Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00166
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
STRAP-3
|
|||||
| Structure |
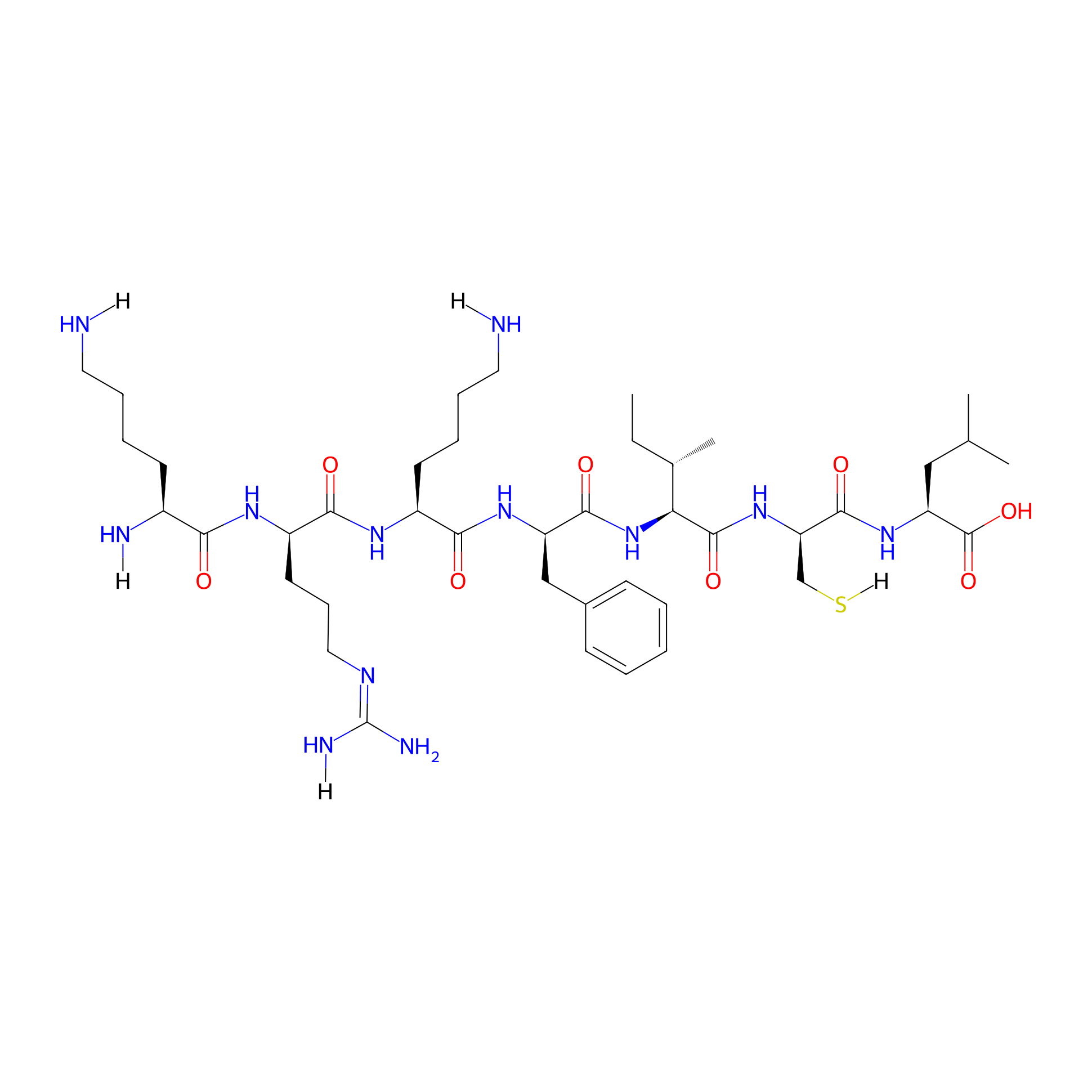
|
|||||
| Sequence |
KRKFICL
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
Cell membrane
|
|||||
| PDC Transmembrane Types | Cell-penetrating peptides (CPPs) | |||||
| Distribution |
All the mice were continuously imaged for 4 h after the STRAP-Cy7 administration. In the healthy mice, the peptide is distributed throughout the body and was seen as surface fluorescence emitted from the liver, lungs, and bladder. Fluorescence emission from the urine in the bladder readily appeared in the mice following the injection. With time, fluorescence is cleared from the lungs and bladder. At the end of four hours, the peptide surface fluorescence was seen in the liver and kidney. In the tumor-bearing mice, the fluorescence emission was observed over the liver and kidney from 20 min. A gradual increase in fluorescence at the tumor site was observed from 2 h, which reached the maximum at 4 h. Ex vivo imaging of organs from the tumor-bearing mouse at 24 h confirmed the retention of peptides only in the tumor and kidneys, indicating the elimination of the drug through the kidneys. Blood was collected from the mice prior to the injection and after the injection for a total of four hours. The highest plasma concentration was observed 20 min after peptide administration with a gradual drop in the plasma concentration over 4 h both in tumor-bearing and healthy animals. This indicates that the plasma clearance of the peptide was unaffected by the tumor status of the mice.
|
|||||
| Formula |
C42H74N12O8S
|
|||||
| Isosmiles |
[H]NCCCC[C@H](N[H])C(=O)N[C@H](CCC/N=C(/N)N[H])C(=O)N[C@@H](CCCCN[H])C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@]([H])(C(=O)N[C@H](CS[H])C(=O)N[C@@H](CC(C)C)C(=O)O)[C@@H](C)CC
|
|||||
| InChI |
InChI=1S/C42H74N12O8S/c1-5-26(4)34(40(60)53-33(24-63)39(59)52-32(41(61)62)22-25(2)3)54-38(58)31(23-27-14-7-6-8-15-27)51-37(57)29(17-10-12-20-44)50-36(56)30(18-13-21-48-42(46)47)49-35(55)28(45)16-9-11-19-43/h6-8,14-15,25-26,28-34,63H,5,9-13,16-24,43-45H2,1-4H3,(H,49,55)(H,50,56)(H,51,57)(H,52,59)(H,53,60)(H,54,58)(H,61,62)(H4,46,47,48)/t26-,28-,29-,30+,31+,32-,33+,34-/m0/s1
