Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00010
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Kb-C04
|
|||||
| Structure |
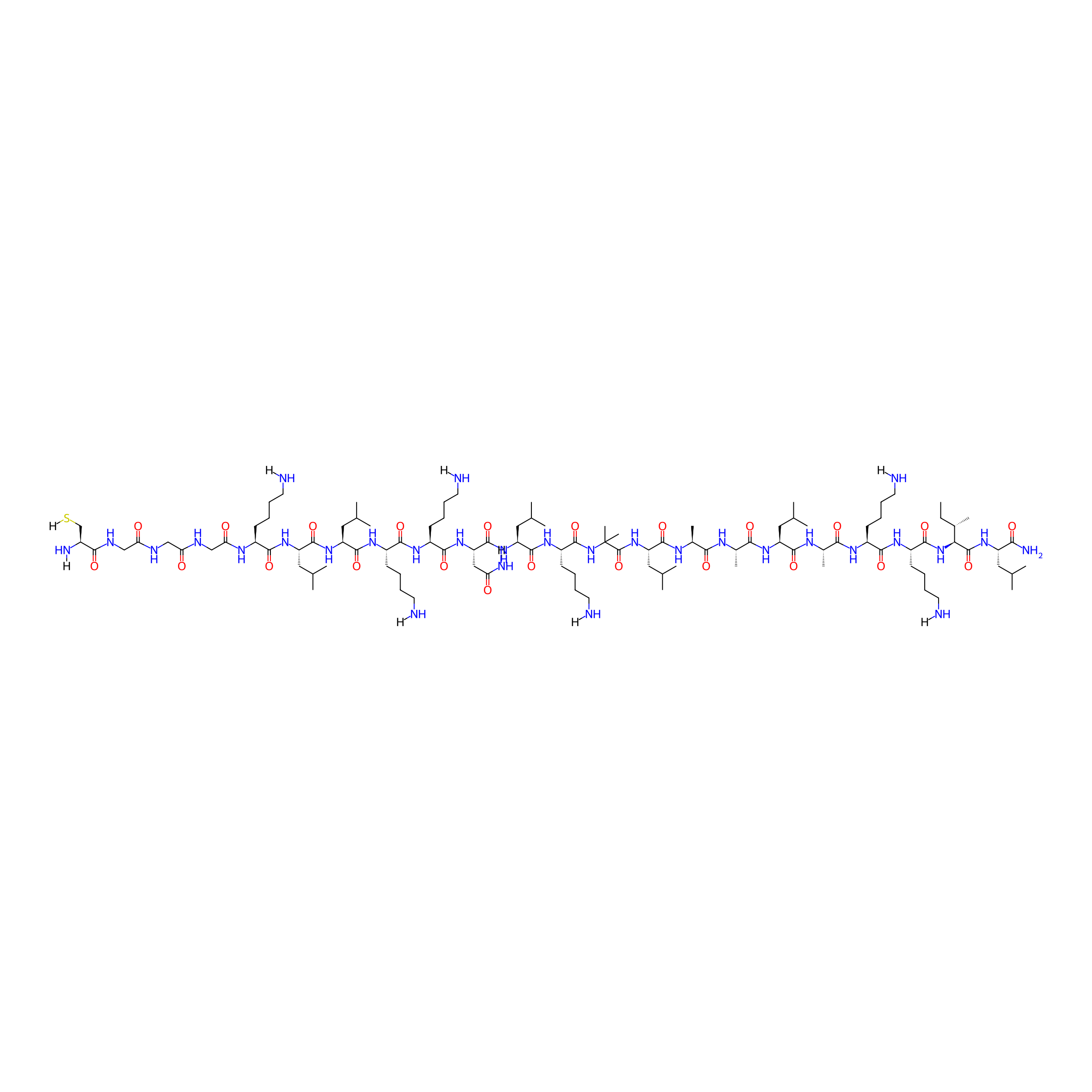
|
|||||
| Sequence |
CGGGKLLKKNLK-Aib-LAALAKKIL-NH2
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Cell membrane
|
|||||
| PDC Transmembrane Types | Cell-penetrating peptides (CPPs) | |||||
| Formula |
C104H194N30O23S
|
|||||
| Isosmiles |
[H]NCCCC[C@H](NC(=O)CNC(=O)CNC(=O)CNC(=O)[C@H](CS[H])N[H])C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN[H])C(=O)N[C@@H](CCCCN[H])C(=O)N[C@@H](CC(=O)N[H])C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN[H])C(=O)NC(C)(C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN[H])C(=O)N[C@@H](CCCCN[H])C(=O)N[C@]([H])(C(=O)N[C@@H](CC(C)C)C(N)=O)[C@@H](C)CC
|
|||||
| InChI |
InChI=1S/C104H194N30O23S/c1-20-62(14)84(102(156)126-73(85(113)139)45-56(2)3)133-94(148)71(37-25-31-43-109)123-91(145)68(34-22-28-40-106)121-87(141)65(17)119-95(149)74(46-57(4)5)127-88(142)64(16)117-86(140)63(15)118-96(150)78(50-61(12)13)132-103(157)104(18,19)134-101(155)72(38-26-32-44-110)125-98(152)76(48-59(8)9)130-100(154)79(51-80(112)135)131-93(147)70(36-24-30-42-108)122-92(146)69(35-23-29-41-107)124-97(151)75(47-58(6)7)129-99(153)77(49-60(10)11)128-90(144)67(33-21-27-39-105)120-83(138)54-115-81(136)52-114-82(137)53-116-89(143)66(111)55-158/h56-79,84,158H,20-55,105-111H2,1-19H3,(H2,112,135)(H2,113,139)(H,114,137)(H,115,136)(H,116,143)(H,117,140)(H,118,150)(H,119,149)(H,120,138)(H,121,141)(H,122,146)(H,123,145)(H,124,151)(H,125,152)(H,126,156)(H,127,142)(H,128,144)(H,129,153)(H,130,154)(H,131,147)(H,132,157)(H,133,148)(H,134,155)/t62-,63-,64-,65-,66-,67-,68-,69-,70-,71-,72-,73-,74-,75-,76-,77-,78-,79-,84-/m0/s1
|
|||||
| InChIKey |
VOSXVFJGUSISRV-JUZUOIPZSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
2264.95
|
Polar area
|
879.42
|
||
|
Complexity
|
2263.465383
|
xlogp Value
|
-5.7544
|
|||
|
Heavy Count
|
158
|
Rot Bonds
|
92
|
|||
|
Hbond acc
|
31
|
Hbond Donor
|
31
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
Kb-CC04 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.14 ± 0.02 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.32 ± 0.02 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT-116/CPT cell | CVCL_0291 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Gastric cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.38 ± 0.01 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Gastric carcinoma | SGC-7901 cell | CVCL_0520 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.43 ± 0.02 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Gastric cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.00 ± 0.08 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Human papillomavirus-related cervical adenocarcinoma | SGC-7901/CPT cell | CVCL_0520 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.05 ± 0.03 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma of no special type | MCF7/C4 cell | CVCL_GX99 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Colon cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
13.4 ± 0.4 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | NCM460 cell | CVCL_0460 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Gastric cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
15.5 ± 0.6 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | GES1 cell | CVCL_EQ22 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Liver cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
