Linker Information
General Information of This Linker
| Linker ID |
LIN00160
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
Boc-aminooxy acetic acid
|
|||||
| Linker Type |
Enzyme-sensitive linkers
|
|||||
| Structure |
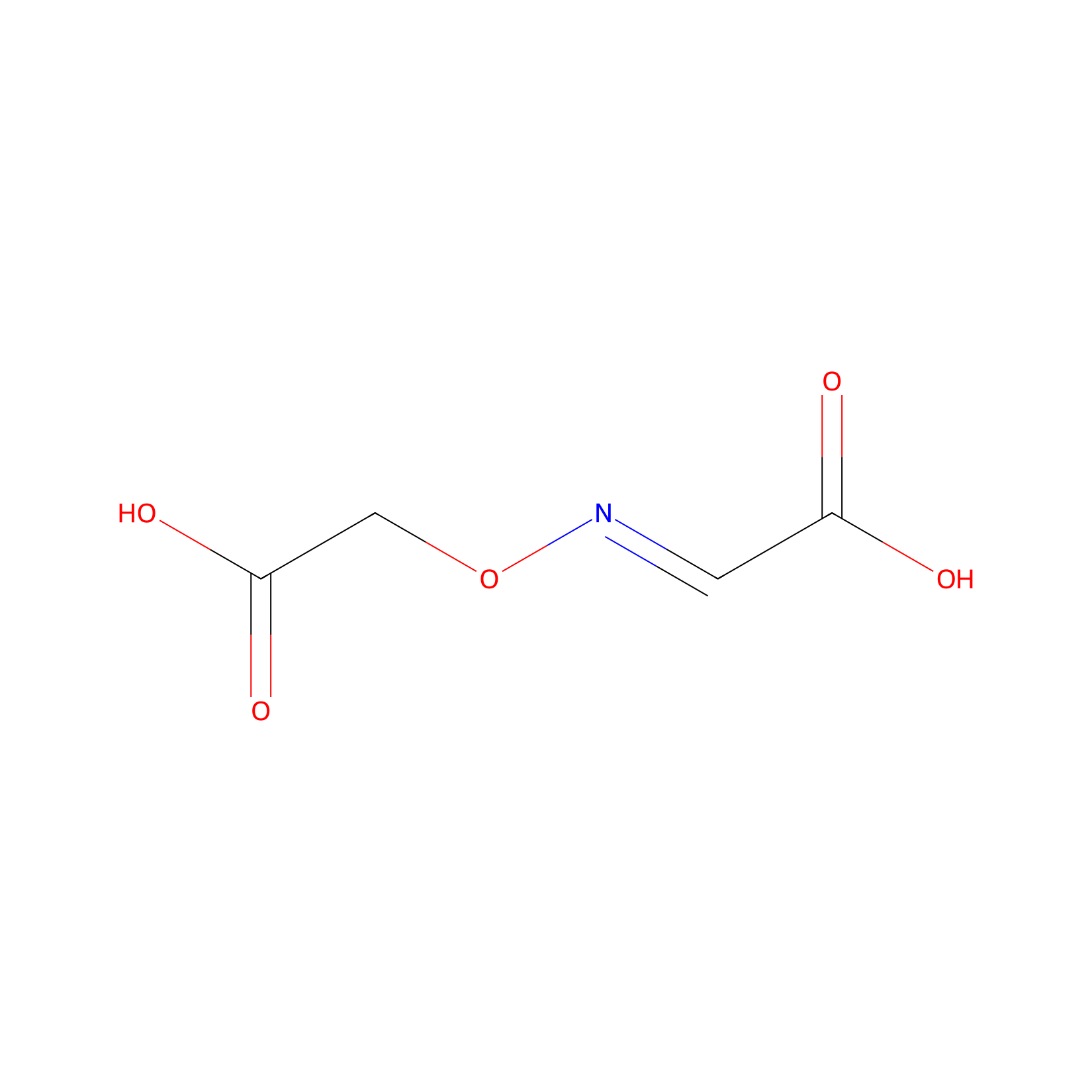
|
|||||
| Formula |
C4H5NO5
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 147.086 | ||||
| Lipid-water partition coefficient (xlogp) | -0.842 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 4 | |||||
| Rotatable Bond Count (rotbonds) | 4 | |||||
| Canonical smiles |
O=C(O)C=NOCC(=O)O
|
|||||
| InChI |
InChI=1S/C4H5NO5/c6-3(7)1-5-10-2-4(8)9/h1H,2H2,(H,6,7)(H,8,9)/b5-1+
|
|||||
| InChIKey |
HLYYFIJFVHWHOM-ORCRQEGFSA-N
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
GOXG2 [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Relative intensity |
5%
|
|||
| Administration Time | 60 min | ||||
| Administration Dosage | 1 μg/ml | ||||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
GOXG2 was the least stable, followed by GOXG1, while GN4OXG was the most stable.
|
||||
| In Vivo Model | Human plasma. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Relative intensity |
0%
|
|||
| Administration Time | 10 h | ||||
| Administration Dosage | 1 μM | ||||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
This experiment showed that the more labile pro-drug was GOXG2, which was almost fully degraded in less than 10 hours while GN4OXG and GOXG1 showed similar, enhanced stability compared to GOXG2.
