Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00160
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
123B9
|
|||||
| Structure |
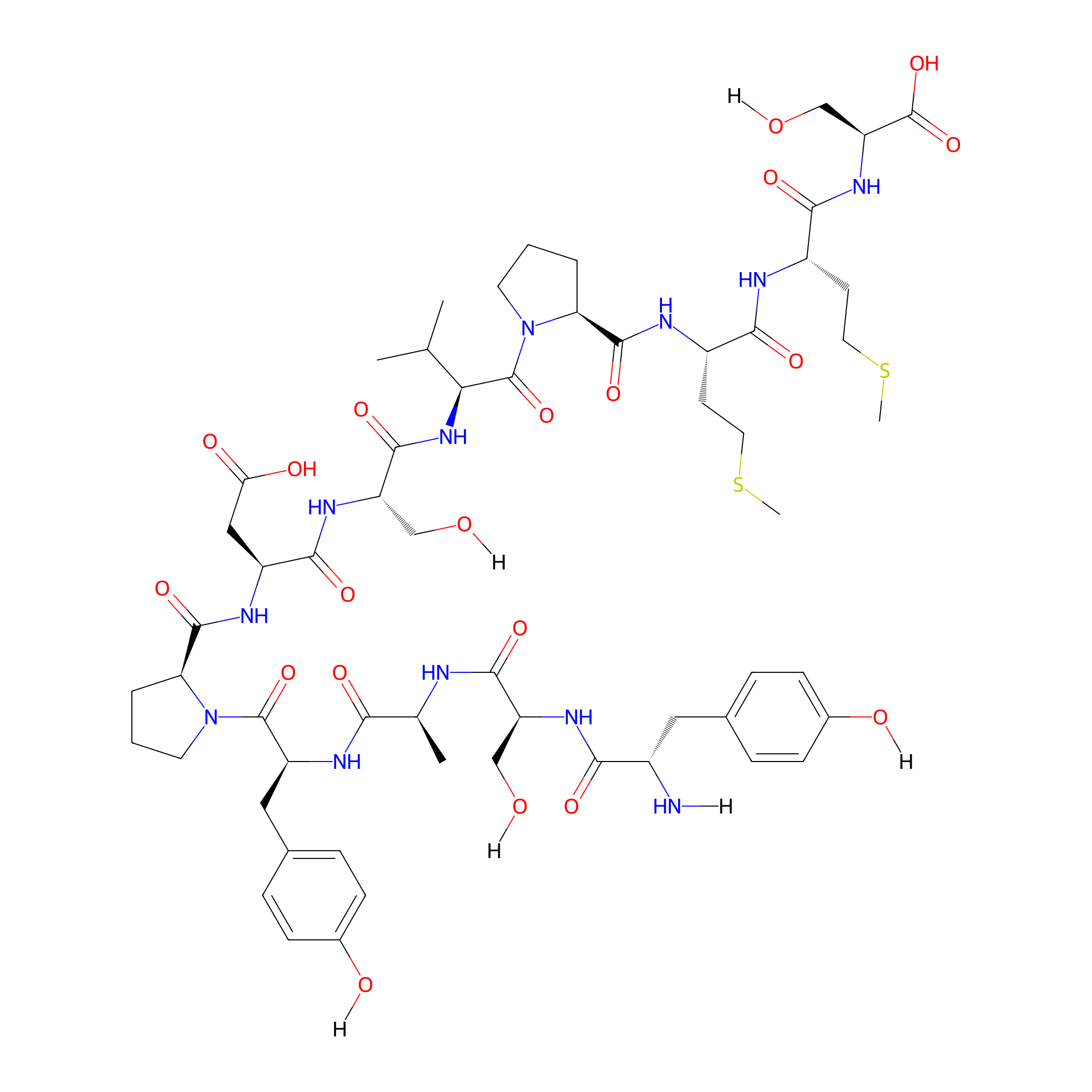
|
|||||
| Sequence |
YSAYPDSVPMMS
|
|||||
| Peptide Type |
Linear
|
|||||
| Peptide Half Life Period |
>> 2 h
|
|||||
| Receptor Name |
Ephrin type-A receptor 2 (EPHA2)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Metabolism |
The mean plasma concentration over time following 123B9 IV administration (30 mg/kg, n = 2) indicated that the compound was detectable in vivo at each time point tested (30 min, 1 hr, 2 hr, 4 hr), with a mean concentration in plasma of 447 ± 268 ng/ml and 569 ± 86 ng/ml still detectable at 2 hr and 4 hr, respectively.
|
|||||
| Excretion |
EphA2 ligand 123B9, lacking the free N-terminal amine, was very stable, with over 95% intact compound detected in plasma after 120 min (Figure 2A; t1/2 >> 2 hr).
|
|||||
| Formula |
C59H86N12O20S2
|
|||||
| Isosmiles |
[H]N[C@@H](Cc1ccc(O[H])cc1)C(=O)N[C@@H](CO[H])C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1ccc(O[H])cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CO[H])C(=O)N[C@H](C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CO[H])C(=O)O)C(C)C
|
|||||
| InChI |
InChI=1S/C59H86N12O20S2/c1-30(2)47(58(89)71-21-7-9-45(71)55(86)63-38(19-23-93-5)50(81)62-37(18-22-92-4)51(82)68-43(29-74)59(90)91)69-54(85)42(28-73)67-52(83)39(26-46(77)78)64-56(87)44-8-6-20-70(44)57(88)40(25-33-12-16-35(76)17-13-33)65-48(79)31(3)61-53(84)41(27-72)66-49(80)36(60)24-32-10-14-34(75)15-11-32/h10-17,30-31,36-45,47,72-76H,6-9,18-29,60H2,1-5H3,(H,61,84)(H,62,81)(H,63,86)(H,64,87)(H,65,79)(H,66,80)(H,67,83)(H,68,82)(H,69,85)(H,77,78)(H,90,91)/t31-,36-,37-,38-,39-,40-,41-,42-,43-,44-,45-,47-/m0/s1
|
|||||
| InChIKey |
WMDHJFJOXRIVNH-JZPYIPNMSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1347.535
|
Polar area
|
504.29
|
||
|
Complexity
|
1346.552275
|
xlogp Value
|
-4.7262
|
|||
|
Heavy Count
|
93
|
Rot Bonds
|
43
|
|||
|
Hbond acc
|
21
|
Hbond Donor
|
17
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| Binding constant | 3.9 μM | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
To further verify the binding affinity and selectivity of the resulting conjugates for the EphA2 ligand-binding domain (LBD), we expressed and purified the EphA2 and EphA4 ligand-binding domains (EphA2-LBD and EphA4-LBD). These proteins were dissolved to final concentrations of 100 μM in 50 mM phosphate buffer (pH = 6.5) containing 100 mM NaCl. The isothermal-titration-calorimetry (ITC) measurements under these experimental conditions revealed that 123B9 and the (123B9)2-motif bound to EphA2 with similarKdvalues of 3.9 and 4.9 μM, respectively (Figure
Click to Show/Hide
|
|||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
(123B9)2L2PTX [Investigative]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Number of Lung metastases nodules |
5
|
|||
| Administration Time | 2 weeks | ||||
| Administration Dosage | 24.5 mg/kg | ||||
| Description |
We observed the clear and significant (p < 0.0001) beneficial effects of the dimer drug on lung metastasis (Figure5A,B), with a reduction of the gross lung-metastasis count by more than 75% compared with those in the control and Abraxane groups.
|
||||
| In Vivo Model | BALB/c mice syngeneic breast-cancer metastasis model. | ||||
References
