Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00171
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Bicycle toxin conjugate precursor
|
|||||
| Structure |
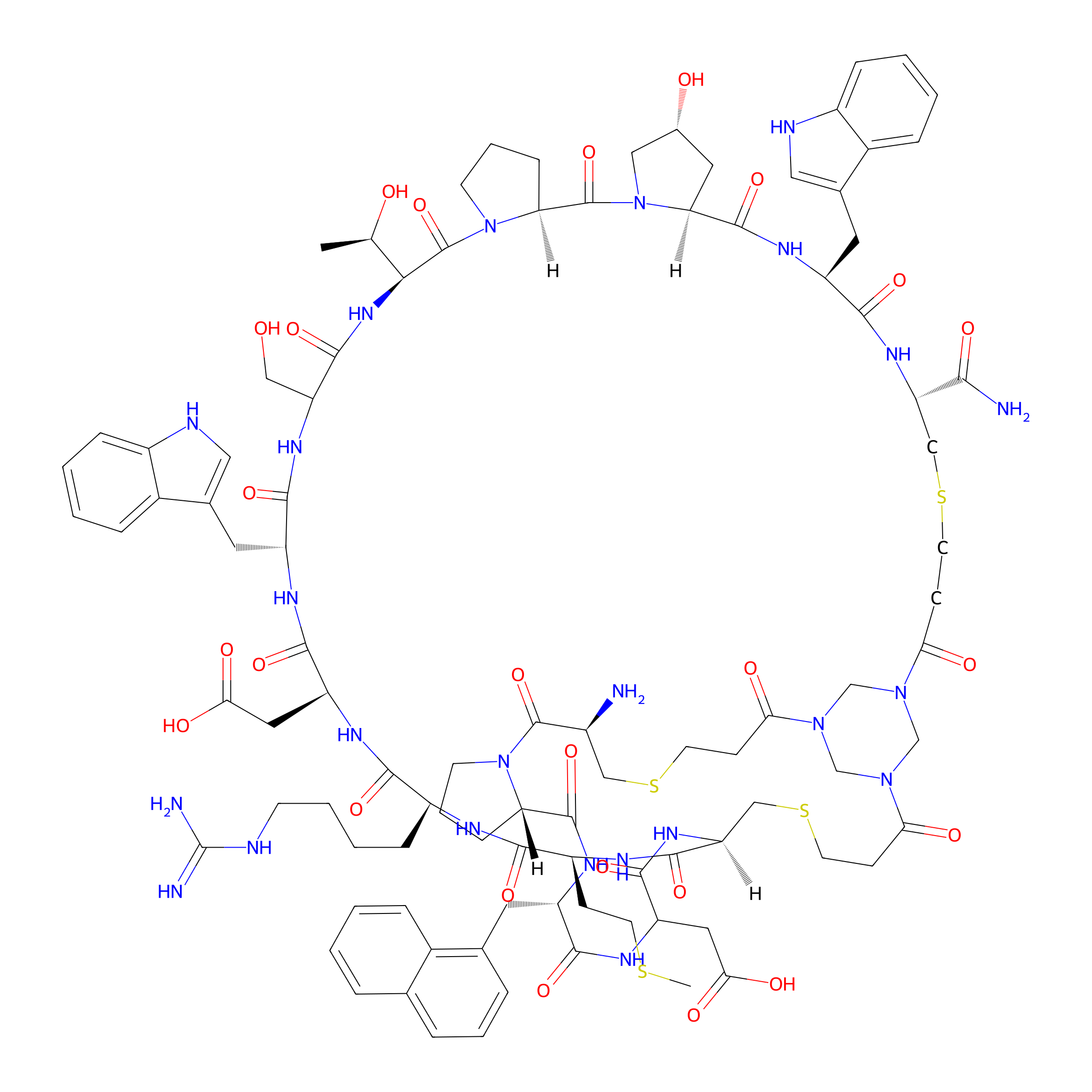
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Nectin-4 (NECTIN4)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C98H130N24O25S4
|
|||||
| Isosmiles |
CSCC[C@@H]1NC(=O)[C@@H]2CSCCC(=O)N3CN(CN(C3)C(=O)CCSC[C@H](N)C(=O)N3CCC[C@H]3C(=O)N[C@@H](Cc3cccc4ccccc34)C(=O)NC(CC(=O)O)C(=O)N2)C(=O)CCSC[C@@H](C(N)=O)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H]2C[C@@H](O)CN2C(=O)[C@@H]2CCCN2C(=O)[C@H]([C@@H](C)O)NC(=O)C(CO)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CCCCNC(=N)N)NC1=O
|
|||||
| InChI |
InChI=1S/C98H130N24O25S4/c1-52(124)82-97(147)121-31-13-24-75(121)96(146)122-44-57(125)39-76(122)94(144)112-68(38-56-43-105-63-21-8-6-19-60(56)63)88(138)114-72(83(100)133)47-150-34-27-78(127)118-49-117-50-119(51-118)79(128)28-35-151-48-73(115-90(140)70(41-81(131)132)110-86(136)66(36-54-16-11-15-53-14-3-4-17-58(53)54)111-93(143)74-23-12-30-120(74)95(145)61(99)46-149-33-26-77(117)126)92(142)107-65(25-32-148-2)85(135)106-64(22-9-10-29-103-98(101)102)84(134)109-69(40-80(129)130)89(139)108-67(87(137)113-71(45-123)91(141)116-82)37-55-42-104-62-20-7-5-18-59(55)62/h3-8,11,14-21,42-43,52,57,61,64-76,82,104-105,123-125H,9-10,12-13,22-41,44-51,99H2,1-2H3,(H2,100,133)(H,106,135)(H,107,142)(H,108,139)(H,109,134)(H,110,136)(H,111,143)(H,112,144)(H,113,137)(H,114,138)(H,115,140)(H,116,141)(H,129,130)(H,131,132)(H4,101,102,103)/t52-,57-,61+,64+,65+,66+,67-,68+,69+,70?,71?,72+,73+,74+,75+,76+,82+/m1/s1
|
|||||
| InChIKey |
FAJMTKQFCKVNHJ-OCLUNYMCSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
2172.529
|
Polar area
|
739.84
|
||
|
Complexity
|
2170.85218
|
xlogp Value
|
-4.58253
|
|||
|
Heavy Count
|
151
|
Rot Bonds
|
21
|
|||
|
Hbond acc
|
29
|
Hbond Donor
|
23
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1] | |||||
| KD | 2.5 nM | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
KD determined by surface plasmon resonance (SPR)
|
|||||
| Experimental Condition | CM5 (GE Healthcare) or CMD5000 (Xantec) chip in the vitro(Nectin-4 of the parent Bicycle) | |||||
| Peptide Activity Information 2 | [1] | |||||
| KD | 12.9 nM | |||||
| Binding Affinity Assay |
KD determined by surface plasmon resonance (SPR)
|
|||||
| Experimental Condition | CM5 (GE Healthcare) or CMD5000 (Xantec) chip in the vitro(endogenous Nectin-4 on cells) | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
BT8009 [Phase 2/3]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
23%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 1 mg/kg | ||||
| MOA of PDC |
