Linker Information
General Information of This Linker
| Linker ID |
LIN00149
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
3-Disulfaneylpropanoic acid
|
|||||
| Linker Type |
GSH concentration-sensitive linkers
|
|||||
| Structure |
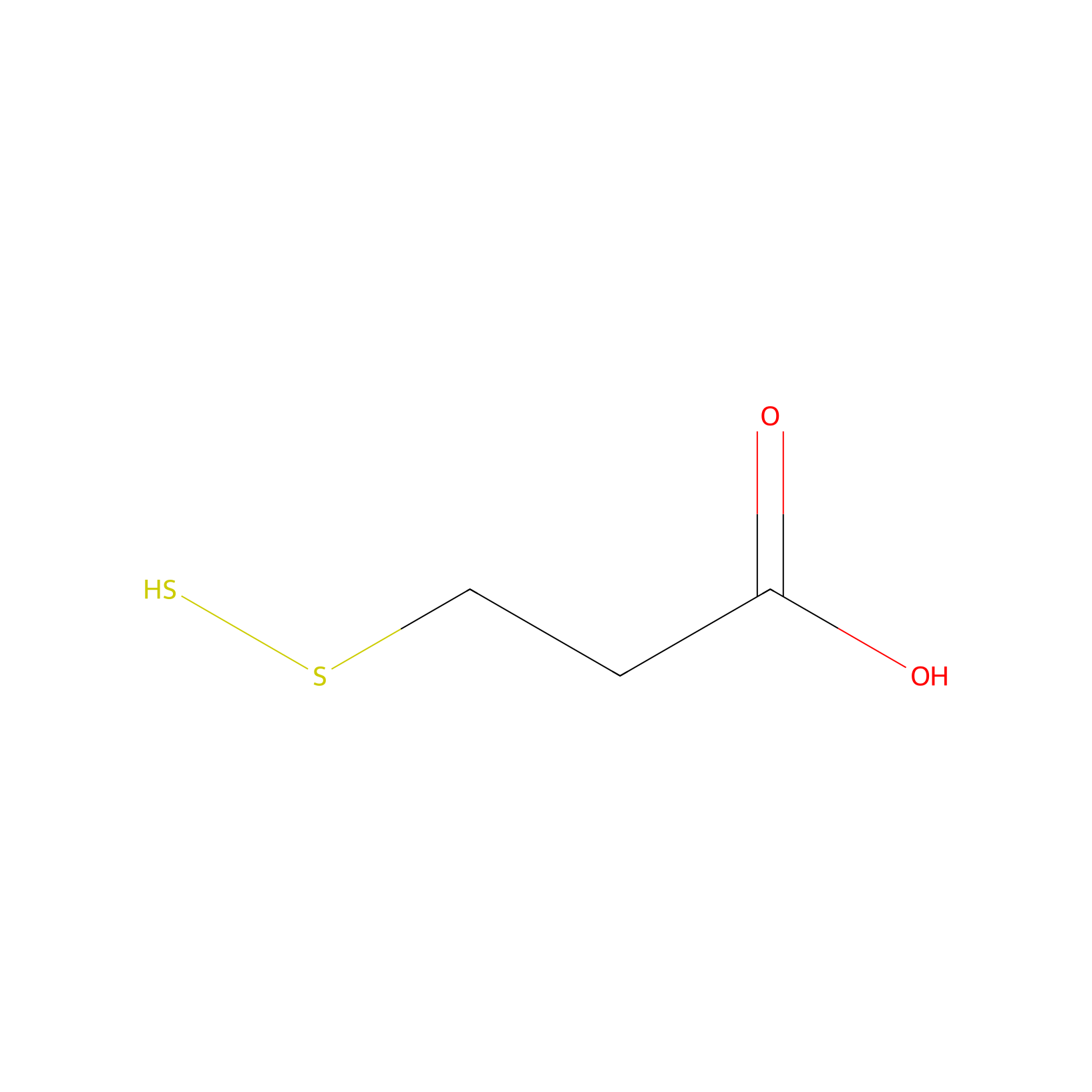
|
|||||
| Formula |
C3H6O2S2
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 138.213 | ||||
| Lipid-water partition coefficient (xlogp) | 1.0391 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||
| Rotatable Bond Count (rotbonds) | 3 | |||||
| Canonical smiles |
O=C(O)CCSS
|
|||||
| InChI |
InChI=1S/C3H6O2S2/c4-3(5)1-2-7-6/h6H,1-2H2,(H,4,5)
|
|||||
| InChIKey |
CXXDUKKBFUFJDF-UHFFFAOYSA-N
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
Sal-A6 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian serous cystadenocarcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
5.45 ± 0.41 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CCK8 assay | ||||
| MOA of PDC |
In this study, a disulfide bond was employed as a linker to couple peptide A6 with Sal, yielding the novel conjugate Sal-A6. Compared to Sal, Sal-A6 not only displayed increased activity and solubility but also demonstrated a notable improvement in targeting specificity. Additionally, Sal-A6 can overcome drug-resistance in cells, leading to a sensitization effect. Surprisingly, Sal-A6 also exhibited a bystander killing effect, capable of eliminating tumor cells with low CD44 expression. Furthermore, at lower doses, it exerts antitumor effects in vivo. The glutathione (GSH) content in tumor cells resistant to cisplatin (CDDP) is significantly higher than that in non-resistant tumor cells. GSH possesses a strong binding affinity with CDDP, resulting in the inactivation of CDDP. Nevertheless, Sal can effectively decrease the intracellular GSH content in tumor cells and elevate the level of intracellular reactive oxygen species (ROS). Moreover, the disulfide bond utilized in Sal-A6 can efficiently diminish the intra-cellular GSH content, thereby overcoming cellular resistance to CDDP and improving drug sensitivity.
