Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00146
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
P2
|
|||||
| Structure |
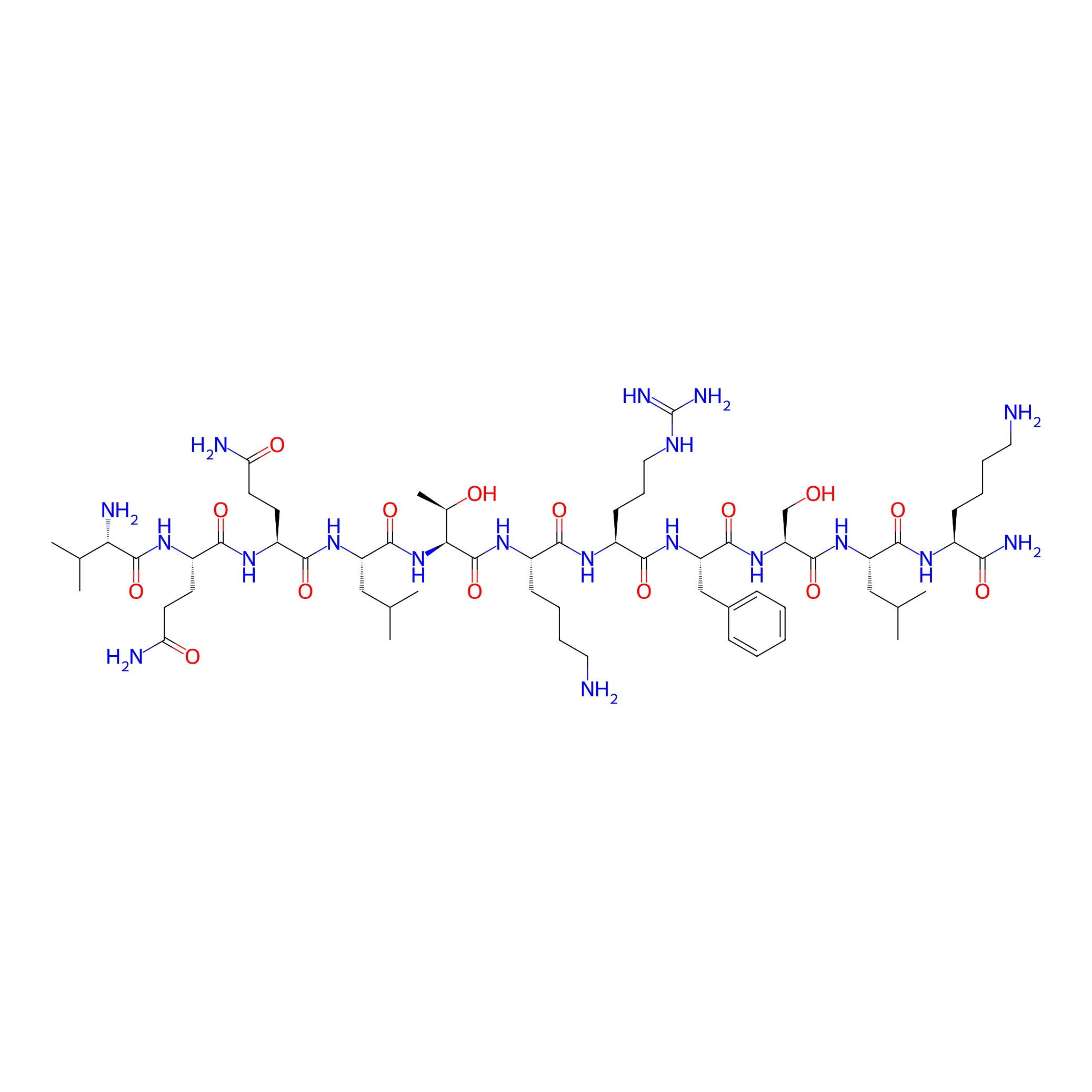
|
|||||
| Sequence |
VQQLTKRFSLK
|
|||||
| Peptide Type |
Linear
|
|||||
| PDC Transmembrane Types | Cell-penetrating peptides (CPPs) | |||||
| Formula |
C61H107N19O15
|
|||||
| Isosmiles |
[H][C@@](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)C(C)C)(C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(N)=O)[C@@H](C)O
|
|||||
| InChI |
InChI=1S/C61H107N19O15/c1-32(2)28-42(55(90)71-37(50(67)85)18-11-13-25-62)77-58(93)45(31-81)79-56(91)44(30-36-16-9-8-10-17-36)78-52(87)39(20-15-27-70-61(68)69)72-51(86)38(19-12-14-26-63)75-60(95)49(35(7)82)80-57(92)43(29-33(3)4)76-54(89)40(21-23-46(64)83)73-53(88)41(22-24-47(65)84)74-59(94)48(66)34(5)6/h8-10,16-17,32-35,37-45,48-49,81-82H,11-15,18-31,62-63,66H2,1-7H3,(H2,64,83)(H2,65,84)(H2,67,85)(H,71,90)(H,72,86)(H,73,88)(H,74,94)(H,75,95)(H,76,89)(H,77,93)(H,78,87)(H,79,91)(H,80,92)(H4,68,69,70)/t35-,37+,38+,39+,40+,41+,42+,43+,44+,45+,48+,49+/m1/s1
|
|||||
| InChIKey |
WAFRYYJMNJZCGQ-XFLLRNPZSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1346.645
|
Polar area
|
600.69
|
||
|
Complexity
|
1345.819404
|
xlogp Value
|
-5.93673
|
|||
|
Heavy Count
|
95
|
Rot Bonds
|
48
|
|||
|
Hbond acc
|
19
|
Hbond Donor
|
21
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
MP-P2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Zika virus infection | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 50 µM | |||
| Administration Time | 1 h | ||||
| Evaluation Method | GraphPad Prism assay | ||||
| MOA of PDC |
Strategies for conjugating BBBpS to drug payloads have been actively explored over recent decades, with the number of new shuttles steadily increasing. Some recent entries, moreover, have shown to be capable not only of carrying drugs into but also removing toxins from the brain, preventing their accumulation. For our part, we have reported that peptide-porphyrin conjugates (PPCs), where a BBBpS and an antiviral porphyrin are covalently linked by an amide bond (amino and carboxyl groups in BBBpS and porphyrin, respectively) can successfully pass the BBB and act against brain-targeting viruses