The cell adhesion molecule Nectin-4 shows elevated expression in multiple tumor types correlated with poor prognosis. Nectin-4-directed ADCs show efficacy in multiple xenograft tumor models. The ADC enfortumab vedotin (EV) delivers the antimitotic toxin MMAE to Nectin-4-expressing cells via internalization and cleavage of a valine-citrulline dipeptide linker component. Clinical validation of Nectin-4 as a target in urothelial cancer has been demonstrated with EV. In 2021, the FDA-approved EV for patients with locally advanced or metastatic urothelial cancer who had previously received a PD-1 or PD-L1 inhibitor and platinum-containing chemotherapy or were ineligible for cisplatin containing chemotherapy and had received one or more prior lines of therapy. Bicycle toxin conjugates (BTC) are structurally constrained bicyclic peptides conjugated through cleavable linkers to a toxin. Various, well-described linker toxin combinations can be incorporated into the BTC molecule (e.g., BT1718 and BT5528; refs.). They are of low molecular weight (approximately 4-4.5 kDa) and being chemically synthesized can be optimized for appropriate affinity, stability, and solubility relatively simply. Through intravenous administration, high systemic Cmax values can be attained, which, along with BTCs relatively small size, helps drive rapid diffusion into extra-vascular compartment, as reflected in a volume of distribution similar to extracellular fluid. We believe that delivery of a high number of BTCs, each carrying a reduced toxin load (peptide toxin ratio of 1:1) should improve tumor penetration and reduce the impact of the binding site barrier. BTCs show moderate clearance from the systemic vasculature, predominantly by the renal route. This overall profile marks them out from most ADCs and provides the possibility of enhanced clinical efficacy with a wider therapeutic index. BTCs are new therapeutic modality that shows a very different pharmacokinetic and structural profile to classic ADCs, whereas possessing robust tumoricidal properties. BTCs targeting MT1-MMP and EphA2 are currently in clinical trials. BT8009 is the most recent BTC to enter clinical trial. BT8009 is highly selective for Nectin-4 over other nectin family members and an extensive range of cell membrane expressed proteins. It shares the same cleavable linker and toxin combination as EV and can be cleaved by proteases in the TME, releasing cell penetrant MMAE that diffuses into tumor cells, or bystander stromal-supportive cells, evoking cell death, and tumor regression. It shows a robust and dose-dependent antitumor activity in multiple CDX and PDX models, representing lung, breast, bladder, head and neck cancers. Optimal tumor regression is associated with membrane expression of Nectin-4, in conjunction with MMAE sensitivity. Tumor regression rates are comparable for small and large tumors, indicative of deep and rapid penetration throughout the tumor. BT8009 provides tumoricidal activity with several regimens, potentially allowing for clinical titration of dose and dose interval if required.
