Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00075
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Cyclic NGR 5
|
|||||
| Structure |
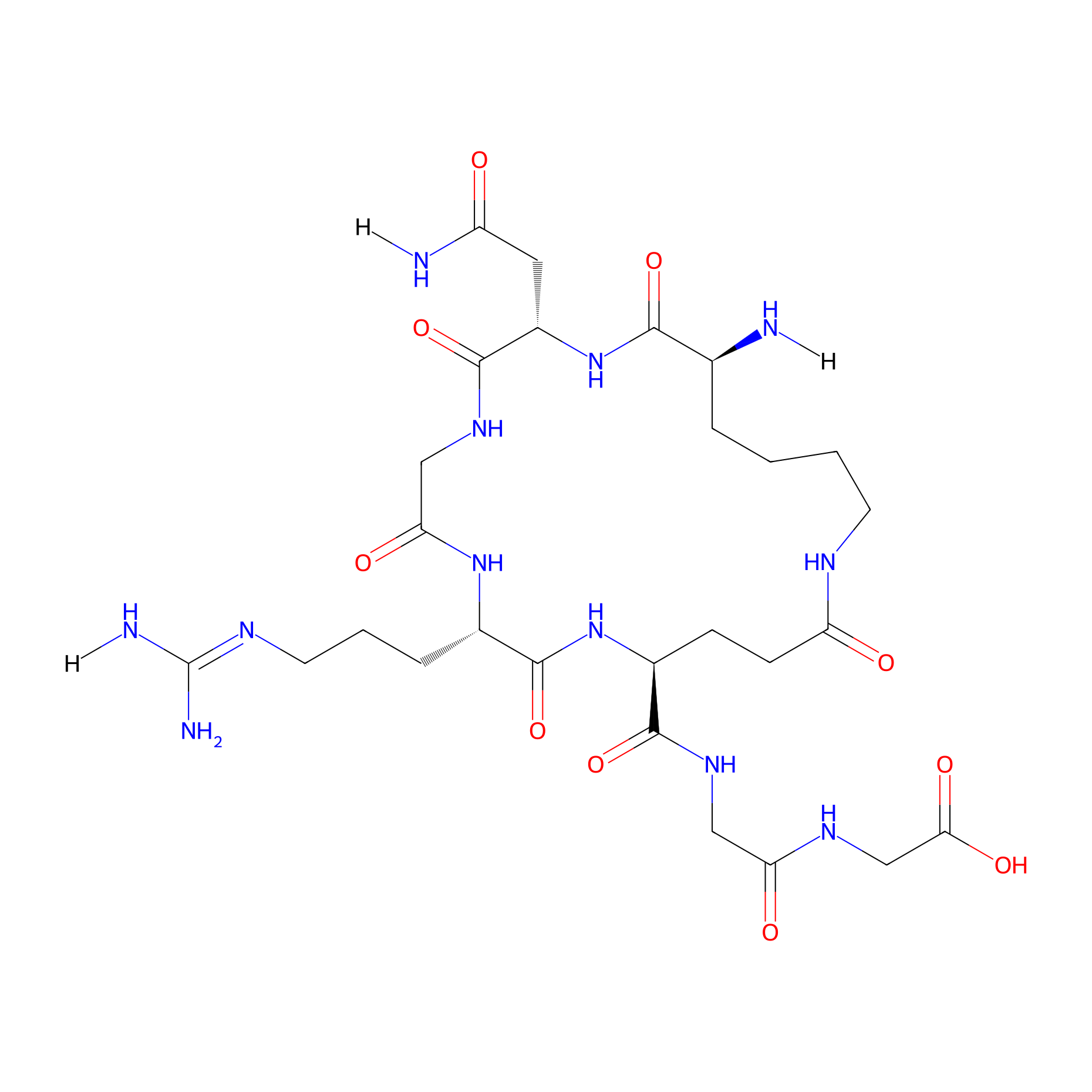
|
|||||
| Sequence |
KNGREGG
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Aminopeptidase N (ANPEP)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C27H46N12O10
|
|||||
| Isosmiles |
[H]NC(=O)C[C@@H]1NC(=O)[C@@H](N[H])CCCCNC(=O)CC[C@@H](C(=O)NCC(=O)NCC(=O)O)NC(=O)[C@H](CCC/N=C(\N)N[H])NC(=O)CNC1=O
|
|||||
| InChI |
InChI=1S/C27H46N12O10/c28-14-4-1-2-8-32-19(41)7-6-16(24(47)35-11-20(42)34-13-22(44)45)38-26(49)15(5-3-9-33-27(30)31)37-21(43)12-36-25(48)17(10-18(29)40)39-23(14)46/h14-17H,1-13,28H2,(H2,29,40)(H,32,41)(H,34,42)(H,35,47)(H,36,48)(H,37,43)(H,38,49)(H,39,46)(H,44,45)(H4,30,31,33)/t14-,15-,16-,17-/m0/s1
|
|||||
| InChIKey |
ZDNYFGMYNXXVSB-QAETUUGQSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
698.739
|
Polar area
|
374.51
|
||
|
Complexity
|
698.3459857
|
xlogp Value
|
-6.7894
|
|||
|
Heavy Count
|
49
|
Rot Bonds
|
12
|
|||
|
Hbond acc
|
11
|
Hbond Donor
|
12
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
Cyclic NGR peptidedaunomycin conjugates 5 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.4 ± 0.1 µM
|
|||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
3.0 ± 0.6 µM
|
|||
| Administration Time | 72 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
5.7 ± 0.5 µM
|
|||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
8.7 ± 1.2 µM
|
|||
| Administration Time | 6 h | ||||
| Description |
In contrast, conjugate 2 (containing a disulfide bridge) showed preferential toxicity to HT-1080 cells in the cytostatic experiments, resulting in the highest selectivity. The antitumor effect of 2 increased in time (72 h treatment), but its selectivity decreased significantly. In comparison to conjugates 3 (thioether linkage) and 5 (amide bond), both containing free Lys in the cycle, conjugate 5 had slightly better activity against HT-1080 cells. However, the selectivity of conjugate 5 to CD13 receptors (as judged by the relative toxicity against the two cell lines) was not significant after 6 h treatment, whereas it was significantly more toxic to HT-1080 cells in the 72 h experiment. This can also be explained by its higher chemostability that results in longer exposure the cells to the unmodified NGR peptide-drug conjugate. In contrast, their analogs in which the drug molecule is connected to the side chain of Lys in the cycle (conjugates 4 and 6) were more specific but less toxic, especially in the cytotoxicity experiments. Interestingly, there was no correlation between the specificity and the chemostability of the conjugates.
Click to Show/Hide
|
||||
| In Vitro Model | Colon adenocarcinoma | CD13-negative HT29 cell | CVCL_0320 | ||
References
