efficiently cross the BBB/BBTB and specifically deliver therapeutic drugs into glioma cells while minimizing systemic toxicity. Here we demonstrated that glucose-regulated protein 78 (GRP78) and dopamine receptor D2 were highly expressed in patient-derived glioma tissues, and dopamine receptors were highly expressed on the BBB. Subsequently, we synthesized a novel Y-shaped peptide and compared the effects of different linkers on the receptor affinity and targeting ability of the peptide. A peptide-drug conjugate (pHA-AOHX-VAP-doxorubicin conjugate, pHA-AOHX-VAP-DOX) with a better affinity for glioma cells and higher solubility was derived for glioma treatment. pHA-AOHX-VAP-DOX could cross both BBB and BBTB via dopamine receptor and GRP78 receptor, and finally target glioma cells, significantly prolonging the survival time of nude mice bearing intracranial glioma. Furthermore, pHA-AOHX-VAP-DOX significantly reduced the toxicity of DOX and increased the maximum tolerated dose (MTD). Collectively, this work paves a new avenue for overcoming multiple barriers and effectively delivering chemotherapeutic agents to glioma cells while providing key evidence to identify potential receptors for glioma-targeted drug delivery.
Click to Show/Hide
|
||||
| Description |
The coupling of peptides with chemotherapeutic drugs often increased the solubility of the drugs, so the solubility of peptide-drug conjugates was determined, and the results were shown in Fig. S9. DOX free base exhibited poor solubility, with a solubility of approximately 0.22 ± 0.03 mg/mL in PBS. However, upon formation of a peptide-drug conjugate, the hydrophilicity of the peptide significantly enhanced the solubility of DOX. The solubility of pHA-AHX-VAP-DOX and pHA-AOHX-VAP-DOX drastically increased to 7.09 ± 0.15 mg/mL and 17.29 ± 0.43 mg/mL, which was 32-fold and 78-fold higher than that of the DOX free base, respectively. The improved solubility performance was consistent with their LogP values predicted by the ALOGPS 2.1 program.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
References