Click to Show/Hide
|
||||
| Description |
BT8009 demonstrated dose-related effects on tumor growth in CDX and PDX models over the range 1-3 mg/kg when administered qw. Full tumor regressions were routinely achieved in both models with 3 mg/kg, with no tumor regrowth in subsequent weeks off treatment. Stable disease was delivered by 2 mg/kg and at this dose, in the PDX model, tumor growth resumed after drug cessation. In both models animals from the vehicle treated group were treated with 3 mg/kg BT8009 when tumors reached approximately 800 mm3or 1,000 mm3. Profound tumor regression was rapidly initiated in response. In the majority of studies 3 mg/kg, qw, was adopted as the standard dosing regimen.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice non-small cell lung cell-derived xenograft model. | ||||
| Half life period | 1-2 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
60%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 1 mg/kg | ||||
| MOA of PDC |
The cell adhesion molecule Nectin-4 shows elevated expression in multiple tumor types correlated with poor prognosis. Nectin-4-directed ADCs show efficacy in multiple xenograft tumor models. The ADC enfortumab vedotin (EV) delivers the antimitotic toxin MMAE to Nectin-4-expressing cells via internalization and cleavage of a valine-citrulline dipeptide linker component. Clinical validation of Nectin-4 as a target in urothelial cancer has been demonstrated with EV. In 2021, the FDA-approved EV for patients with locally advanced or metastatic urothelial cancer who had previously received a PD-1 or PD-L1 inhibitor and platinum-containing chemotherapy or were ineligible for cisplatin containing chemotherapy and had received one or more prior lines of therapy. Bicycle toxin conjugates (BTC) are structurally constrained bicyclic peptides conjugated through cleavable linkers to a toxin. Various, well-described linker toxin combinations can be incorporated into the BTC molecule (e.g., BT1718 and BT5528; refs.). They are of low molecular weight (approximately 4-4.5 kDa) and being chemically synthesized can be optimized for appropriate affinity, stability, and solubility relatively simply. Through intravenous administration, high systemic Cmax values can be attained, which, along with BTCs relatively small size, helps drive rapid diffusion into extra-vascular compartment, as reflected in a volume of distribution similar to extracellular fluid. We believe that delivery of a high number of BTCs, each carrying a reduced toxin load (peptide toxin ratio of 1:1) should improve tumor penetration and reduce the impact of the binding site barrier. BTCs show moderate clearance from the systemic vasculature, predominantly by the renal route. This overall profile marks them out from most ADCs and provides the possibility of enhanced clinical efficacy with a wider therapeutic index. BTCs are new therapeutic modality that shows a very different pharmacokinetic and structural profile to classic ADCs, whereas possessing robust tumoricidal properties. BTCs targeting MT1-MMP and EphA2 are currently in clinical trials. BT8009 is the most recent BTC to enter clinical trial. BT8009 is highly selective for Nectin-4 over other nectin family members and an extensive range of cell membrane expressed proteins. It shares the same cleavable linker and toxin combination as EV and can be cleaved by proteases in the TME, releasing cell penetrant MMAE that diffuses into tumor cells, or bystander stromal-supportive cells, evoking cell death, and tumor regression. It shows a robust and dose-dependent antitumor activity in multiple CDX and PDX models, representing lung, breast, bladder, head and neck cancers. Optimal tumor regression is associated with membrane expression of Nectin-4, in conjunction with MMAE sensitivity. Tumor regression rates are comparable for small and large tumors, indicative of deep and rapid penetration throughout the tumor. BT8009 provides tumoricidal activity with several regimens, potentially allowing for clinical titration of dose and dose interval if required.