|
|||||
| InChIKey |
XSVYSYBQSSNRAI-GHHKMHCSSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
907.197
|
Polar area
|
354.36
|
||
|
Complexity
|
906.5473283
|
xlogp Value
|
-1.1166
|
|||
|
Heavy Count
|
63
|
Rot Bonds
|
36
|
|||
|
Hbond acc
|
12
|
Hbond Donor
|
13
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
STRAP-4-MTX [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.01 ± 0.22 µM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Electrostatics modulates the interactions of CPPs with the corresponding cell surface. Therefore, for the design of the amino acid side chain sequences, we reverse-engineered the spatial electrostatic potential distribution of the previously designed peptides with cell penetration ability. The spatial electrostatic fingerprints of the earlier designed peptides were generated as described previously. The reverse-engineered sequences with a shorter (7-mer) chain length were compared to the previous designs through multiple iterations of amino acid sequences. Their respective electrostatic profiles were used for the design of four syndiotactic, cationic, amphipathic peptides with optimal sequence selection. These syndiotactic re-engineered amphipathic peptides (STRAPs) were tested for their ability to penetrate cells and deliver a small molecule (methotrexate). The amino acid constitutions of STRAP-1, STRAP-2, STRAP-3, and STRAP-4 are the same. However, STRAP-2 is the stereochemically reversed STRAP-1; similarly, STRAP-4 is the stereochemically reversed STRAP-3. The use of LDLD and DLDL stereochemical sequences in the design of STRAPs resulted in the differential electrostatic signatures for the same amino acid sequences. The designed peptides have a higher cellular uptake in TNBC cells (MDA-MB-231) than the standard control TAT peptide. They could penetrate cells by both active and passive processes, and their activity is not reduced in biological fluids (human plasma and bovine serum). Furthermore, the delivery of MTX as STRAP-MTX conjugates helped to overcome the drug resistance of the MDA-MB-231 cells under in vitro conditions. The delivery of the STARP-4-MTX conjugate in TNBC xenograft tumors was more effective in the reduction of both the tumor size and metastasis to the lungs, liver, spleen, and lymph nodes.
