Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00164
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
c[DKP-RGD]-PEG<sub>4</sub>-sC18
|
|||||
| Structure |
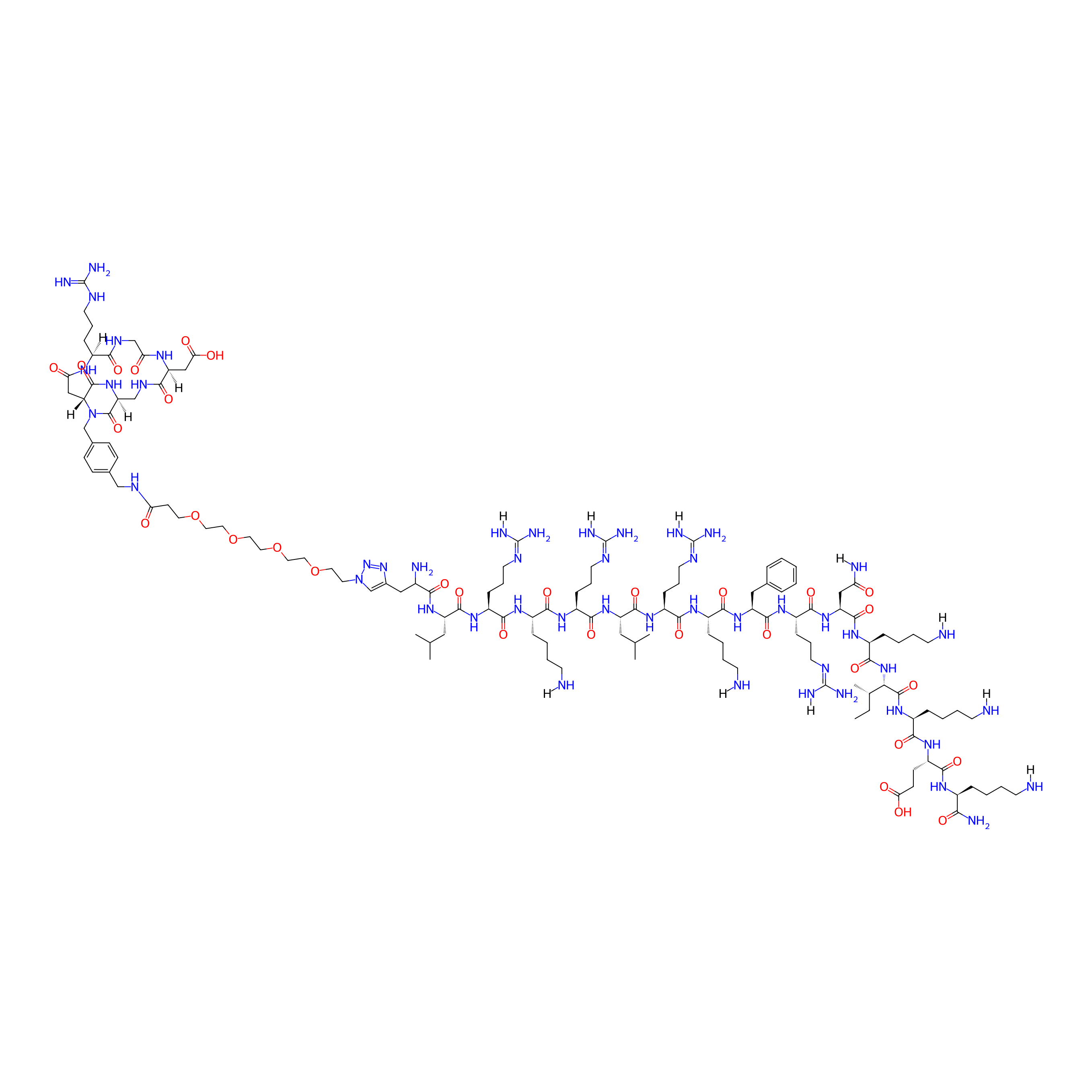
|
|||||
| Sequence |
LRKRLRKFRNKIKEK
|
|||||
| Peptide Type |
Linear
|
|||||
| PDC Transmembrane Types | Cell-penetrating peptides (CPPs) | |||||
| Formula |
C133H228N48O32
|
|||||
| Isosmiles |
[H]NCCCC[C@H](NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CCCCN[H])NC(=O)[C@@]([H])(NC(=O)[C@H](CCCCN[H])NC(=O)[C@H](CC(=O)N[H])NC(=O)[C@H](CCC/N=C(/N)N[H])NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCCCN[H])NC(=O)[C@H](CCC/N=C(/N)N[H])NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC/N=C(/N)N[H])NC(=O)[C@H](CCCCN[H])NC(=O)[C@H](CCC/N=C(/N)N[H])NC(=O)[C@H](CC(C)C)NC(=O)C(N)Cc1cn(CCOCCOCCOCCOCCC(=O)NCc2ccc(CN3C(=O)[C@@H]4CNC(=O)[C@H](CC(=O)O)NC(=O)CNC(=O)[C@H](CCCNC(=N)N)NC(=O)C[C@@H]3C(=O)N4)cc2)nn1)[C@@H](C)CC)C(N)=O
|
|||||
| InChI |
InChI=1S/C133H228N48O32/c1-7-78(6)108(127(208)171-88(31-14-19-48-137)114(195)170-94(42-43-106(186)187)120(201)162-84(109(141)190)28-11-16-45-134)177-121(202)89(32-15-20-49-138)166-125(206)98(68-102(140)182)175-119(200)93(37-25-54-156-133(150)151)169-124(205)97(66-79-26-9-8-10-27-79)174-117(198)87(30-13-18-47-136)164-116(197)91(35-23-52-154-131(146)147)168-123(204)96(65-77(4)5)173-118(199)92(36-24-53-155-132(148)149)165-113(194)86(29-12-17-46-135)163-115(196)90(34-22-51-153-130(144)145)167-122(203)95(64-76(2)3)172-110(191)83(139)67-82-75-180(179-178-82)55-57-211-59-61-213-63-62-212-60-58-210-56-44-103(183)157-71-80-38-40-81(41-39-80)74-181-101-70-104(184)160-85(33-21-50-152-129(142)143)111(192)159-73-105(185)161-99(69-107(188)189)112(193)158-72-100(128(181)209)176-126(101)207/h8-10,26-27,38-41,75-78,83-101,108H,7,11-25,28-37,42-74,134-139H2,1-6H3,(H2,140,182)(H2,141,190)(H,157,183)(H,158,193)(H,159,192)(H,160,184)(H,161,185)(H,162,201)(H,163,196)(H,164,197)(H,165,194)(H,166,206)(H,167,203)(H,168,204)(H,169,205)(H,170,195)(H,171,208)(H,172,191)(H,173,199)(H,174,198)(H,175,200)(H,176,207)(H,177,202)(H,186,187)(H,188,189)(H4,142,143,152)(H4,144,145,153)(H4,146,147,154)(H4,148,149,155)(H4,150,151,156)/t78-,83?,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101+,108-/m0/s1