Click to Show/Hide
|
||||
| Description |
BT8009 demonstrated dose-related effects on tumor growth in CDX and PDX models over the range 1-3 mg/kg when administered qw. Full tumor regressions were routinely achieved in both models with 3 mg/kg, with no tumor regrowth in subsequent weeks off treatment. Stable disease was delivered by 2 mg/kg and at this dose, in the PDX model, tumor growth resumed after drug cessation. In both models animals from the vehicle treated group were treated with 3 mg/kg BT8009 when tumors reached approximately 800 mm3or 1,000 mm3. Profound tumor regression was rapidly initiated in response. In the majority of studies 3 mg/kg, qw, was adopted as the standard dosing regimen.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice triple-negative breast cancer cell-derived xenograft model. | ||||
| Half life period | 1-2 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
74%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 2 mg/kg | ||||
| MOA of PDC |
The cell adhesion molecule Nectin-4 shows elevated expression in multiple tumor types correlated with poor prognosis. Nectin-4-directed ADCs show efficacy in multiple xenograft tumor models. The ADC enfortumab vedotin (EV) delivers the antimitotic toxin MMAE to Nectin-4-expressing cells via internalization and cleavage of a valine-citrulline dipeptide linker component. Clinical validation of Nectin-4 as a target in urothelial cancer has been demonstrated with EV. In 2021, the FDA-approved EV for patients with locally advanced or metastatic urothelial cancer who had previously received a PD-1 or PD-L1 inhibitor and platinum-containing chemotherapy or were ineligible for cisplatin containing chemotherapy and had received one or more prior lines of therapy. Bicycle toxin conjugates (BTC) are structurally constrained bicyclic peptides conjugated through cleavable linkers to a toxin. Various, well-described linker toxin combinations can be incorporated into the BTC molecule (e.g., BT1718 and BT5528; refs.). They are of low molecular weight (approximately 4-4.5 kDa) and being chemically synthesized can be optimized for appropriate affinity, stability, and solubility relatively simply. Through intravenous administration, high systemic Cmax values can be attained, which, along with BTCs relatively small size, helps drive rapid diffusion into extra-vascular compartment, as reflected in a volume of distribution similar to extracellular fluid. We believe that delivery of a high number of BTCs, each carrying a reduced toxin load (peptide toxin ratio of 1:1) should improve tumor penetration and reduce the impact of the binding site barrier. BTCs show moderate clearance from the systemic vasculature, predominantly by the renal route. This overall profile marks them out from most ADCs and provides the possibility of enhanced clinical efficacy with a wider therapeutic index. BTCs are new therapeutic modality that shows a very different pharmacokinetic and structural profile to classic ADCs, whereas possessing robust tumoricidal properties. BTCs targeting MT1-MMP and EphA2 are currently in clinical trials. BT8009 is the most recent BTC to enter clinical trial. BT8009 is highly selective for Nectin-4 over other nectin family members and an extensive range of cell membrane expressed proteins. It shares the same cleavable linker and toxin combination as EV and can be cleaved by proteases in the TME, releasing cell penetrant MMAE that diffuses into tumor cells, or bystander stromal-supportive cells, evoking cell death, and tumor regression. It shows a robust and dose-dependent antitumor activity in multiple CDX and PDX models, representing lung, breast, bladder, head and neck cancers. Optimal tumor regression is associated with membrane expression of Nectin-4, in conjunction with MMAE sensitivity. Tumor regression rates are comparable for small and large tumors, indicative of deep and rapid penetration throughout the tumor. BT8009 provides tumoricidal activity with several regimens, potentially allowing for clinical titration of dose and dose interval if required.