Click to Show/Hide
|
||||
| Description |
To deliver the functional molecule into the cells, we attached an anticancer drug, methotrexate (MTX), to the N-terminus of STRAPs. The peptides were tested for their capability to deliver the active drug molecule in breast and cervical cancer cells. MDA-MB-231 and HeLa cells were treated with varying concentrations of MTX and STRAP-MTX conjugates for 72 h. Cell viability was assessed using the MTT assay. The cytotoxicity of MTX increased when delivered as peptide-MTX conjugates to the cells. This resulted in an overall reduction in the inhibitory concentration cytotoxic to 50% of cells (IC50) for MTX. The MTX resistance for MDA-MB-231 cells is well-established. The IC50 for MTX in MDA-MB-231 cells was minimum when delivered as a STRAP-4-MTX conjugate. Similar lowering of IC50 values in HeLa cells was observed. We also treated HEK-293 cells with MTX-STRAP conjugates. The peptide-drug conjugates showed no significant toxicity to the HEK-293 cells under the tested conditions, thereby confirming the earlier reported observation of higher uptake rates in cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.34 ± 0.19 µM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Electrostatics modulates the interactions of CPPs with the corresponding cell surface. Therefore, for the design of the amino acid side chain sequences, we reverse-engineered the spatial electrostatic potential distribution of the previously designed peptides with cell penetration ability. The spatial electrostatic fingerprints of the earlier designed peptides were generated as described previously. The reverse-engineered sequences with a shorter (7-mer) chain length were compared to the previous designs through multiple iterations of amino acid sequences. Their respective electrostatic profiles were used for the design of four syndiotactic, cationic, amphipathic peptides with optimal sequence selection. These syndiotactic re-engineered amphipathic peptides (STRAPs) were tested for their ability to penetrate cells and deliver a small molecule (methotrexate). The amino acid constitutions of STRAP-1, STRAP-2, STRAP-3, and STRAP-4 are the same. However, STRAP-2 is the stereochemically reversed STRAP-1; similarly, STRAP-4 is the stereochemically reversed STRAP-3. The use of LDLD and DLDL stereochemical sequences in the design of STRAPs resulted in the differential electrostatic signatures for the same amino acid sequences. The designed peptides have a higher cellular uptake in TNBC cells (MDA-MB-231) than the standard control TAT peptide. They could penetrate cells by both active and passive processes, and their activity is not reduced in biological fluids (human plasma and bovine serum). Furthermore, the delivery of MTX as STRAP-MTX conjugates helped to overcome the drug resistance of the MDA-MB-231 cells under in vitro conditions. The delivery of the STARP-4-MTX conjugate in TNBC xenograft tumors was more effective in the reduction of both the tumor size and metastasis to the lungs, liver, spleen, and lymph nodes.