|
|||||
| InChIKey |
RBAKWJKQPHNSAD-MSYGNZFVSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
3011.591
|
Polar area
|
1335.44
|
||
|
Complexity
|
3009.768927
|
xlogp Value
|
-13.80203
|
|||
|
Heavy Count
|
213
|
Rot Bonds
|
112
|
|||
|
Hbond acc
|
44
|
Hbond Donor
|
42
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| IC50 | 2.5 ± 0.2 nM | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
Human integrin receptors avb3 (R&D Systems, Minneapolis, MN, USA) and avb5 (EMD Millipore Corporation, Inc., Temecula, CA, USA) were diluted to 0.5 μg/mL in coating buffer containing 20 mM Tris-HCl (pH 7.4), 150 mM NaCl, 1 mM MnCl2, 2 mM CaCl2, and 1 mM MgCl2. An aliquot of diluted receptor (100 μL/well) was added to 96-well microtiter plates (Nunc MaxiSorp, Termo Fisher Scientific, Roskilde, DK) and incubated overnight at 4 °C. The plates were incubated with blocking solution (coating buffer plus 1% bovine serum albumin) for additional 2 h at room temperature to block nonspecific binding. After washing 2 times with blocking solution, plates were incubated shaking in the dark for 3 h at room temperature, with various concentrations (10-5-10-12 M) of test compounds in the presence of 1 μg/mL vitronectin (Molecular Innovations, Novi, MI, USA) biotinylated using an EZ-Link Sulfo-NHS-Biotinylation kit (Pierce, Rockford, IL, USA). After washing 3 times, the plates were incubated shaking for 1 h in the dark, at room temperature, with streptavidin-biotinylated peroxidase complex (Amersham Biosciences, Uppsala, Sweden). After washing 3 times with blocking solution, plates were incubated with 100 μL/well of Substrate Reagent Solution (R&D Systems, Minneapolis, MN, USA) for 30 min shaking in the dark, before stopping the reaction with the addition of 50 μL/well 2 N H2SO4. Absorbance at 415 nm was read in a Synergy HT Multi-Detection Microplate Reader (BioTek Instruments, Inc.). Each data point represents the average of triplicate wells; data analysis was carried out by nonlinear regression analysis with GraphPad Prism software. Each experiment was repeated in duplicate.
