Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00149
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Cyclic peptide [W(WR)4K(βAla)]
|
|||||
| Structure |
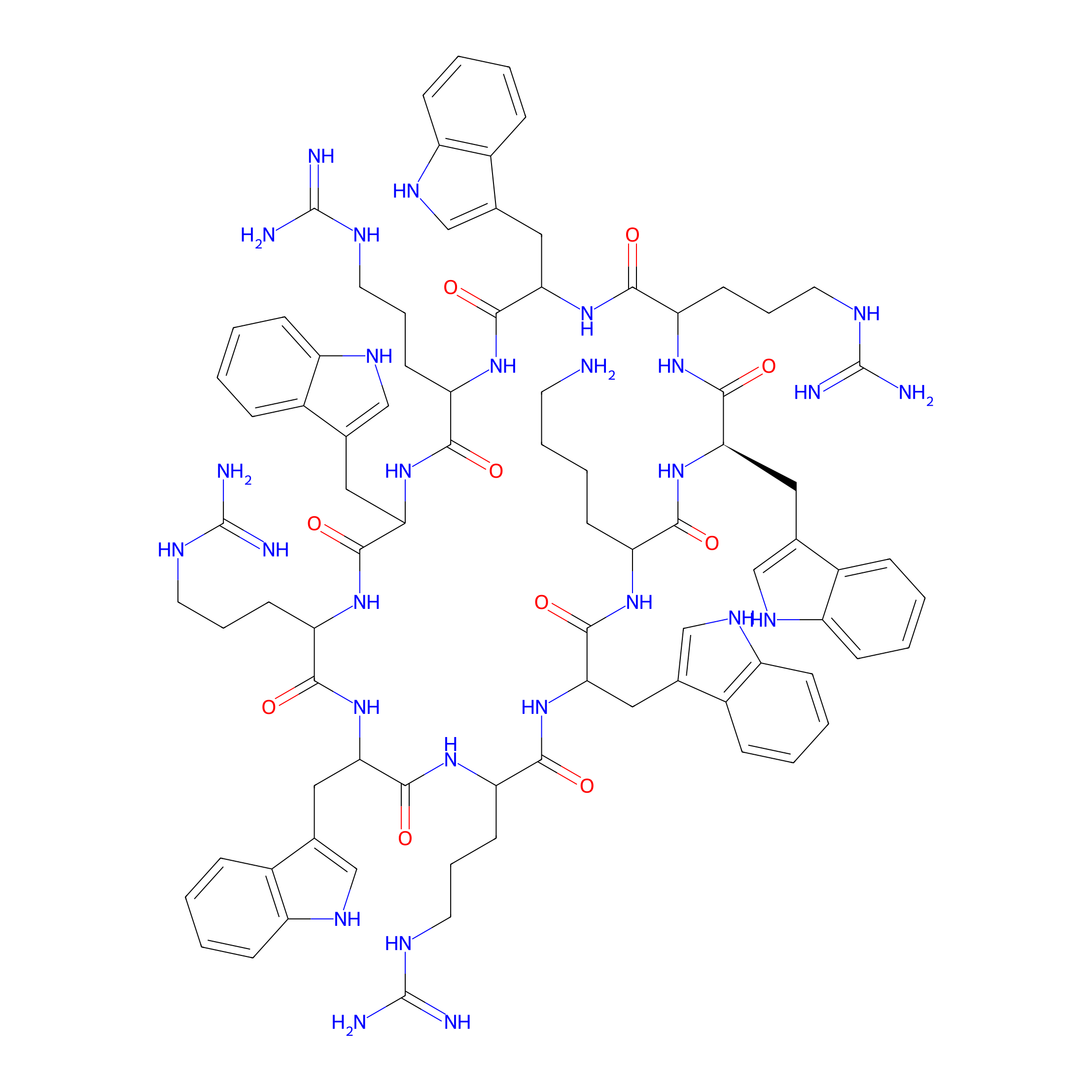
|
|||||
| Sequence |
WWRWRWRWRK
|
|||||
| Peptide Type |
Cyclic
|
|||||
| PDC Transmembrane Types | Cell-penetrating peptides (CPPs) | |||||
| Formula |
C85H110N28O10
|
|||||
| Isosmiles |
N=C(N)NCCCC1NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(CCCNC(=N)N)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(CCCNC(=N)N)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)C(CCCCN)NC(=O)C(Cc2c[nH]c3ccccc23)NC(=O)C(CCCNC(=N)N)NC(=O)C(Cc2c[nH]c3ccccc23)NC1=O
|
|||||
| InChI |
InChI=1S/C85H110N28O10/c86-32-12-11-27-62-72(114)109-68(38-48-43-100-58-23-7-2-18-53(48)58)78(120)105-64(29-14-34-96-83(89)90)74(116)111-70(40-50-45-102-60-25-9-4-20-55(50)60)80(122)107-66(31-16-36-98-85(93)94)76(118)113-71(41-51-46-103-61-26-10-5-21-56(51)61)81(123)108-65(30-15-35-97-84(91)92)75(117)112-69(39-49-44-101-59-24-8-3-19-54(49)59)79(121)106-63(28-13-33-95-82(87)88)73(115)110-67(77(119)104-62)37-47-42-99-57-22-6-1-17-52(47)57/h1-10,17-26,42-46,62-71,99-103H,11-16,27-41,86H2,(H,104,119)(H,105,120)(H,106,121)(H,107,122)(H,108,123)(H,109,114)(H,110,115)(H,111,116)(H,112,117)(H,113,118)(H4,87,88,95)(H4,89,90,96)(H4,91,92,97)(H4,93,94,98)/t62?,63?,64?,65?,66?,67?,68-,69?,70?,71?/m1/s1
|
|||||
| InChIKey |
CIOVDCFIYHKAPQ-NVVJDERTSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1684.001
|
Polar area
|
643.57
|
||
|
Complexity
|
1682.895972
|
xlogp Value
|
0.65008
|
|||
|
Heavy Count
|
123
|
Rot Bonds
|
30
|
|||
|
Hbond acc
|
15
|
Hbond Donor
|
28
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
PDC-CPT1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
38%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
Antiproliferative results showed that PTX1 inhibited cell proliferation by 18.7%. The anti-proliferative activity of CPT1 was diminished by 1.9-fold as compared to CPT whereas the activity of CPT2 was comparable to CPT, since CPT2 reduced the cell viability to 61%.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
PDC-PTX1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
60%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
Antiproliferative results showed that PTX1 inhibited cell proliferation by 18.7%. The anti-proliferative activity of CPT1 was diminished by 1.9-fold as compared to CPT whereas the activity of CPT2 was comparable to CPT, since CPT2 reduced the cell viability to 61%.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
80%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
The cytotoxicity of PTX and PTX1 was further evaluated in the normal human embryonic kidney cells (HEK-293) at 5 uM which showed reduced cell proliferation by ~34% and 18%, respectively, after 72 h using MTT assay, as shown in Figure 2.
|
||||
| In Vitro Model | Normal | HEK-298 cell | Homo sapiens | ||
PDC-CPT2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
80%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
Antiproliferative results showed that PTX1 inhibited cell proliferation by 18.7%. The anti-proliferative activity of CPT1 was diminished by 1.9-fold as compared to CPT whereas the activity of CPT2 was comparable to CPT, since CPT2 reduced the cell viability to 61%.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
References