Click to Show/Hide
|
||||
| Description |
Cell Counting Kit-8 (CCK8) was used to evaluate the in vitro anti-tumor activity and selectivity of Sal-A6 cell viability inhibitory effect of Sal, A6, compound E, and Sal-A6 was assessed in both CD44+ SKOV3 and CD44? A2780 cells. The results showed that peptide A6 exhibited no significant inhibitory effect on any tumor cells, in line with literature reports. Conversely, Sal and compound E significantly inhibited tumor cells proliferation while Sal-A6 conjugate exhibited the ability to suppress the cell viability of CD44+ SKOV3, demonstrating superior activity compared to Sal. However, it showed no significant inhibitory effect on CD44? A2780 cells, exerting only a moderate effect under high concentration conditions, possibly due to the target-mediated cellular entry of Sal-A6. To further validate whether the cytotoxicity of Sal-A6 is related to targeting via CD44, CD44+ SKOV3 cells co-incubated with A6 peptide and Sal-A6 at concentrations of 100 μM and 10 μM, respectively. Peptide A6 partially alleviated the cytotoxicity of Sal-A6, especially at saturated concentration (100 μM), leading to a significant reduction in the in vitro activity of Sal-A6 at both low (10 μM) and high (100 μM) concentrations. These results suggest that cells with low CD44 expression or treated with peptide A6 can mitigate the cellular inhibitory effects of Sal-A6. The onset of action of Sal-A6 was slower than that of Sal, which may be due to the fact that Sal-A6 exerts its efficacy through receptor-mediated entry into the cell and has a slower onset of action than the highly fat-soluble Sal. Overall, the competitive binding of cells with low CD44 expression and peptide A6 confirms that the in vitro activity of Sal-A6 is related to CD44 expression.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
47.09 ± 2.69 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CCK8 assay | ||||
| MOA of PDC |
In this study, a disulfide bond was employed as a linker to couple peptide A6 with Sal, yielding the novel conjugate Sal-A6. Compared to Sal, Sal-A6 not only displayed increased activity and solubility but also demonstrated a notable improvement in targeting specificity. Additionally, Sal-A6 can overcome drug-resistance in cells, leading to a sensitization effect. Surprisingly, Sal-A6 also exhibited a bystander killing effect, capable of eliminating tumor cells with low CD44 expression. Furthermore, at lower doses, it exerts antitumor effects in vivo. The glutathione (GSH) content in tumor cells resistant to cisplatin (CDDP) is significantly higher than that in non-resistant tumor cells. GSH possesses a strong binding affinity with CDDP, resulting in the inactivation of CDDP. Nevertheless, Sal can effectively decrease the intracellular GSH content in tumor cells and elevate the level of intracellular reactive oxygen species (ROS). Moreover, the disulfide bond utilized in Sal-A6 can efficiently diminish the intra-cellular GSH content, thereby overcoming cellular resistance to CDDP and improving drug sensitivity.