Click to Show/Hide
|
||||
| Description |
To deliver the functional molecule into the cells, we attached an anticancer drug, methotrexate (MTX), to the N-terminus of STRAPs. The peptides were tested for their capability to deliver the active drug molecule in breast and cervical cancer cells. MDA-MB-231 and HeLa cells were treated with varying concentrations of MTX and STRAP-MTX conjugates for 72 h. Cell viability was assessed using the MTT assay. The cytotoxicity of MTX increased when delivered as peptide-MTX conjugates to the cells. This resulted in an overall reduction in the inhibitory concentration cytotoxic to 50% of cells (IC50) for MTX. The MTX resistance for MDA-MB-231 cells is well-established. The IC50 for MTX in MDA-MB-231 cells was minimum when delivered as a STRAP-4-MTX conjugate. Similar lowering of IC50 values in HeLa cells was observed. We also treated HEK-293 cells with MTX-STRAP conjugates. The peptide-drug conjugates showed no significant toxicity to the HEK-293 cells under the tested conditions, thereby confirming the earlier reported observation of higher uptake rates in cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Renal cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 50 µM | |||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Electrostatics modulates the interactions of CPPs with the corresponding cell surface. Therefore, for the design of the amino acid side chain sequences, we reverse-engineered the spatial electrostatic potential distribution of the previously designed peptides with cell penetration ability. The spatial electrostatic fingerprints of the earlier designed peptides were generated as described previously. The reverse-engineered sequences with a shorter (7-mer) chain length were compared to the previous designs through multiple iterations of amino acid sequences. Their respective electrostatic profiles were used for the design of four syndiotactic, cationic, amphipathic peptides with optimal sequence selection. These syndiotactic re-engineered amphipathic peptides (STRAPs) were tested for their ability to penetrate cells and deliver a small molecule (methotrexate). The amino acid constitutions of STRAP-1, STRAP-2, STRAP-3, and STRAP-4 are the same. However, STRAP-2 is the stereochemically reversed STRAP-1; similarly, STRAP-4 is the stereochemically reversed STRAP-3. The use of LDLD and DLDL stereochemical sequences in the design of STRAPs resulted in the differential electrostatic signatures for the same amino acid sequences. The designed peptides have a higher cellular uptake in TNBC cells (MDA-MB-231) than the standard control TAT peptide. They could penetrate cells by both active and passive processes, and their activity is not reduced in biological fluids (human plasma and bovine serum). Furthermore, the delivery of MTX as STRAP-MTX conjugates helped to overcome the drug resistance of the MDA-MB-231 cells under in vitro conditions. The delivery of the STARP-4-MTX conjugate in TNBC xenograft tumors was more effective in the reduction of both the tumor size and metastasis to the lungs, liver, spleen, and lymph nodes.
Click to Show/Hide
|
||||
| Description |
To deliver the functional molecule into the cells, we attached an anticancer drug, methotrexate (MTX), to the N-terminus of STRAPs. The peptides were tested for their capability to deliver the active drug molecule in breast and cervical cancer cells. MDA-MB-231 and HeLa cells were treated with varying concentrations of MTX and STRAP-MTX conjugates for 72 h. Cell viability was assessed using the MTT assay. The cytotoxicity of MTX increased when delivered as peptide-MTX conjugates to the cells. This resulted in an overall reduction in the inhibitory concentration cytotoxic to 50% of cells (IC50) for MTX. The MTX resistance for MDA-MB-231 cells is well-established. The IC50 for MTX in MDA-MB-231 cells was minimum when delivered as a STRAP-4-MTX conjugate. Similar lowering of IC50 values in HeLa cells was observed. We also treated HEK-293 cells with MTX-STRAP conjugates. The peptide-drug conjugates showed no significant toxicity to the HEK-293 cells under the tested conditions, thereby confirming the earlier reported observation of higher uptake rates in cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | HEK293 cell | CVCL_0045 | ||
STRAP-3-MTX [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.22 ± 0.18 µM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Electrostatics modulates the interactions of CPPs with the corresponding cell surface. Therefore, for the design of the amino acid side chain sequences, we reverse-engineered the spatial electrostatic potential distribution of the previously designed peptides with cell penetration ability. The spatial electrostatic fingerprints of the earlier designed peptides were generated as described previously. The reverse-engineered sequences with a shorter (7-mer) chain length were compared to the previous designs through multiple iterations of amino acid sequences. Their respective electrostatic profiles were used for the design of four syndiotactic, cationic, amphipathic peptides with optimal sequence selection. These syndiotactic re-engineered amphipathic peptides (STRAPs) were tested for their ability to penetrate cells and deliver a small molecule (methotrexate). The amino acid constitutions of STRAP-1, STRAP-2, STRAP-3, and STRAP-4 are the same. However, STRAP-2 is the stereochemically reversed STRAP-1; similarly, STRAP-4 is the stereochemically reversed STRAP-3. The use of LDLD and DLDL stereochemical sequences in the design of STRAPs resulted in the differential electrostatic signatures for the same amino acid sequences. The designed peptides have a higher cellular uptake in TNBC cells (MDA-MB-231) than the standard control TAT peptide. They could penetrate cells by both active and passive processes, and their activity is not reduced in biological fluids (human plasma and bovine serum). Furthermore, the delivery of MTX as STRAP-MTX conjugates helped to overcome the drug resistance of the MDA-MB-231 cells under in vitro conditions. The delivery of the STARP-4-MTX conjugate in TNBC xenograft tumors was more effective in the reduction of both the tumor size and metastasis to the lungs, liver, spleen, and lymph nodes.