Click to Show/Hide
|
||||
| Description |
BT8009 demonstrated dose-related effects on tumor growth in CDX and PDX models over the range 1-3 mg/kg when administered qw. Full tumor regressions were routinely achieved in both models with 3 mg/kg, with no tumor regrowth in subsequent weeks off treatment. Stable disease was delivered by 2 mg/kg and at this dose, in the PDX model, tumor growth resumed after drug cessation. In both models animals from the vehicle treated group were treated with 3 mg/kg BT8009 when tumors reached approximately 800 mm3or 1,000 mm3. Profound tumor regression was rapidly initiated in response. In the majority of studies 3 mg/kg, qw, was adopted as the standard dosing regimen.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice triple-negative breast cancer cell-derived xenograft model. | ||||
| Half life period | 1-2 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
77%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 2 mg/kg | ||||
| MOA of PDC |
The cell adhesion molecule Nectin-4 shows elevated expression in multiple tumor types correlated with poor prognosis. Nectin-4-directed ADCs show efficacy in multiple xenograft tumor models. The ADC enfortumab vedotin (EV) delivers the antimitotic toxin MMAE to Nectin-4-expressing cells via internalization and cleavage of a valine-citrulline dipeptide linker component. Clinical validation of Nectin-4 as a target in urothelial cancer has been demonstrated with EV. In 2021, the FDA-approved EV for patients with locally advanced or metastatic urothelial cancer who had previously received a PD-1 or PD-L1 inhibitor and platinum-containing chemotherapy or were ineligible for cisplatin containing chemotherapy and had received one or more prior lines of therapy. Bicycle toxin conjugates (BTC) are structurally constrained bicyclic peptides conjugated through cleavable linkers to a toxin. Various, well-described linker toxin combinations can be incorporated into the BTC molecule (e.g., BT1718 and BT5528; refs.). They are of low molecular weight (approximately 4-4.5 kDa) and being chemically synthesized can be optimized for appropriate affinity, stability, and solubility relatively simply. Through intravenous administration, high systemic Cmax values can be attained, which, along with BTCs relatively small size, helps drive rapid diffusion into extra-vascular compartment, as reflected in a volume of distribution similar to extracellular fluid. We believe that delivery of a high number of BTCs, each carrying a reduced toxin load (peptide toxin ratio of 1:1) should improve tumor penetration and reduce the impact of the binding site barrier. BTCs show moderate clearance from the systemic vasculature, predominantly by the renal route. This overall profile marks them out from most ADCs and provides the possibility of enhanced clinical efficacy with a wider therapeutic index. BTCs are new therapeutic modality that shows a very different pharmacokinetic and structural profile to classic ADCs, whereas possessing robust tumoricidal properties. BTCs targeting MT1-MMP and EphA2 are currently in clinical trials. BT8009 is the most recent BTC to enter clinical trial. BT8009 is highly selective for Nectin-4 over other nectin family members and an extensive range of cell membrane expressed proteins. It shares the same cleavable linker and toxin combination as EV and can be cleaved by proteases in the TME, releasing cell penetrant MMAE that diffuses into tumor cells, or bystander stromal-supportive cells, evoking cell death, and tumor regression. It shows a robust and dose-dependent antitumor activity in multiple CDX and PDX models, representing lung, breast, bladder, head and neck cancers. Optimal tumor regression is associated with membrane expression of Nectin-4, in conjunction with MMAE sensitivity. Tumor regression rates are comparable for small and large tumors, indicative of deep and rapid penetration throughout the tumor. BT8009 provides tumoricidal activity with several regimens, potentially allowing for clinical titration of dose and dose interval if required.
Click to Show/Hide
|
||||
| Description |
BT8009 demonstrated dose-related effects on tumor growth in CDX and PDX models over the range 1-3 mg/kg when administered qw. Full tumor regressions were routinely achieved in both models with 3 mg/kg, with no tumor regrowth in subsequent weeks off treatment. Stable disease was delivered by 2 mg/kg and at this dose, in the PDX model, tumor growth resumed after drug cessation. In both models animals from the vehicle treated group were treated with 3 mg/kg BT8009 when tumors reached approximately 800 mm3or 1,000 mm3. Profound tumor regression was rapidly initiated in response. In the majority of studies 3 mg/kg, qw, was adopted as the standard dosing regimen.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice non-small cell lung cell-derived xenograft model. | ||||
| Half life period | 1-2 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
96%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 3 mg/kg | ||||
| MOA of PDC |
The cell adhesion molecule Nectin-4 shows elevated expression in multiple tumor types correlated with poor prognosis. Nectin-4-directed ADCs show efficacy in multiple xenograft tumor models. The ADC enfortumab vedotin (EV) delivers the antimitotic toxin MMAE to Nectin-4-expressing cells via internalization and cleavage of a valine-citrulline dipeptide linker component. Clinical validation of Nectin-4 as a target in urothelial cancer has been demonstrated with EV. In 2021, the FDA-approved EV for patients with locally advanced or metastatic urothelial cancer who had previously received a PD-1 or PD-L1 inhibitor and platinum-containing chemotherapy or were ineligible for cisplatin containing chemotherapy and had received one or more prior lines of therapy. Bicycle toxin conjugates (BTC) are structurally constrained bicyclic peptides conjugated through cleavable linkers to a toxin. Various, well-described linker toxin combinations can be incorporated into the BTC molecule (e.g., BT1718 and BT5528; refs.). They are of low molecular weight (approximately 