Linker Information
General Information of This Linker
| Linker ID |
LIN00063
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
Maleimidocaproyl (MC) moiety-valine-citrulline (VC) dipeptide-para-aminobenzyloxycarbamoyl (PABC) spacer (MC-VC-PABC)
|
|||||
| Linker Type |
Enzyme-sensitive linkers
|
|||||
| Structure |
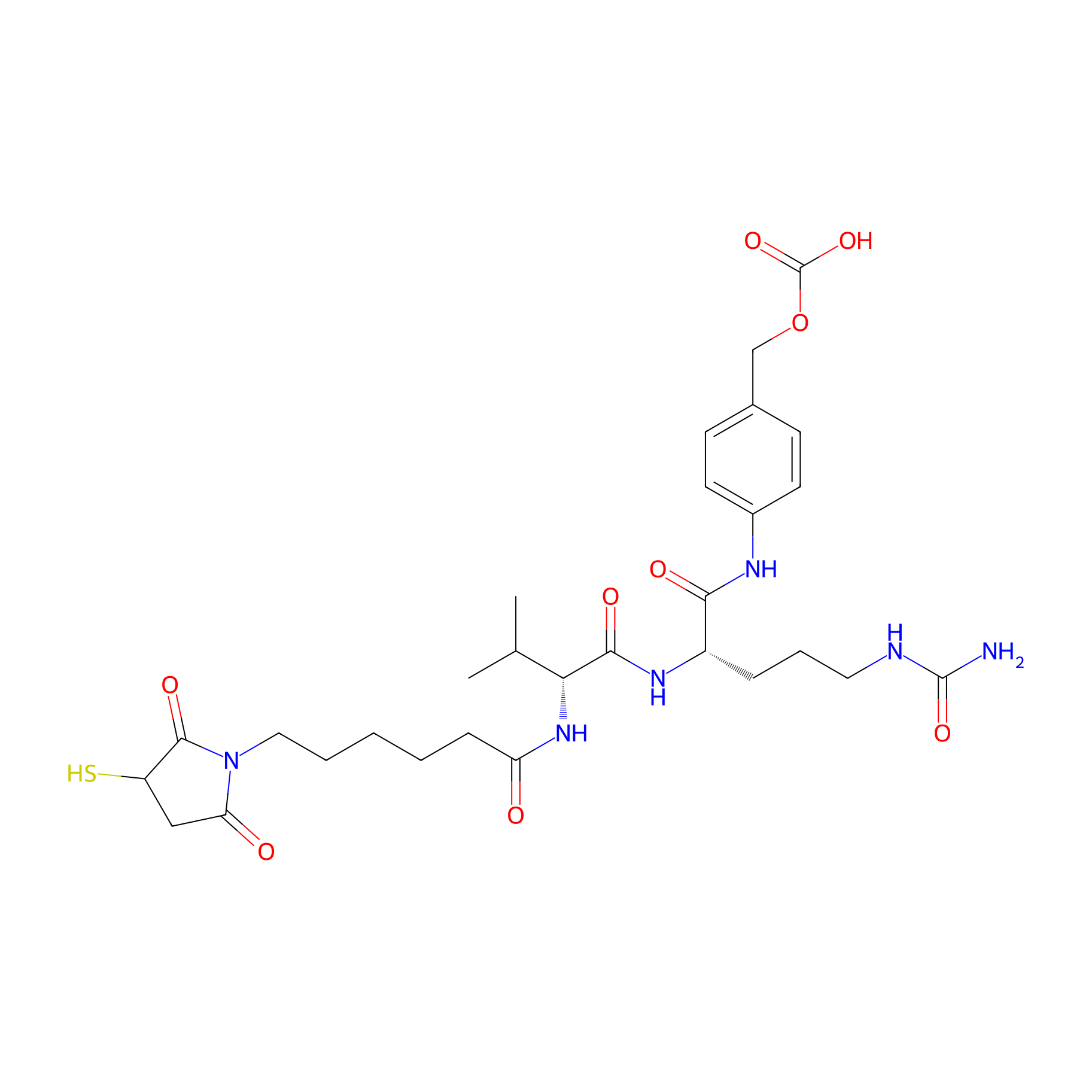
|
|||||
| Formula |
C29H42N6O9S
|
|||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight (mw) | 650.755 | ||||
| Lipid-water partition coefficient (xlogp) | 1.5115 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 7 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 9 | |||||
| Rotatable Bond Count (rotbonds) | 18 | |||||
| Canonical smiles |
CC(C)C(NC(=O)CCCCCN1C(=O)CC(S)C1=O)C(=O)NC(CCCNC(N)=O)C(=O)Nc1ccc(COC(=O)O)cc1
|
|||||
| InChI |
InChI=1S/C29H42N6O9S/c1-17(2)24(34-22(36)8-4-3-5-14-35-23(37)15-21(45)27(35)40)26(39)33-20(7-6-13-31-28(30)41)25(38)32-19-11-9-18(10-12-19)16-44-29(42)43/h9-12,17,20-21,24,45H,3-8,13-16H2,1-2H3,(H,32,38)(H,33,39)(H,34,36)(H,42,43)(H3,30,31,41)/t20-,21?,24+/m0/s1
|
|||||
| InChIKey |
LEBKZSAGLVOOHK-WRLRJORWSA-N
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
D-Cys6-LHRH vedotin [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
4 nM
|
|||
| Administration Time | 3 days | ||||
| MOA of PDC |
Ovarian cancer is the most deadly female gynaecological malignancy in developed countries and new treatments are urgently needed. The luteinising hormone releasing hormone (LHRH) peptide drug conjugate Zoptarelin doxorubicin is one such potential new drug modality that entered clinical trials for treating LHRH receptor-positive gynaecological cancers. However, development stopped after disappointing Phase 3 results in 2017. We believe the lack of efficacy was due to linker instability and payload potency. In this work, we replaced its linker-toxin with vedotin (MC-VC-PABC-MMAE), yielding the novel peptide drug conjugate D-Cys6-LHRH vedotin. A GI50 and cell specificity comparison against cancerous and non-cancerous ovarian cell lines showed significantly superior bioactivity and selectivity over Zoptarelin doxorubicin (GI50 4 vs. 453 nM) and other chemotherapeutic drugs used for treating ovarian cancers. Our results suggest D-Cys6-LHRH vedotin can potentially be used as a treatment for ovarian cancer.
Click to Show/Hide
|
||||
| Description |