Click to Show/Hide
|
||||
| Description |
To deliver the functional molecule into the cells, we attached an anticancer drug, methotrexate (MTX), to the N-terminus of STRAPs. The peptides were tested for their capability to deliver the active drug molecule in breast and cervical cancer cells. MDA-MB-231 and HeLa cells were treated with varying concentrations of MTX and STRAP-MTX conjugates for 72 h. Cell viability was assessed using the MTT assay. The cytotoxicity of MTX increased when delivered as peptide-MTX conjugates to the cells. This resulted in an overall reduction in the inhibitory concentration cytotoxic to 50% of cells (IC50) for MTX. The MTX resistance for MDA-MB-231 cells is well-established. The IC50 for MTX in MDA-MB-231 cells was minimum when delivered as a STRAP-4-MTX conjugate. Similar lowering of IC50 values in HeLa cells was observed. We also treated HEK-293 cells with MTX-STRAP conjugates. The peptide-drug conjugates showed no significant toxicity to the HEK-293 cells under the tested conditions, thereby confirming the earlier reported observation of higher uptake rates in cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) |
1.79 ± 0.31 µM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Electrostatics modulates the interactions of CPPs with the corresponding cell surface. Therefore, for the design of the amino acid side chain sequences, we reverse-engineered the spatial electrostatic potential distribution of the previously designed peptides with cell penetration ability. The spatial electrostatic fingerprints of the earlier designed peptides were generated as described previously. The reverse-engineered sequences with a shorter (7-mer) chain length were compared to the previous designs through multiple iterations of amino acid sequences. Their respective electrostatic profiles were used for the design of four syndiotactic, cationic, amphipathic peptides with optimal sequence selection. These syndiotactic re-engineered amphipathic peptides (STRAPs) were tested for their ability to penetrate cells and deliver a small molecule (methotrexate). The amino acid constitutions of STRAP-1, STRAP-2, STRAP-3, and STRAP-4 are the same. However, STRAP-2 is the stereochemically reversed STRAP-1; similarly, STRAP-4 is the stereochemically reversed STRAP-3. The use of LDLD and DLDL stereochemical sequences in the design of STRAPs resulted in the differential electrostatic signatures for the same amino acid sequences. The designed peptides have a higher cellular uptake in TNBC cells (MDA-MB-231) than the standard control TAT peptide. They could penetrate cells by both active and passive processes, and their activity is not reduced in biological fluids (human plasma and bovine serum). Furthermore, the delivery of MTX as STRAP-MTX conjugates helped to overcome the drug resistance of the MDA-MB-231 cells under in vitro conditions. The delivery of the STARP-4-MTX conjugate in TNBC xenograft tumors was more effective in the reduction of both the tumor size and metastasis to the lungs, liver, spleen, and lymph nodes.