such as HIV. As for the BPB, the literature is scarce, with only a few described examples of peptides able to pass it. Herein, we report our results in developing new PPCs able to penetrate both BPB and BBB and act against ZIKV. The PPC production strategy, involving porphyrin (P), C- or N-terminal conjugation to a BBBpS, has been detailed in a recent publication and is illustrated in Scheme 1 and Scheme S1. In this paper we describe eight PPCs resulting from the combination of four BBBpS and two porphyrins, and their evaluation in terms of barrier crossing and anti-ZIKV activity. One of the conjugates, PP-P1, emerges as particularly effective against ZIKV, having also the ability to translocate across BPB and BBB.
Click to Show/Hide
|
||||
| Description |
The five PPCs that successfully translocated BBB and BPB were evaluated for ZIKV inactivation in vitro, using a plaque assay. Of them, two showed significant activity, namely MP-P5 (IC50 = 25.07 ± 0.05 μM, similar to activity against HIV) and PP-P1 (IC50 = 1.08 ± 0.14 μM). Additionally, a treatment assay performed with MP-P5 and PP-P1 revealed that both PPCs efficiently inhibit ZIKV replication when added 1 h and 7 h post-infection. As observed for HIV, non-conjugated porphyrins did not show activity against ZIKV, reinforcing the claim that BBBpS conjugation is not only critical for BBB/BPB translocation but also for antiviral activity. On the other hand, and somewhat unexpectedly, PP-P1, shown to be inactive in vitro against HIV, emerged as the most active anti-ZIKV conjugate. As described in the literature, the light-independent mechanism of action of porphyrins is based on a direct perturbation of the viral envelope. Porphyrins interact and accumulate on the envelope lipid membrane, causing a decrease in order and a consequent phase alteration that impairs viral entry processes. Since we ensured no-light conditions and no metal cations are coordinated to the porphyrin rings of the PPCsavoiding generation of reactive oxygen speciesthe antiviral light-independent mechanism is the only plausible one. Thus, one may confidently suggest that the PP-P1 specific anti-ZIKV activity is guided by preferential interaction with the ZIKV viral envelope and/or components.
