Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00175
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Unique variant of pHLIP
|
|||||
| Structure |
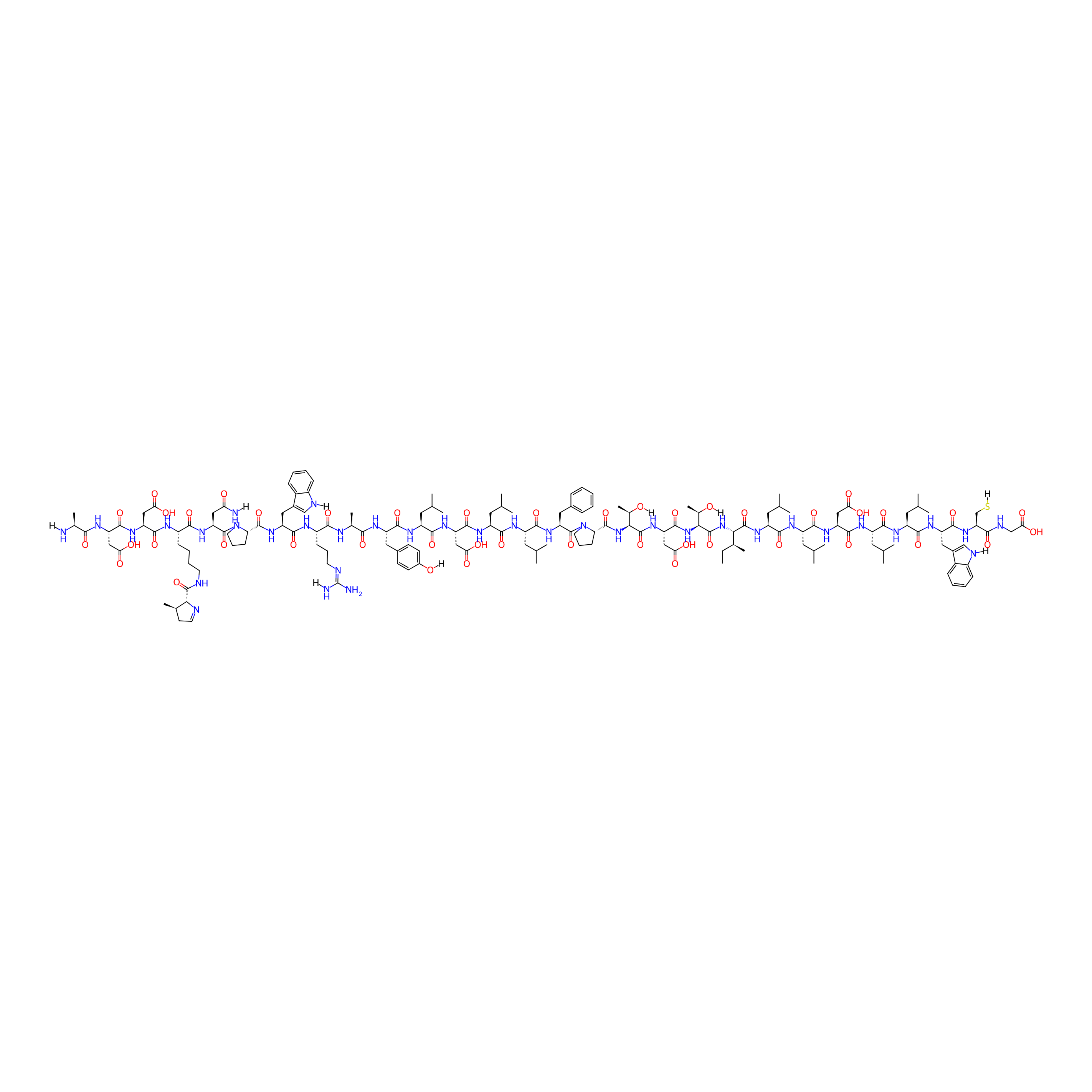
|
|||||
| Sequence |
ADDONPWRAYLDLLFPTDTILLDLLWCG
|
|||||
| Peptide Type |
Linear
|
|||||
| Receptor Name |
DNA topoisomerase 1 (TOP1)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Distribution |
Acidity is produced not only by cancerous tissue, but also by any tissue that is experiencing hypoxia: the lack of oxygen in the tissue triggers cells to switch to a high rate of the anaerobic, glycolytic energy production pathway, consequently resulting in acidification. pHLIPs have been shown to target infections in lungs, inflammatory arthritis, and ischemic myocardium, a consequence of heart disease, and could be used for diagnosis and treatment thereof. It is also possible that pHLIPs could be used as targeted treatment in ischemic strokes. In contrast to diseased tissue, healthy tissue is typically not associated with increased acidity except in the gastrointestinal tract and kidney, the pHs of which might be regulated by the implementation of a special diet or supplementary drinks if their acidity proves problematic.
