Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00078
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
P-(A5G27scrm)-FITC
|
|||||
| Structure |
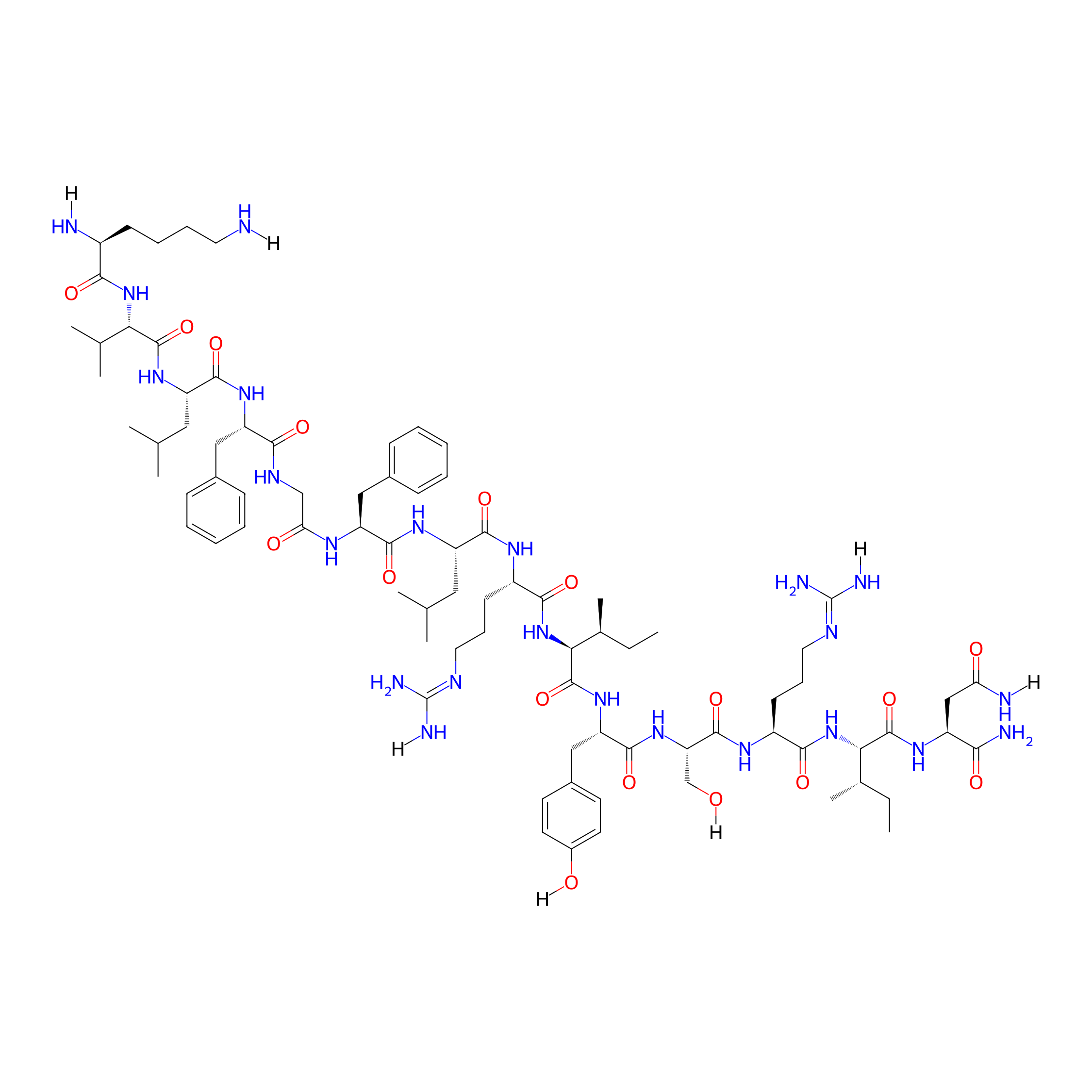
|
|||||
| Sequence |
KVLFGFLRIYSRIN-NH2
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
CD44 antigen (CD44)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C83H133N23O17
|
|||||
| Isosmiles |
[H]NCCCC[C@H](N[H])C(=O)N[C@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC/N=C(\N)N[H])C(=O)N[C@]([H])(C(=O)N[C@@H](Cc1ccc(O[H])cc1)C(=O)N[C@@H](CO[H])C(=O)N[C@@H](CCC/N=C(\N)N[H])C(=O)N[C@]([H])(C(=O)N[C@@H](CC(=O)N[H])C(N)=O)[C@@H](C)CC)[C@@H](C)CC)C(C)C
|
|||||
| InChI |
InChI=1S/C83H133N23O17/c1-11-48(9)67(80(122)98-57(69(87)111)42-64(86)109)105-73(115)56(29-22-36-93-83(90)91)97-78(120)63(44-107)103-77(119)62(41-52-30-32-53(108)33-31-52)102-81(123)68(49(10)12-2)106-72(114)55(28-21-35-92-82(88)89)96-74(116)58(37-45(3)4)99-76(118)61(40-51-25-17-14-18-26-51)95-65(110)43-94-71(113)60(39-50-23-15-13-16-24-50)100-75(117)59(38-46(5)6)101-79(121)66(47(7)8)104-70(112)54(85)27-19-20-34-84/h13-18,23-26,30-33,45-49,54-63,66-68,107-108H,11-12,19-22,27-29,34-44,84-85H2,1-10H3,(H2,86,109)(H2,87,111)(H,94,113)(H,95,110)(H,96,116)(H,97,120)(H,98,122)(H,99,118)(H,100,117)(H,101,121)(H,102,123)(H,103,119)(H,104,112)(H,105,115)(H,106,114)(H4,88,89,92)(H4,90,91,93)/t48-,49-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,66-,67-,68-/m0/s1
|
|||||
| InChIKey |
FRUJCYBMVDKHFQ-NWDMPNHUSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1725.121
|
Polar area
|
685.78
|
||
|
Complexity
|
1724.02498
|
xlogp Value
|
-3.8892
|
|||
|
Heavy Count
|
123
|
Rot Bonds
|
61
|
|||
|
Hbond acc
|
21
|
Hbond Donor
|