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
494 ± 93 nM
|
|||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
The most potent among them was found to be pro-drug GOXG2 with IC50 494 ± 93 nM and 675 ± 82 nM against DU145 and PC3, respectively. Regarding the other two conjugates, GN4OXG was found to be the next most cytotoxic compound against DU145 with IC50 590 ± 62 nM, followed by the least toxic GOXG1 with IC50 611 ± 80 nM. In contrast, GOXG1 was the second more toxic against PC3 with IC50 754 ± 142 nM, followed by the least toxic GN4OXG which showed IC50 833 ± 27 nM. The results are summarized in Table 2.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
675 ± 82 nM
|
|||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
The most potent among them was found to be pro-drug GOXG2 with IC50 494 ± 93 nM and 675 ± 82 nM against DU145 and PC3, respectively. Regarding the other two conjugates, GN4OXG was found to be the next most cytotoxic compound against DU145 with IC50 590 ± 62 nM, followed by the least toxic GOXG1 with IC50 611 ± 80 nM. In contrast, GOXG1 was the second more toxic against PC3 with IC50 754 ± 142 nM, followed by the least toxic GN4OXG which showed IC50 833 ± 27 nM. The results are summarized in Table 2.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
GOXG1 [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Relative intensity |
30%
|
|||
| Administration Time | 60 min | ||||
| Administration Dosage | 1 μg/ml | ||||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
GOXG2 was the least stable, followed by GOXG1, while GN4OXG was the most stable.
|
||||
| In Vivo Model | Human plasma. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Relative intensity |
10%
|
|||
| Administration Time | 10 h | ||||
| Administration Dosage | 1 μM | ||||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
This experiment showed that the more labile pro-drug was GOXG2, which was almost fully degraded in less than 10 hours while GN4OXG and GOXG1 showed similar, enhanced stability compared to GOXG2.
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
611 ± 80 nM
|
|||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
The most potent among them was found to be pro-drug GOXG2 with IC50 494 ± 93 nM and 675 ± 82 nM against DU145 and PC3, respectively. Regarding the other two conjugates, GN4OXG was found to be the next most cytotoxic compound against DU145 with IC50 590 ± 62 nM, followed by the least toxic GOXG1 with IC50 611 ± 80 nM. In contrast, GOXG1 was the second more toxic against PC3 with IC50 754 ± 142 nM, followed by the least toxic GN4OXG which showed IC50 833 ± 27 nM. The results are summarized in Table 2.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
754 ± 142 nM
|
|||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
The most potent among them was found to be pro-drug GOXG2 with IC50 494 ± 93 nM and 675 ± 82 nM against DU145 and PC3, respectively. Regarding the other two conjugates, GN4OXG was found to be the next most cytotoxic compound against DU145 with IC50 590 ± 62 nM, followed by the least toxic GOXG1 with IC50 611 ± 80 nM. In contrast, GOXG1 was the second more toxic against PC3 with IC50 754 ± 142 nM, followed by the least toxic GN4OXG which showed IC50 833 ± 27 nM. The results are summarized in Table 2.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
GN4OXG [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Relative intensity |
75%
|
|||
| Administration Time | 60 min | ||||
| Administration Dosage | 1 μg/ml | ||||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
GOXG2 was the least stable, followed by GOXG1, while GN4OXG was the most stable.
|
||||
| In Vivo Model | Human plasma. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Relative intensity |
12%
|
|||
| Administration Time | 10 h | ||||
| Administration Dosage | 1 μM | ||||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
This experiment showed that the more labile pro-drug was GOXG2, which was almost fully degraded in less than 10 hours while GN4OXG and GOXG1 showed similar, enhanced stability compared to GOXG2.
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
590 ± 62 nM
|
|||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
The most potent among them was found to be pro-drug GOXG2 with IC50 494 ± 93 nM and 675 ± 82 nM against DU145 and PC3, respectively. Regarding the other two conjugates, GN4OXG was found to be the next most cytotoxic compound against DU145 with IC50 590 ± 62 nM, followed by the least toxic GOXG1 with IC50 611 ± 80 nM. In contrast, GOXG1 was the second more toxic against PC3 with IC50 754 ± 142 nM, followed by the least toxic GN4OXG which showed IC50 833 ± 27 nM. The results are summarized in Table 2.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
833 ± 27 nM
|
|||
| MOA of PDC |
We aimed to construct PDCs with linker controllable drug release rates simply by manipulating the linker unit. For a more rapid drug-release rate we developed GOXG1 and GOXG2. These conjugates bear a carboxylate ester linker directly attached to the primary and the secondary alcohol group of the drug respectively, followed by oxime and amide bond. The primary alcohol of gemcitabine has been used since it is involved in the phosphorylation process, through which gemcitabine exerts its cytotoxic effect. Therefore, we aimed to block the primary alcohol and examine its effect (GOXG1) and also take advantage of the secondary alcohol which could lead to a PDC with a completely different profile (GOXG2), although they share structural similarities. For a slower drug release rate, we designed and developed the PDC GN4OXG that contains an amide bond on the 4-N position of the parent drug followed by click oxime ligation and another amide bond. The stability of this molecule should be enhanced since it is devoid of rapidly hydrolyzable ester bonds. Furthermore, in this PDC since the 4-NH2 moiety of gemcitabine is capped it could further surmount the rapid gemcitabine metabolism that leads to the formation of dFdU, after the enzymatic 4-N deamination of gemcitabine by cytidine deaminase (CDA).
Click to Show/Hide
|
||||
| Description |
The most potent among them was found to be pro-drug GOXG2 with IC50 494 ± 93 nM and 675 ± 82 nM against DU145 and PC3, respectively. Regarding the other two conjugates, GN4OXG was found to be the next most cytotoxic compound against DU145 with IC50 590 ± 62 nM, followed by the least toxic GOXG1 with IC50 611 ± 80 nM. In contrast, GOXG1 was the second more toxic against PC3 with IC50 754 ± 142 nM, followed by the least toxic GN4OXG which showed IC50 833 ± 27 nM. The results are summarized in Table 2.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
References