Click to Show/Hide
|
|||||
| Experimental Condition | Biotinylated vitronectin(αvβ3) | |||||
| Peptide Activity Information 2 | [1] | |||||
| IC50 | 15.3 ± 5.2 nM | |||||
| Binding Affinity Assay |
Human integrin receptors avb3 (R&D Systems, Minneapolis, MN, USA) and avb5 (EMD Millipore Corporation, Inc., Temecula, CA, USA) were diluted to 0.5 μg/mL in coating buffer containing 20 mM Tris-HCl (pH 7.4), 150 mM NaCl, 1 mM MnCl2, 2 mM CaCl2, and 1 mM MgCl2. An aliquot of diluted receptor (100 μL/well) was added to 96-well microtiter plates (Nunc MaxiSorp, Termo Fisher Scientific, Roskilde, DK) and incubated overnight at 4 °C. The plates were incubated with blocking solution (coating buffer plus 1% bovine serum albumin) for additional 2 h at room temperature to block nonspecific binding. After washing 2 times with blocking solution, plates were incubated shaking in the dark for 3 h at room temperature, with various concentrations (10-5-10-12 M) of test compounds in the presence of 1 μg/mL vitronectin (Molecular Innovations, Novi, MI, USA) biotinylated using an EZ-Link Sulfo-NHS-Biotinylation kit (Pierce, Rockford, IL, USA). After washing 3 times, the plates were incubated shaking for 1 h in the dark, at room temperature, with streptavidin-biotinylated peroxidase complex (Amersham Biosciences, Uppsala, Sweden). After washing 3 times with blocking solution, plates were incubated with 100 μL/well of Substrate Reagent Solution (R&D Systems, Minneapolis, MN, USA) for 30 min shaking in the dark, before stopping the reaction with the addition of 50 μL/well 2 N H2SO4. Absorbance at 415 nm was read in a Synergy HT Multi-Detection Microplate Reader (BioTek Instruments, Inc.). Each data point represents the average of triplicate wells; data analysis was carried out by nonlinear regression analysis with GraphPad Prism software. Each experiment was repeated in duplicate.
Click to Show/Hide
|
|||||
| Experimental Condition | Biotinylated vitronectin(αvβ5) | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
c[DKP-RGD]-PEG4-sC18(dau=Aoa-GFLG-Lys8) [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
2.7 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
3.9 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
7.8 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
11 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
23.8 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
89.1 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
c[DKP-RGD]-PEG4-sC18(dau=Aoa-Lys8) [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
3 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
5.6 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
9.1 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
12.5 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds 1b and 3b demonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure 6). Importantly, 1b was more active than 3b demonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that 2b was less efficient than 1b but marginally more active than 3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that 3c is still significantly more active in U87 cells expressing integrin receptors. By contrast, 1c kept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to 1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
50.5 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds 1b and 3b demonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure 6). Importantly, 1b was more active than 3b demonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that 2b was less efficient than 1b but marginally more active than 3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that 3c is still significantly more active in U87 cells expressing integrin receptors. By contrast, 1c kept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to 1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
53.4 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds 1b and 3b demonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure 6). Importantly, 1b was more active than 3b demonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that 2b was less efficient than 1b but marginally more active than 3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that 3c is still significantly more active in U87 cells expressing integrin receptors. By contrast, 1c kept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to 1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
sC18(dau-Aoa-Lys8) [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
5 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
5.8 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
9.2 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
25.1 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to3b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
42.2 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 100 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to2b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
References