GI50s are summarised in Table 1. Our cell panel consisted of LHRH receptor-positive (OVCAR-3) and receptor-negative (SK-OV-3) ovarian cancer cell lines used in Zoptarelin doxorubicin's preclinical development studies. Non-cancerous human ovarian (H-6036) and lung fibroblast (MRC-5) cell lines were also included for selectivity comparison. Paclitaxel, a first-line chemotherapeutic drug used for ovarian cancer, was observed to be highly cytotoxic against all cell lines, including non-cancerous ovarian and human fibroblasts (GI50s 1-5 nM). Unsurprisingly, common and serious side-effects experienced by more than 50% patients dosed with this drug included anaemia, neutropenia, peripheral neuropathy, nausea, vomiting, myalgia, arthralgia and alopecia. Similarly, doxorubicin was also shown to be non-specific towards all the cell lines (GI50s 60-311 nM), below the reported average 631 nM GI50 in a study involving 39 cancer cell lines. It is noteworthy that doxorubicin exhibited moderate GI50s of 311 and 218 nM against OVCAR-3 and SK-OV-3, respectively, supporting earlier in vitro data. Tubulin binder MMAE was found to be ultra-toxic against all cell lines (GI50s 0.5-1.1 nM), supporting the findings from an earlier report. It is therefore unsurprising that MMAE has never been approved as a drug on its own. The LHRH analog and carrier peptide, D-Cys6-LHRH, was inactive against all cell lines (GI50s >50,000 nM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
49 nM
|
|||
| Administration Time | 3 days | ||||
| MOA of PDC |
Ovarian cancer is the most deadly female gynaecological malignancy in developed countries and new treatments are urgently needed. The luteinising hormone releasing hormone (LHRH) peptide drug conjugate Zoptarelin doxorubicin is one such potential new drug modality that entered clinical trials for treating LHRH receptor-positive gynaecological cancers. However, development stopped after disappointing Phase 3 results in 2017. We believe the lack of efficacy was due to linker instability and payload potency. In this work, we replaced its linker-toxin with vedotin (MC-VC-PABC-MMAE), yielding the novel peptide drug conjugate D-Cys6-LHRH vedotin. A GI50 and cell specificity comparison against cancerous and non-cancerous ovarian cell lines showed significantly superior bioactivity and selectivity over Zoptarelin doxorubicin (GI50 4 vs. 453 nM) and other chemotherapeutic drugs used for treating ovarian cancers. Our results suggest D-Cys6-LHRH vedotin can potentially be used as a treatment for ovarian cancer.