16.4 ± 0.2 µM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Cationic anticancer peptides (CAPs) refer to peptide-based molecules possessing a net positive charge, allowing them to preferentially combined with cancer cells which exhibit an overabundance of anionic components on their surface. Cationic peptides additionally induce cell membrane destabilization through carpet-like, toroidal pore or sinking raft et al. mechanism, or penetrate cell membrane and act on cellular sub-structures. Besides, cell-penetrating peptides (CPPs) represent a special subset of CAPs, renowned for their rapid and low-toxic cellular translocation abilities, making them promising vehicles for drug delivery. Therefore, CAPs are emerging as a class of potential anticancer agents and gaining widespread attention by researchers. As reported, the construction of bioactive peptides with cytotoxic payloads via a chemical linker provides a kind of prodrugs known as peptide-drug conjugates (PDCs), which represents an efficient approach to address the limitations of small molecules by leveraging the properties of peptides. In previous study, we have discovered the CAP of KM8-Aib (sequence: Lys-Leu-Leu-Lys-Lys-Asn-Leu-Lys-Aib-Leu-Ala-Ala-Leu-Ala-Lys-Lys-Ile-Leu-NH2), which derived from venom peptide Mastoparan, exhibiting favorable antiproliferative effect against several cancer cells but less toxic to benign cells. Herein, we attempted to integrate CPT with KM8-Aib, obtaining a series of peptide-CPT conjugates, to improve the solubility, selectivity of CPT, and achieve enhanced therapeutic outcomes. Based on preliminary in vitro assays, the PDC Kb-CC07 demonstrated exceptional selective cytotoxicity against various cancer cell lines, including drug-resistant variants, surpassing that of other conjugates. The solubility of Kb-CC07 was also significantly increased, which is ˜ 100 fold higher than that of CPT. In addition, we evaluated the in vivo antitumor efficacy of Kb-CC07 via nude-xenograft model. It is showed that mice treated with Kb-CC07 had substantially lower tumor volumes and minimum organ toxicities than CPT-treated group. As such, Kb-CC07 is suggested to be a promising anticancer agent and warrant thorough exploration.
Click to Show/Hide
|
||||
| Description |
MTT assay was preformed to measure the in vitro cytotoxicities of the compounds against cancer cells (HCT-116, SGC-7901, MCF-7, HCT-116/CPT, SGC-7901/CPT and MCF-7/C4) and benign cells (NCM460, GES-1 and LO2). As present in Table 1, all the peptides (Kb-C01, 0207) exhibited certain selective anticancer activities, which are comparable to their parent counterpart KM8-Aib. The target PDCs (Kb-CC01, 0207) were further obtained by coupling the peptides with CPT, displaying markedly improved antiproliferative activities and selectivities against cancer cell lines over CPT. Studies have revealed that cancer cell membranes are negatively charged due to the presence of substantial amounts of acid phospholipids. Therefore, positively charged CAPs are capable of binding to cancer cell surface through electrostatic interactions, making them specific to cancer cells. In this project, since the PDCs were composed of CAP KM8-Aib and CPT, they potentially inherited the antineoplastic activities of KM8-Aib against cancer cells, as well as low toxicities to non-malignant cells.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | LO #2 cell | CVCL_C7SD | ||
References