4-4.5 kDa) and being chemically synthesized can be optimized for appropriate affinity, stability, and solubility relatively simply. Through intravenous administration, high systemic Cmax values can be attained, which, along with BTCs relatively small size, helps drive rapid diffusion into extra-vascular compartment, as reflected in a volume of distribution similar to extracellular fluid. We believe that delivery of a high number of BTCs, each carrying a reduced toxin load (peptide toxin ratio of 1:1) should improve tumor penetration and reduce the impact of the binding site barrier. BTCs show moderate clearance from the systemic vasculature, predominantly by the renal route. This overall profile marks them out from most ADCs and provides the possibility of enhanced clinical efficacy with a wider therapeutic index. BTCs are new therapeutic modality that shows a very different pharmacokinetic and structural profile to classic ADCs, whereas possessing robust tumoricidal properties. BTCs targeting MT1-MMP and EphA2 are currently in clinical trials. BT8009 is the most recent BTC to enter clinical trial. BT8009 is highly selective for Nectin-4 over other nectin family members and an extensive range of cell membrane expressed proteins. It shares the same cleavable linker and toxin combination as EV and can be cleaved by proteases in the TME, releasing cell penetrant MMAE that diffuses into tumor cells, or bystander stromal-supportive cells, evoking cell death, and tumor regression. It shows a robust and dose-dependent antitumor activity in multiple CDX and PDX models, representing lung, breast, bladder, head and neck cancers. Optimal tumor regression is associated with membrane expression of Nectin-4, in conjunction with MMAE sensitivity. Tumor regression rates are comparable for small and large tumors, indicative of deep and rapid penetration throughout the tumor. BT8009 provides tumoricidal activity with several regimens, potentially allowing for clinical titration of dose and dose interval if required.
Click to Show/Hide
|
||||
| Description |
BT8009 demonstrated dose-related effects on tumor growth in CDX and PDX models over the range 1-3 mg/kg when administered qw. Full tumor regressions were routinely achieved in both models with 3 mg/kg, with no tumor regrowth in subsequent weeks off treatment. Stable disease was delivered by 2 mg/kg and at this dose, in the PDX model, tumor growth resumed after drug cessation. In both models animals from the vehicle treated group were treated with 3 mg/kg BT8009 when tumors reached approximately 800 mm3or 1,000 mm3. Profound tumor regression was rapidly initiated in response. In the majority of studies 3 mg/kg, qw, was adopted as the standard dosing regimen.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice triple-negative breast cancer cell-derived xenograft model. | ||||
| Half life period | 1-2 h | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
98%
|
|||
| Administration Time | 28 days | ||||
| Administration Dosage | 3 mg/kg | ||||
| MOA of PDC |
The cell adhesion molecule Nectin-4 shows elevated expression in multiple tumor types correlated with poor prognosis. Nectin-4-directed ADCs show efficacy in multiple xenograft tumor models. The ADC enfortumab vedotin (EV) delivers the antimitotic toxin MMAE to Nectin-4-expressing cells via internalization and cleavage of a valine-citrulline dipeptide linker component. Clinical validation of Nectin-4 as a target in urothelial cancer has been demonstrated with EV. In 2021, the FDA-approved EV for patients with locally advanced or metastatic urothelial cancer who had previously received a PD-1 or PD-L1 inhibitor and platinum-containing chemotherapy or were ineligible for cisplatin containing chemotherapy and had received one or more prior lines of therapy. Bicycle toxin conjugates (BTC) are structurally constrained bicyclic peptides conjugated through cleavable linkers to a toxin. Various, well-described linker toxin combinations can be incorporated into the BTC molecule (e.g., BT1718 and BT5528; refs.). They are of low molecular weight (approximately 4-4.5 kDa) and being chemically synthesized can be optimized for appropriate affinity, stability, and solubility relatively simply. Through intravenous administration, high systemic Cmax values can be attained, which, along with BTCs relatively small size, helps drive rapid diffusion into extra-vascular compartment, as reflected in a volume of distribution similar to extracellular fluid. We believe that delivery of a high number of BTCs, each carrying a reduced toxin load (peptide toxin ratio of 1:1) should improve tumor penetration and reduce the impact of the binding site barrier. BTCs show moderate clearance from the systemic vasculature, predominantly by the renal route. This overall profile marks them out from most ADCs and provides the possibility of enhanced clinical efficacy with a wider therapeutic index. BTCs are new therapeutic modality that shows a very different pharmacokinetic and structural profile to classic ADCs, whereas possessing robust tumoricidal properties. BTCs targeting MT1-MMP and EphA2 are currently in clinical trials. BT8009 is the most recent BTC to enter clinical trial. BT8009 is highly selective for Nectin-4 over other nectin family members and an extensive range of cell membrane expressed proteins. It shares the same cleavable linker and toxin combination as EV and can be cleaved by proteases in the TME, releasing cell penetrant MMAE that diffuses into tumor cells, or bystander stromal-supportive cells, evoking cell death, and tumor regression. It shows a robust and dose-dependent antitumor activity in multiple CDX and PDX models, representing lung, breast, bladder, head and neck cancers. Optimal tumor regression is associated with membrane expression of Nectin-4, in conjunction with MMAE sensitivity. Tumor regression rates are comparable for small and large tumors, indicative of deep and rapid penetration throughout the tumor. BT8009 provides tumoricidal activity with several regimens, potentially allowing for clinical titration of dose and dose interval if required.