Click to Show/Hide
|
||||
| Description |
GI50s are summarised in Table 1. Our cell panel consisted of LHRH receptor-positive (OVCAR-3) and receptor-negative (SK-OV-3) ovarian cancer cell lines used in Zoptarelin doxorubicin's preclinical development studies. Non-cancerous human ovarian (H-6036) and lung fibroblast (MRC-5) cell lines were also included for selectivity comparison. Paclitaxel, a first-line chemotherapeutic drug used for ovarian cancer, was observed to be highly cytotoxic against all cell lines, including non-cancerous ovarian and human fibroblasts (GI50s 1-5 nM). Unsurprisingly, common and serious side-effects experienced by more than 50% patients dosed with this drug included anaemia, neutropenia, peripheral neuropathy, nausea, vomiting, myalgia, arthralgia and alopecia. Similarly, doxorubicin was also shown to be non-specific towards all the cell lines (GI50s 60-311 nM), below the reported average 631 nM GI50 in a study involving 39 cancer cell lines. It is noteworthy that doxorubicin exhibited moderate GI50s of 311 and 218 nM against OVCAR-3 and SK-OV-3, respectively, supporting earlier in vitro data. Tubulin binder MMAE was found to be ultra-toxic against all cell lines (GI50s 0.5-1.1 nM), supporting the findings from an earlier report. It is therefore unsurprising that MMAE has never been approved as a drug on its own. The LHRH analog and carrier peptide, D-Cys6-LHRH, was inactive against all cell lines (GI50s >50,000 nM).
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
52 nM
|
|||
| Administration Time | 3 days | ||||
| MOA of PDC |
Ovarian cancer is the most deadly female gynaecological malignancy in developed countries and new treatments are urgently needed. The luteinising hormone releasing hormone (LHRH) peptide drug conjugate Zoptarelin doxorubicin is one such potential new drug modality that entered clinical trials for treating LHRH receptor-positive gynaecological cancers. However, development stopped after disappointing Phase 3 results in 2017. We believe the lack of efficacy was due to linker instability and payload potency. In this work, we replaced its linker-toxin with vedotin (MC-VC-PABC-MMAE), yielding the novel peptide drug conjugate D-Cys6-LHRH vedotin. A GI50 and cell specificity comparison against cancerous and non-cancerous ovarian cell lines showed significantly superior bioactivity and selectivity over Zoptarelin doxorubicin (GI50 4 vs. 453 nM) and other chemotherapeutic drugs used for treating ovarian cancers. Our results suggest D-Cys6-LHRH vedotin can potentially be used as a treatment for ovarian cancer.