Click to Show/Hide
|
||||
| In Vitro Model | ZIKV infection | Zika virus | 64320 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 50 µM | |||
| Administration Time | 1 h | ||||
| Evaluation Method | GraphPad Prism assay | ||||
| MOA of PDC |
Strategies for conjugating BBBpS to drug payloads have been actively explored over recent decades, with the number of new shuttles steadily increasing. Some recent entries, moreover, have shown to be capable not only of carrying drugs into but also removing toxins from the brain, preventing their accumulation. For our part, we have reported that peptide-porphyrin conjugates (PPCs), where a BBBpS and an antiviral porphyrin are covalently linked by an amide bond (amino and carboxyl groups in BBBpS and porphyrin, respectively) can successfully pass the BBB and act against brain-targeting viruses such as HIV. As for the BPB, the literature is scarce, with only a few described examples of peptides able to pass it. Herein, we report our results in developing new PPCs able to penetrate both BPB and BBB and act against ZIKV. The PPC production strategy, involving porphyrin (P), C- or N-terminal conjugation to a BBBpS, has been detailed in a recent publication and is illustrated in Scheme 1 and Scheme S1. In this paper we describe eight PPCs resulting from the combination of four BBBpS and two porphyrins, and their evaluation in terms of barrier crossing and anti-ZIKV activity. One of the conjugates, PP-P1, emerges as particularly effective against ZIKV, having also the ability to translocate across BPB and BBB.
Click to Show/Hide
|
||||
| Description |
The five PPCs that successfully translocated BBB and BPB were evaluated for ZIKV inactivation in vitro, using a plaque assay. Of them, two showed significant activity, namely MP-P5 (IC50 = 25.07 ± 0.05 μM, similar to activity against HIV) and PP-P1 (IC50 = 1.08 ± 0.14 μM). Additionally, a treatment assay performed with MP-P5 and PP-P1 revealed that both PPCs efficiently inhibit ZIKV replication when added 1 h and 7 h post-infection. As observed for HIV, non-conjugated porphyrins did not show activity against ZIKV, reinforcing the claim that BBBpS conjugation is not only critical for BBB/BPB translocation but also for antiviral activity. On the other hand, and somewhat unexpectedly, PP-P1, shown to be inactive in vitro against HIV, emerged as the most active anti-ZIKV conjugate. As described in the literature, the light-independent mechanism of action of porphyrins is based on a direct perturbation of the viral envelope. Porphyrins interact and accumulate on the envelope lipid membrane, causing a decrease in order and a consequent phase alteration that impairs viral entry processes. Since we ensured no-light conditions and no metal cations are coordinated to the porphyrin rings of the PPCsavoiding generation of reactive oxygen speciesthe antiviral light-independent mechanism is the only plausible one. Thus, one may confidently suggest that the PP-P1 specific anti-ZIKV activity is guided by preferential interaction with the ZIKV viral envelope and/or components.
Click to Show/Hide
|
||||
| In Vitro Model | HIV Infection | Human immunodeficiency virus | 12721 | ||
PP-P2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Zika virus infection | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 50 µM | |||
| Administration Time | 1 h | ||||
| Evaluation Method | GraphPad Prism assay | ||||
| MOA of PDC |
Strategies for conjugating BBBpS to drug payloads have been actively explored over recent decades, with the number of new shuttles steadily increasing. Some recent entries, moreover, have shown to be capable not only of carrying drugs into but also removing toxins from the brain, preventing their accumulation. For our part, we have reported that peptide-porphyrin conjugates (PPCs), where a BBBpS and an antiviral porphyrin are covalently linked by an amide bond (amino and carboxyl groups in BBBpS and porphyrin, respectively) can successfully pass the BBB and act against brain-targeting viruses such as HIV. As for the BPB, the literature is scarce, with only a few described examples of peptides able to pass it. Herein, we report our results in developing new PPCs able to penetrate both BPB and BBB and act against ZIKV. The PPC production strategy, involving porphyrin (P), C- or N-terminal conjugation to a BBBpS, has been detailed in a recent publication and is illustrated in Scheme 1 and Scheme S1. In this paper we describe eight PPCs resulting from the combination of four BBBpS and two porphyrins, and their evaluation in terms of barrier crossing and anti-ZIKV activity. One of the conjugates, PP-P1, emerges as particularly effective against ZIKV, having also the ability to translocate across BPB and BBB.
