Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_01571
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Tetrapeptide 42 - AZT monophosphate conjugate
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
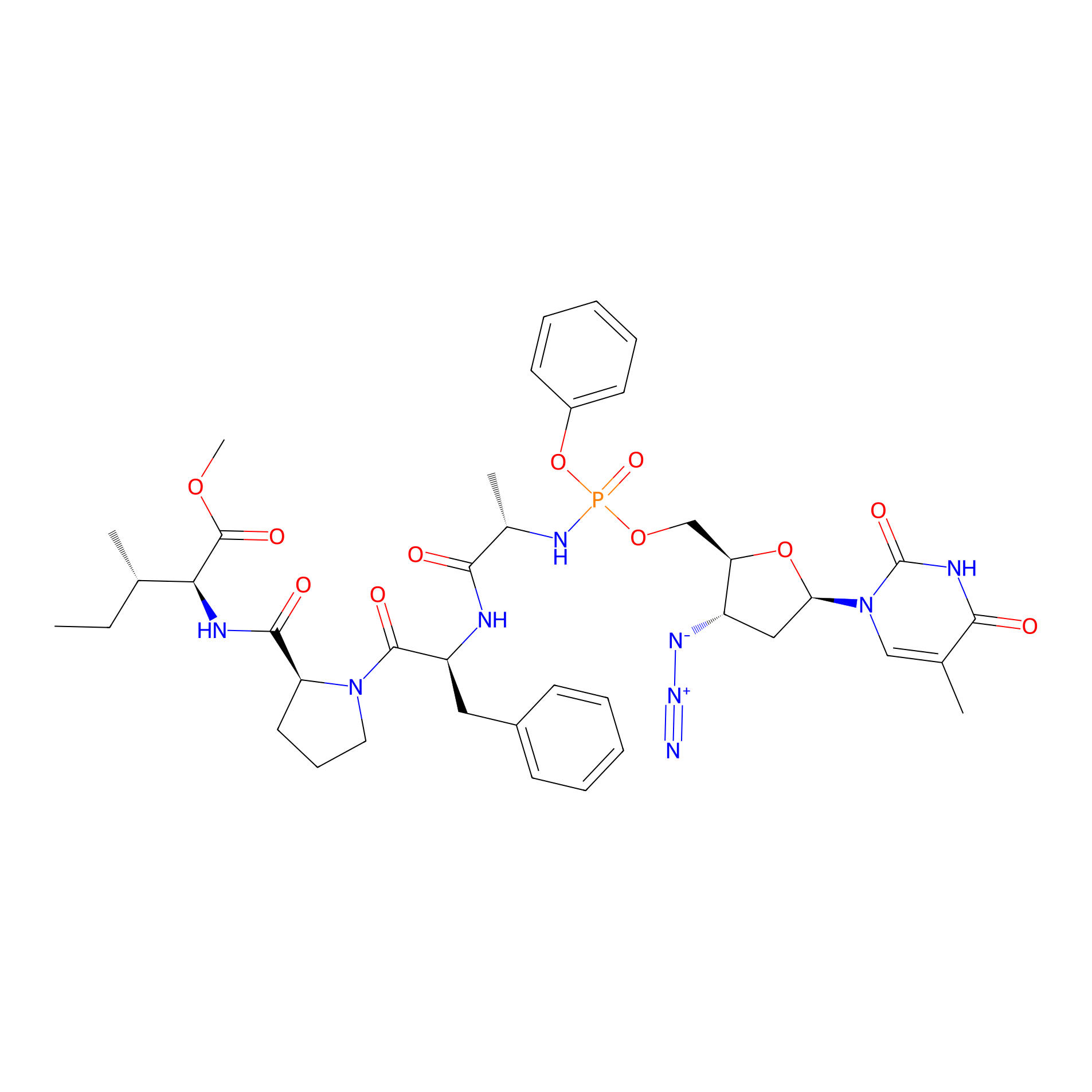
|
|||||
| Peptide Name |
Tetrapeptide 42
|
Peptide Info | ||||
| Receptor Name |
HIV-1 protease
|
Receptor Info | ||||
| Drug Name |
Zidovudine monophosphate
|
Drug Info | ||||
| Linker Name |
Phosphoramidate bond
|
Linker Info | ||||
| Formula |
C40H52N9O11P
|
|||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight | 865.882 | ||||
| Lipid-water partition coefficient (xlogp) | 3.2471 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 4 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 13 | |||||
| Rotatable Bond Count (rotbonds) | 19 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
1.1 μM
|
|||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
2.4 μM
|
|||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
70 μM
|
|||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM cell | CVCL_0207 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
77 μM
|
|||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
105 mM
|
|||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
Concerning the phosphoramidate series, low anti-HIV activities ranging from 70 nM to 1.9 μM and from 0.5 to 6.1 μM were measured when TK+ CEM-SS and MT-4 cells were incubated with these derivatives, respectively. Although these prodrugs degraded very rapidly, these values are much higher than that of AZT, indicating that (i) AZT is not the major metabolite that has been released upon hydrolysis in the extra- and intra-cellular medium, (ii) AZT-MP has not been released at an efficient inhibitory level into the intracellular medium, and/or (iii) the other metabolites are poor anti-HIV active agents. It is noteworthy that the highest antiviral activity was achieved by the prodrug that released the largest amount of AZT, that is, 21. That its antiviral activity is related to AZT and not to AZT-MP is further supported by the absence of inhibition measured on TK- cells. However, among the eight phosphoramidate conjugates 15-22, six were found to have a weak inhibitory activity on HIV replication in TK- CEM cells with IC50 values of about 40-100 μM (for example, 15-18, 20 and 22). This could indicate that a low amount of free AZT-MP has been released into the cells. However, two of these conjugates (15 and 16) were found to be moderately toxic in CEM-SS and MT-4 cells (CC50 of 40 and 18 μM in CEM-SS and 19 and 2 μM in MT-4 cells, respectively). On TK- CEM cells, the IC50 and the CC50 of compound 15 are very close, indicating a non-specific antiviral activity.
Click to Show/Hide
|
||||
| In Vitro Model | HIV Infection | Human immunodeficiency virus | 12721 | ||
References