23
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| Percentage of labeled cells | 62% | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
4T1-Luc and B16-F10 cells were collected after trypsinization of cell monolayers. Cells (5 105) were incubated in 3% BSA for 30 min at room temperature and then treated with 50 μg/mL of the FITC-labeled P-(A5G27) or P-(A5G27scrm) in 0.5 mL of PBS containing 1% BSA for 1 h at 4 °C or for 4 h at 37 °C. Cells were then washed twice with PBS, and the fluorescence intensity was analyzed by Guava easyCyte flow cytometer (EMD Millipore, excitation at 488 nm and emission at 525 nm).
Click to Show/Hide
|
|||||
| Experimental Condition | 4T1-Luc and B16-F10 cells | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
P-(A5G27scrm)-PTX [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic Tumor | ||||
| Efficacy Data | Median overall survival (mOS) |
40.5 days
|
|||
| Administration Dosage | 15 mg/kg PTX equivalent dose | ||||
| Description |
The median survival of mice in the P-(A5G27)-PTX treatment group was longer than in P-(A5G27scrm)-PTX, P-(A5G27), and free PTX-treated groups (48.50 vs 40.5, 43, and 45.5, respectively); however, differences were nonsignificant (Figure 6B).
|
||||
| In Vivo Model | 4T1 tumor-bearing mice. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic Tumor | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
0.221 ± 0.055 µM
|
|||
| Administration Time | 72 h | ||||
| Description |
P-(A5G27)-PTX was ˜4-fold more toxic than the nontargeted P-(A5G27scrm)-PTX copolymer, suggesting a faster (receptor-mediated) internalization (Figure 4).
|
||||
| In Vitro Model | Mammary carcinoma | 4T1 cell | CVCL_0125 | ||
References