Click to Show/Hide
|
||||
| Description |
BT8009 demonstrated dose-related effects on tumor growth in CDX and PDX models over the range 1-3 mg/kg when administered qw. Full tumor regressions were routinely achieved in both models with 3 mg/kg, with no tumor regrowth in subsequent weeks off treatment. Stable disease was delivered by 2 mg/kg and at this dose, in the PDX model, tumor growth resumed after drug cessation. In both models animals from the vehicle treated group were treated with 3 mg/kg BT8009 when tumors reached approximately 800 mm3or 1,000 mm3. Profound tumor regression was rapidly initiated in response. In the majority of studies 3 mg/kg, qw, was adopted as the standard dosing regimen.
Click to Show/Hide
|
||||
| In Vivo Model | BALB/c nude mice non-small cell lung cell-derived xenograft model. | ||||
| Half life period | 1-2 h | ||||
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
81%
|
|||
| Administration Time | 21 days | ||||
| Administration Dosage | 3 mg/kg qw | ||||
| MOA of PDC |
Nectin-4 is a cell adhesion molecule from the Nectin and Nectin-like family. It is a clinically validated tumor target and has been reported to be highly expressed in a wide range of solid tumors, including bladder, esophageal, pancreatic, and lung, but with limited distribution in healthy tissues.An antibody drug conjugate (PADCEV, enfortumab vedotin) that targets Nectin-4 was recently approved for the treatment of bladder cancer following the generation of positive data in a number of clinical studies. Herein we describe the discovery via phage display and subsequent chemical optimization of a Nectin-4 binding Bicycle and its incorporation into BT8009, a BTC that is currently under clinical evaluation. A detailed report of the pharmacologic properties of BT8009 has recently been described.In this, BT8009 shows potent efficacy in multiple tumor models, including patient-derived xenografts, across a variety of tumor indications and is well-tolerated in preclinical safety studies. In several models it demonstrated superior or equivalent efficacy to an analogue of the ADC PADCEV.
Click to Show/Hide
|
||||
| Description |
The efficacy of BT8009 was evaluated in a cell-derived xenograft (CDX) model using breast adenocarcinoma (MDA-MB-468) cells, which express Nectin-4.When tested at a dose of 3 mg/kg once weekly, significant antitumor activity was observed. At a dose of 3 mg/kg twice weekly or 5 mg/kg once weekly, potent efficacy was achieved with almost complete regression of the tumor after 18 days. Importantly, following cessation of dosing after 18 days, animals from the 5 mg/kg once weekly dosing group were monitored up to day 42, and no tumor regrowth was observed. Consistent animal body weights throughout the study indicate that BT8009 appeared to be well-tolerated at all doses tested. In additional studies reported elsewhere, BT8009 has shown preclinical efficacy in a wide range of CDX and PDX tumor types with full tumor regression seen in small and large tumors, where efficacy broadly correlates with Nectin-4 expression. Additionally, when BT8009 was codosed with an excess of an MMAE-free analogue, efficacy was attenuated, and a BTC incorporating a nonbinding Bicycle analogue showed a demonstrably lower rate and degree of tumor regression.