Click to Show/Hide
|
||||
| Description |
To deliver the functional molecule into the cells, we attached an anticancer drug, methotrexate (MTX), to the N-terminus of STRAPs. The peptides were tested for their capability to deliver the active drug molecule in breast and cervical cancer cells. MDA-MB-231 and HeLa cells were treated with varying concentrations of MTX and STRAP-MTX conjugates for 72 h. Cell viability was assessed using the MTT assay. The cytotoxicity of MTX increased when delivered as peptide-MTX conjugates to the cells. This resulted in an overall reduction in the inhibitory concentration cytotoxic to 50% of cells (IC50) for MTX. The MTX resistance for MDA-MB-231 cells is well-established. The IC50 for MTX in MDA-MB-231 cells was minimum when delivered as a STRAP-4-MTX conjugate. Similar lowering of IC50 values in HeLa cells was observed. We also treated HEK-293 cells with MTX-STRAP conjugates. The peptide-drug conjugates showed no significant toxicity to the HEK-293 cells under the tested conditions, thereby confirming the earlier reported observation of higher uptake rates in cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Renal cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 50 µM | |||
| Administration Time | 72 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Electrostatics modulates the interactions of CPPs with the corresponding cell surface. Therefore, for the design of the amino acid side chain sequences, we reverse-engineered the spatial electrostatic potential distribution of the previously designed peptides with cell penetration ability. The spatial electrostatic fingerprints of the earlier designed peptides were generated as described previously. The reverse-engineered sequences with a shorter (7-mer) chain length were compared to the previous designs through multiple iterations of amino acid sequences. Their respective electrostatic profiles were used for the design of four syndiotactic, cationic, amphipathic peptides with optimal sequence selection. These syndiotactic re-engineered amphipathic peptides (STRAPs) were tested for their ability to penetrate cells and deliver a small molecule (methotrexate). The amino acid constitutions of STRAP-1, STRAP-2, STRAP-3, and STRAP-4 are the same. However, STRAP-2 is the stereochemically reversed STRAP-1; similarly, STRAP-4 is the stereochemically reversed STRAP-3. The use of LDLD and DLDL stereochemical sequences in the design of STRAPs resulted in the differential electrostatic signatures for the same amino acid sequences. The designed peptides have a higher cellular uptake in TNBC cells (MDA-MB-231) than the standard control TAT peptide. They could penetrate cells by both active and passive processes, and their activity is not reduced in biological fluids (human plasma and bovine serum). Furthermore, the delivery of MTX as STRAP-MTX conjugates helped to overcome the drug resistance of the MDA-MB-231 cells under in vitro conditions. The delivery of the STARP-4-MTX conjugate in TNBC xenograft tumors was more effective in the reduction of both the tumor size and metastasis to the lungs, liver, spleen, and lymph nodes.
Click to Show/Hide
|
||||
| Description |
To deliver the functional molecule into the cells, we attached an anticancer drug, methotrexate (MTX), to the N-terminus of STRAPs. The peptides were tested for their capability to deliver the active drug molecule in breast and cervical cancer cells. MDA-MB-231 and HeLa cells were treated with varying concentrations of MTX and STRAP-MTX conjugates for 72 h. Cell viability was assessed using the MTT assay. The cytotoxicity of MTX increased when delivered as peptide-MTX conjugates to the cells. This resulted in an overall reduction in the inhibitory concentration cytotoxic to 50% of cells (IC50) for MTX. The MTX resistance for MDA-MB-231 cells is well-established. The IC50 for MTX in MDA-MB-231 cells was minimum when delivered as a STRAP-4-MTX conjugate. Similar lowering of IC50 values in HeLa cells was observed. We also treated HEK-293 cells with MTX-STRAP conjugates. The peptide-drug conjugates showed no significant toxicity to the HEK-293 cells under the tested conditions, thereby confirming the earlier reported observation of higher uptake rates in cancerous cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | HEK293 cell | CVCL_0045 | ||
References