Click to Show/Hide
|
||||
| Description |
The five PPCs that successfully translocated BBB and BPB were evaluated for ZIKV inactivation in vitro, using a plaque assay. Of them, two showed significant activity, namely MP-P5 (IC50 = 25.07 ± 0.05 μM, similar to activity against HIV) and PP-P1 (IC50 = 1.08 ± 0.14 μM). Additionally, a treatment assay performed with MP-P5 and PP-P1 revealed that both PPCs efficiently inhibit ZIKV replication when added 1 h and 7 h post-infection. As observed for HIV, non-conjugated porphyrins did not show activity against ZIKV, reinforcing the claim that BBBpS conjugation is not only critical for BBB/BPB translocation but also for antiviral activity. On the other hand, and somewhat unexpectedly, PP-P1, shown to be inactive in vitro against HIV, emerged as the most active anti-ZIKV conjugate. As described in the literature, the light-independent mechanism of action of porphyrins is based on a direct perturbation of the viral envelope. Porphyrins interact and accumulate on the envelope lipid membrane, causing a decrease in order and a consequent phase alteration that impairs viral entry processes. Since we ensured no-light conditions and no metal cations are coordinated to the porphyrin rings of the PPCsavoiding generation of reactive oxygen speciesthe antiviral light-independent mechanism is the only plausible one. Thus, one may confidently suggest that the PP-P1 specific anti-ZIKV activity is guided by preferential interaction with the ZIKV viral envelope and/or components.
Click to Show/Hide
|
||||
| In Vitro Model | ZIKV infection | Zika virus | 64320 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 50 µM | |||
| Administration Time | 1 h | ||||
| Evaluation Method | GraphPad Prism assay | ||||
| MOA of PDC |
Strategies for conjugating BBBpS to drug payloads have been actively explored over recent decades, with the number of new shuttles steadily increasing. Some recent entries, moreover, have shown to be capable not only of carrying drugs into but also removing toxins from the brain, preventing their accumulation. For our part, we have reported that peptide-porphyrin conjugates (PPCs), where a BBBpS and an antiviral porphyrin are covalently linked by an amide bond (amino and carboxyl groups in BBBpS and porphyrin, respectively) can successfully pass the BBB and act against brain-targeting viruses such as HIV. As for the BPB, the literature is scarce, with only a few described examples of peptides able to pass it. Herein, we report our results in developing new PPCs able to penetrate both BPB and BBB and act against ZIKV. The PPC production strategy, involving porphyrin (P), C- or N-terminal conjugation to a BBBpS, has been detailed in a recent publication and is illustrated in Scheme 1 and Scheme S1. In this paper we describe eight PPCs resulting from the combination of four BBBpS and two porphyrins, and their evaluation in terms of barrier crossing and anti-ZIKV activity. One of the conjugates, PP-P1, emerges as particularly effective against ZIKV, having also the ability to translocate across BPB and BBB.
Click to Show/Hide
|
||||
| Description |
The five PPCs that successfully translocated BBB and BPB were evaluated for ZIKV inactivation in vitro, using a plaque assay. Of them, two showed significant activity, namely MP-P5 (IC50 = 25.07 ± 0.05 μM, similar to activity against HIV) and PP-P1 (IC50 = 1.08 ± 0.14 μM). Additionally, a treatment assay performed with MP-P5 and PP-P1 revealed that both PPCs efficiently inhibit ZIKV replication when added 1 h and 7 h post-infection. As observed for HIV, non-conjugated porphyrins did not show activity against ZIKV, reinforcing the claim that BBBpS conjugation is not only critical for BBB/BPB translocation but also for antiviral activity. On the other hand, and somewhat unexpectedly, PP-P1, shown to be inactive in vitro against HIV, emerged as the most active anti-ZIKV conjugate. As described in the literature, the light-independent mechanism of action of porphyrins is based on a direct perturbation of the viral envelope. Porphyrins interact and accumulate on the envelope lipid membrane, causing a decrease in order and a consequent phase alteration that impairs viral entry processes. Since we ensured no-light conditions and no metal cations are coordinated to the porphyrin rings of the PPCsavoiding generation of reactive oxygen speciesthe antiviral light-independent mechanism is the only plausible one. Thus, one may confidently suggest that the PP-P1 specific anti-ZIKV activity is guided by preferential interaction with the ZIKV viral envelope and/or components.
Click to Show/Hide
|
||||
| In Vitro Model | HIV Infection | Human immunodeficiency virus | 12721 | ||
References