Click to Show/Hide
|
||||
| Description |
Cell Counting Kit-8 (CCK8) was used to evaluate the in vitro anti-tumor activity and selectivity of Sal-A6 cell viability inhibitory effect of Sal, A6, compound E, and Sal-A6 was assessed in both CD44+ SKOV3 and CD44? A2780 cells. The results showed that peptide A6 exhibited no significant inhibitory effect on any tumor cells, in line with literature reports. Conversely, Sal and compound E significantly inhibited tumor cells proliferation while Sal-A6 conjugate exhibited the ability to suppress the cell viability of CD44+ SKOV3, demonstrating superior activity compared to Sal. However, it showed no significant inhibitory effect on CD44? A2780 cells, exerting only a moderate effect under high concentration conditions, possibly due to the target-mediated cellular entry of Sal-A6. To further validate whether the cytotoxicity of Sal-A6 is related to targeting via CD44, CD44+ SKOV3 cells co-incubated with A6 peptide and Sal-A6 at concentrations of 100 μM and 10 μM, respectively. Peptide A6 partially alleviated the cytotoxicity of Sal-A6, especially at saturated concentration (100 μM), leading to a significant reduction in the in vitro activity of Sal-A6 at both low (10 μM) and high (100 μM) concentrations. These results suggest that cells with low CD44 expression or treated with peptide A6 can mitigate the cellular inhibitory effects of Sal-A6. The onset of action of Sal-A6 was slower than that of Sal, which may be due to the fact that Sal-A6 exerts its efficacy through receptor-mediated entry into the cell and has a slower onset of action than the highly fat-soluble Sal. Overall, the competitive binding of cells with low CD44 expression and peptide A6 confirms that the in vitro activity of Sal-A6 is related to CD44 expression.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
CPT-Cyclo-GCGPep Conjugate 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
45.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 10 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Gastric tubular adenocarcinoma | ||||
| Efficacy Data | Cell viability |
45.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 10 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Gastric tubular adenocarcinoma | NCI-N87 cell | CVCL_1603 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Gastric tubular adenocarcinoma | ||||
| Efficacy Data | Cell viability |
62.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 2.5 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Gastric tubular adenocarcinoma | NCI-N87 cell | CVCL_1603 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
69.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 2.5 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
78.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.625 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Gastric tubular adenocarcinoma | ||||
| Efficacy Data | Cell viability |
80.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.625 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Gastric tubular adenocarcinoma | NCI-N87 cell | CVCL_1603 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Cell viability |
85.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.156 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Gastric tubular adenocarcinoma | ||||
| Efficacy Data | Cell viability |
100.00%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 0.156 μM | ||||
| Evaluation Method | CCK-8 assay | ||||
| MOA of PDC |
In this research, based on the binding mode between Trastuzumab and HER2 protein, we first discovered a peptide sequence Leadpep that plays a vital role when antibody binds with HER2 protein. In silico mutations were conducted on Leadpep to screen out HER2-targeted peptides. The binding affinity of Cyclo-GCGPep1 increased when cyclized (Kd = 2.555 10-6M). It was used for constructing PDCs with the cytotoxin Camptothecin. Among them, Conjugate 1 showed best antiproliferative activities. Further research demonstrated that Conjugate 1 has a similar mechanism to Camptothecin. It is worth noting that Conjugate 1 showed a good specific delivery capacity and better penetration than Camptothecin. It could be a new therapeutic tool for HER2-positive cancer.
Click to Show/Hide
|
||||
| Description |
Antiproliferative activity of conjugates were evaluated using HER2-positive SK-BR-3 and NCI-N87 cells. As shown in Fig. 5A, Conjugate 1 reduced the cell viability by 23, 38, 47 and 66% at 0.156, 0.625, 2.5 and 10 μM. Its cytotoxicity is significantly more than Conjugate 2 at 0.625 μM and 10 μM. It showed comparable cytotoxicity with CPT at 10 μM. And Conjugate 3 showed little cytotoxicity at any concentrations other than 10 μM, which may be related to the structure of Conjugate 3 where CPT is conjugated on the peptide by non-breakable bond preventing CPT from acting as an anticancer agent. Cyclo-GCGPep1 exhibited no significant cytotoxicity at any concentrations indicating that peptide part of PDCs is inactive for inducing cancer cell death. Similar results can be seen on NCI-N87 cells. Conjugate 1 reduced significantly more cell viability than Conjugate 2 at 0.156 μM and 2.5 μM. And Conjugate 1 exhibited comparable antiproliferation activity as CPT at 2.5 μM and 10 μM. The cytotoxicity of Conjugate 3 is still weak at any concentrations. Cyclo-GCGPep1 is non-toxic to NCI-N87 cells as well. These results told us antiproliferative activity of conjugates is closely related to the ways drugs are conjugated. The cleavable disulfide bonds in Conjugate 1 and 2 could be broken and then Camptothecin derivatives can be released in cells, which is much related to anti-proliferative activity. In general, Conjugate 1 showed best antiproliferative activity and it was selected for further research.
Click to Show/Hide
|
||||
| In Vitro Model | Gastric tubular adenocarcinoma | NCI-N87 cell | CVCL_1603 | ||
References