|
|||||
| Formula |
C159H236N36O44S
|
|||||
| Isosmiles |
[H]NC(=O)C[C@H](NC(=O)[C@H](CCCCNC(=O)[C@@H]1N=CC[C@H]1C)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](C)N[H])C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1cn([H])c2ccccc12)C(=O)N[C@@H](CCC/N=C(/N)N[H])C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1ccc(O[H])cc1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@]([H])(C(=O)N[C@@H](CC(=O)O)C(=O)N[C@]([H])(C(=O)N[C@]([H])(C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1cn([H])c2ccccc12)C(=O)N[C@@H](CS[H])C(=O)NCC(=O)O)[C@@H](C)CC)[C@@H](C)O[H])[C@@H](C)O[H]
|
|||||
| InChI |
InChI=1S/C159H236N36O44S/c1-22-83(16)128(154(235)186-106(61-82(14)15)141(222)176-103(58-79(8)9)139(220)182-112(69-123(204)205)148(229)179-100(55-76(2)3)136(217)175-102(57-78(6)7)138(219)181-108(64-91-72-167-96-39-28-26-37-94(91)96)145(226)190-117(75-240)133(214)169-74-126(210)211)191-156(237)130(88(21)197)192-150(231)114(71-125(208)209)187-155(236)129(87(20)196)193-152(233)119-44-34-54-195(119)157(238)115(63-89-35-24-23-25-36-89)188-142(223)105(60-81(12)13)177-137(218)101(56-77(4)5)180-149(230)113(70-124(206)207)183-140(221)104(59-80(10)11)178-143(224)107(62-90-45-47-93(198)48-46-90)174-132(213)86(19)170-134(215)99(42-32-51-166-159(162)163)172-144(225)109(65-92-73-168-97-40-29-27-38-95(92)97)185-151(232)118-43-33-53-194(118)158(239)116(66-120(161)199)189-135(216)98(41-30-31-50-165-153(234)127-84(17)49-52-164-127)171-146(227)111(68-122(202)203)184-147(228)110(67-121(200)201)173-131(212)85(18)160/h23-29,35-40,45-48,52,72-73,76-88,98-119,127-130,167-168,196-198,240H,22,30-34,41-44,49-51,53-71,74-75,160H2,1-21H3,(H2,161,199)(H,165,234)(H,169,214)(H,170,215)(H,171,227)(H,172,225)(H,173,212)(H,174,213)(H,175,217)(H,176,222)(H,177,218)(H,178,224)(H,179,229)(H,180,230)(H,181,219)(H,182,220)(H,183,221)(H,184,228)(H,185,232)(H,186,235)(H,187,236)(H,188,223)(H,189,216)(H,190,226)(H,191,237)(H,192,231)(H,193,233)(H,200,201)(H,202,203)(H,204,205)(H,206,207)(H,208,209)(H,210,211)(H4,162,163,166)/t83-,84+,85-,86-,87+,88+,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,127+,128-,129-,130-/m0/s1
|
|||||
| InChIKey |
UUNZFLYWPMXOIH-XGYFDBJRSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
3387.912
|
Polar area
|
1259.16
|
||
|
Complexity
|
3385.705686
|
xlogp Value
|
-5.0611
|
|||
|
Heavy Count
|
240
|
Rot Bonds
|
107
|
|||
|
Hbond acc
|
42
|
Hbond Donor
|
42
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
CBX-12 [Phase 2]
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
58.10%
|
|||
| Administration Time | 23 days | ||||
| Administration Dosage | 5 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
76.50%
|
|||
| Administration Time | 35 days | ||||
| Administration Dosage | 10 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
81.40%
|
|||
| Administration Time | 23 days | ||||
| Administration Dosage | 5 mg/kg with ceralasertib 25 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
95%
|
|||
| Administration Time | 35 days | ||||
| Administration Dosage | 10 mg/kg with ceralasertib 25 mg/kg | ||||
| Evaluation Method | Tumor volume detection | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate |
60 mm3
|
|||
| Administration Time | 45 days | ||||
| Administration Dosage | 5 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate |
75 mm3
|
|||
| Administration Time | 42 days | ||||
| Administration Dosage | 10 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate |
100 mm3
|
|||
| Administration Time | 42 days | ||||
| Administration Dosage | 10 mg/kg with ceralasertib 25 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | MDA-MB-231 xenografts | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Survival rate |
100 mm3
|
|||
| Administration Time | 45 days | ||||
| Administration Dosage | 5 mg/kg with ceralasertib 25 mg/kg | ||||
| Description |
Combination treatment significantly inhibited tumor growth without significant toxicity in both mouse xenografts compared with CBX-12 and ceralasertib monotherapy without significant toxicity.
|
||||
| In Vivo Model | HCT-116 xenografts | ||||
References