Click to Show/Hide
|
||||
| In Vivo Model | MDA-MB-468 cell-derived xenograft (CDX) model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
93%
|
|||
| Administration Time | 21 days | ||||
| Administration Dosage | 3 mg/kg biw | ||||
| MOA of PDC |
Nectin-4 is a cell adhesion molecule from the Nectin and Nectin-like family. It is a clinically validated tumor target and has been reported to be highly expressed in a wide range of solid tumors, including bladder, esophageal, pancreatic, and lung, but with limited distribution in healthy tissues.An antibody drug conjugate (PADCEV, enfortumab vedotin) that targets Nectin-4 was recently approved for the treatment of bladder cancer following the generation of positive data in a number of clinical studies. Herein we describe the discovery via phage display and subsequent chemical optimization of a Nectin-4 binding Bicycle and its incorporation into BT8009, a BTC that is currently under clinical evaluation. A detailed report of the pharmacologic properties of BT8009 has recently been described.In this, BT8009 shows potent efficacy in multiple tumor models, including patient-derived xenografts, across a variety of tumor indications and is well-tolerated in preclinical safety studies. In several models it demonstrated superior or equivalent efficacy to an analogue of the ADC PADCEV.
Click to Show/Hide
|
||||
| Description |
The efficacy of BT8009 was evaluated in a cell-derived xenograft (CDX) model using breast adenocarcinoma (MDA-MB-468) cells, which express Nectin-4.When tested at a dose of 3 mg/kg once weekly, significant antitumor activity was observed. At a dose of 3 mg/kg twice weekly or 5 mg/kg once weekly, potent efficacy was achieved with almost complete regression of the tumor after 18 days. Importantly, following cessation of dosing after 18 days, animals from the 5 mg/kg once weekly dosing group were monitored up to day 42, and no tumor regrowth was observed. Consistent animal body weights throughout the study indicate that BT8009 appeared to be well-tolerated at all doses tested. In additional studies reported elsewhere, BT8009 has shown preclinical efficacy in a wide range of CDX and PDX tumor types with full tumor regression seen in small and large tumors, where efficacy broadly correlates with Nectin-4 expression. Additionally, when BT8009 was codosed with an excess of an MMAE-free analogue, efficacy was attenuated, and a BTC incorporating a nonbinding Bicycle analogue showed a demonstrably lower rate and degree of tumor regression.
Click to Show/Hide
|
||||
| In Vivo Model | MDA-MB-468 cell-derived xenograft (CDX) model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
93%
|
|||
| Administration Time | 21 days | ||||
| Administration Dosage | 5 mg/kg qw | ||||
| MOA of PDC |
Nectin-4 is a cell adhesion molecule from the Nectin and Nectin-like family. It is a clinically validated tumor target and has been reported to be highly expressed in a wide range of solid tumors, including bladder, esophageal, pancreatic, and lung, but with limited distribution in healthy tissues.An antibody drug conjugate (PADCEV, enfortumab vedotin) that targets Nectin-4 was recently approved for the treatment of bladder cancer following the generation of positive data in a number of clinical studies. Herein we describe the discovery via phage display and subsequent chemical optimization of a Nectin-4 binding Bicycle and its incorporation into BT8009, a BTC that is currently under clinical evaluation. A detailed report of the pharmacologic properties of BT8009 has recently been described.In this, BT8009 shows potent efficacy in multiple tumor models, including patient-derived xenografts, across a variety of tumor indications and is well-tolerated in preclinical safety studies. In several models it demonstrated superior or equivalent efficacy to an analogue of the ADC PADCEV.
Click to Show/Hide
|
||||
| Description |
The efficacy of BT8009 was evaluated in a cell-derived xenograft (CDX) model using breast adenocarcinoma (MDA-MB-468) cells, which express Nectin-4.When tested at a dose of 3 mg/kg once weekly, significant antitumor activity was observed. At a dose of 3 mg/kg twice weekly or 5 mg/kg once weekly, potent efficacy was achieved with almost complete regression of the tumor after 18 days. Importantly, following cessation of dosing after 18 days, animals from the 5 mg/kg once weekly dosing group were monitored up to day 42, and no tumor regrowth was observed. Consistent animal body weights throughout the study indicate that BT8009 appeared to be well-tolerated at all doses tested. In additional studies reported elsewhere, BT8009 has shown preclinical efficacy in a wide range of CDX and PDX tumor types with full tumor regression seen in small and large tumors, where efficacy broadly correlates with Nectin-4 expression. Additionally, when BT8009 was codosed with an excess of an MMAE-free analogue, efficacy was attenuated, and a BTC incorporating a nonbinding Bicycle analogue showed a demonstrably lower rate and degree of tumor regression.
Click to Show/Hide
|
||||
| In Vivo Model | MDA-MB-468 cell-derived xenograft (CDX) model. | ||||
| In Vitro Model | Breast adenocarcinoma | MDA-MB-468 cell | CVCL_0419 | ||
References