Click to Show/Hide
|
||||
| Description |
GI50s are summarised in Table 1. Our cell panel consisted of LHRH receptor-positive (OVCAR-3) and receptor-negative (SK-OV-3) ovarian cancer cell lines used in Zoptarelin doxorubicin's preclinical development studies. Non-cancerous human ovarian (H-6036) and lung fibroblast (MRC-5) cell lines were also included for selectivity comparison. Paclitaxel, a first-line chemotherapeutic drug used for ovarian cancer, was observed to be highly cytotoxic against all cell lines, including non-cancerous ovarian and human fibroblasts (GI50s 1-5 nM). Unsurprisingly, common and serious side-effects experienced by more than 50% patients dosed with this drug included anaemia, neutropenia, peripheral neuropathy, nausea, vomiting, myalgia, arthralgia and alopecia. Similarly, doxorubicin was also shown to be non-specific towards all the cell lines (GI50s 60-311 nM), below the reported average 631 nM GI50 in a study involving 39 cancer cell lines. It is noteworthy that doxorubicin exhibited moderate GI50s of 311 and 218 nM against OVCAR-3 and SK-OV-3, respectively, supporting earlier in vitro data. Tubulin binder MMAE was found to be ultra-toxic against all cell lines (GI50s 0.5-1.1 nM), supporting the findings from an earlier report. It is therefore unsurprising that MMAE has never been approved as a drug on its own. The LHRH analog and carrier peptide, D-Cys6-LHRH, was inactive against all cell lines (GI50s >50,000 nM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | H-6036 cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Half Maximal growth inhibition concentration (GI50) |
191 nM
|
|||
| Administration Time | 3 days | ||||
| MOA of PDC |
Ovarian cancer is the most deadly female gynaecological malignancy in developed countries and new treatments are urgently needed. The luteinising hormone releasing hormone (LHRH) peptide drug conjugate Zoptarelin doxorubicin is one such potential new drug modality that entered clinical trials for treating LHRH receptor-positive gynaecological cancers. However, development stopped after disappointing Phase 3 results in 2017. We believe the lack of efficacy was due to linker instability and payload potency. In this work, we replaced its linker-toxin with vedotin (MC-VC-PABC-MMAE), yielding the novel peptide drug conjugate D-Cys6-LHRH vedotin. A GI50 and cell specificity comparison against cancerous and non-cancerous ovarian cell lines showed significantly superior bioactivity and selectivity over Zoptarelin doxorubicin (GI50 4 vs. 453 nM) and other chemotherapeutic drugs used for treating ovarian cancers. Our results suggest D-Cys6-LHRH vedotin can potentially be used as a treatment for ovarian cancer.
Click to Show/Hide
|
||||
| Description |
GI50s are summarised in Table 1. Our cell panel consisted of LHRH receptor-positive (OVCAR-3) and receptor-negative (SK-OV-3) ovarian cancer cell lines used in Zoptarelin doxorubicin's preclinical development studies. Non-cancerous human ovarian (H-6036) and lung fibroblast (MRC-5) cell lines were also included for selectivity comparison. Paclitaxel, a first-line chemotherapeutic drug used for ovarian cancer, was observed to be highly cytotoxic against all cell lines, including non-cancerous ovarian and human fibroblasts (GI50s 1-5 nM). Unsurprisingly, common and serious side-effects experienced by more than 50% patients dosed with this drug included anaemia, neutropenia, peripheral neuropathy, nausea, vomiting, myalgia, arthralgia and alopecia. Similarly, doxorubicin was also shown to be non-specific towards all the cell lines (GI50s 60-311 nM), below the reported average 631 nM GI50 in a study involving 39 cancer cell lines. It is noteworthy that doxorubicin exhibited moderate GI50s of 311 and 218 nM against OVCAR-3 and SK-OV-3, respectively, supporting earlier in vitro data. Tubulin binder MMAE was found to be ultra-toxic against all cell lines (GI50s 0.5-1.1 nM), supporting the findings from an earlier report. It is therefore unsurprising that MMAE has never been approved as a drug on its own. The LHRH analog and carrier peptide, D-Cys6-LHRH, was inactive against all cell lines (GI50s >50,000 nM).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MRC-5 cell | CVCL_0440 | ||
References
