Drug Information
General Information of This Drug
| Drug ID | DRG00001 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Brimonidine | |||||
| Synonyms |
brimonidine; 59803-98-4; UK 14,304; Bromoxidine; 5-bromo-N-(4,5-dihydro-1H-imidazol-2-yl)quinoxalin-6-amine; 5-Bromo-N-(4,5-dihydro-1H-imidazol-2-yl)-6-quinoxalinamine; brimonidinum; 5-Bromo-6-(2-imidazolin-2-ylamino)quinoxaline; UK-14304; 6-Quinoxalinamine, 5-bromo-N-(4,5-dihydro-1H-imidazol-2-yl)-; brimonidina; AGN 190342; E6GNX3HHTE; UNII-E6GNX3HHTE; UK 14304; NSC-318825; BRN 0751629; CHEBI:3175; MLS000069370; AGN-190342 FREE BASE; LK 14304-18; UK-1430418 FREE BASE; MFCD00153878; CHEMBL844; SMR000058355; DTXSID3045221; Brimonidine (INN); NSC 318825; NCGC00016069-09; UK14,304; UK 14304;AGN190342; BRIMONIDINE [INN]; UK 14304 (tartrate);AGN190342 (tartrate); 5-Bromo-N-(2-imidazolin-2-yl)-6-quinoxalinamine; DTXCID1025221; Brimonidine [INN:BAN]; 6-Quinoxalinamine, 5-bromo-N-(4,5-dihydro-1H-imidazol-2-yl)-, (S-(R*,R*))-2,3-dihydroxybutanedioate (1:1); CAS-59803-98-4; SR-01000000023; AGN-190342; Brimodine; 5-bromo-6-(imidazolidinylideneamino)quinoxaline; UK14304; Tocris-0425; [3H]-UK14304; Opera_ID_612; Lopac-U-104; BRIMONIDINE [MI]; brimonidine (UK14304); BRIMONIDINE [VANDF]; cid_2435; Lopac0_001216; SCHEMBL24670; BRIMONIDINE [WHO-DD]; GTPL520; MLS001076349; BIDD:GT0649; GTPL5386; BDBM34572; [3H]brimonidine (UK14304); D11AX21; S01EA05; XYLJNLCSTIOKRM-UHFFFAOYSA-N; HMS3259P09; HMS3263D14; HMS3266O03; HMS3411K05; HMS3675K05; HMS3887K07; AMY22318; BCP12632; EX-A5415; HY-B0659; Tox21_110299; Tox21_501216; AC-162; NSC318825; PDSP1_000640; PDSP2_000635; s9508; AKOS005267239; Brimonidine;AGN190342;UK14304; Tox21_110299_1; CCG-205290; DB00484; GS-3236; LP01216; NC00638; SDCCGSBI-0051183.P002; MRF-0000657; NCGC00016069-01; NCGC00016069-02; NCGC00016069-03; NCGC00016069-04; NCGC00016069-05; NCGC00016069-06; NCGC00016069-07; NCGC00016069-08; NCGC00016069-10; NCGC00016069-11; NCGC00016069-12; NCGC00016069-13; NCGC00016069-24; NCGC00023468-02; NCGC00023468-04; NCGC00023468-05; NCGC00023468-06; NCGC00023468-07; NCGC00261901-01; SY053060; Brimonidine 100 microg/mL in Acetonitrile; B4132; EU-0101216; FT-0650586; NS00006954; EN300-50880; UK 14304-18; C07886; C75796; D07540; 5-bromo-6-(2-imidazolidinylidenamino)quinoxaline; 5-bromo-6-(2-imidazolin-2-ylamino) quinoxaline; 5-Bromo-6-(2-imidazolin-2-ylamino)-quinoxaline; 6-Quinoxalinamine,5-dihydro-1H-imidazol-2-yl)-; A832477; L000615; Q577377; SR-01000000023-2; SR-01000000023-4; BRD-K68264559-001-10-0; 5-Bromo-N-(4,5-dihydro-2-imidazolyl)quinoxalin-6-amine; Z608061562; (5-Bromo-quinoxalin-6-yl)-(4,5-dihydro-1H-imidazol-2-yl)-amine; 5-bromanyl-N-(4,5-dihydro-1H-imidazol-2-yl)quinoxalin-6-amine; 5-BROMO-6-(2-IMIDAZOLIN-2-YLAMINO)QUINOXALINE D-TARTRATE (1:1).; 6-Quinoxalinamine, 5-bromo-N-(4,5-dihydro-1H-imidazol-2-yl)- (9CI)
Click to Show/Hide
|
|||||
| Target(s) | Alpha-2A adrenergic receptor (ADRA2A) | Target Info | ||||
| Structure |
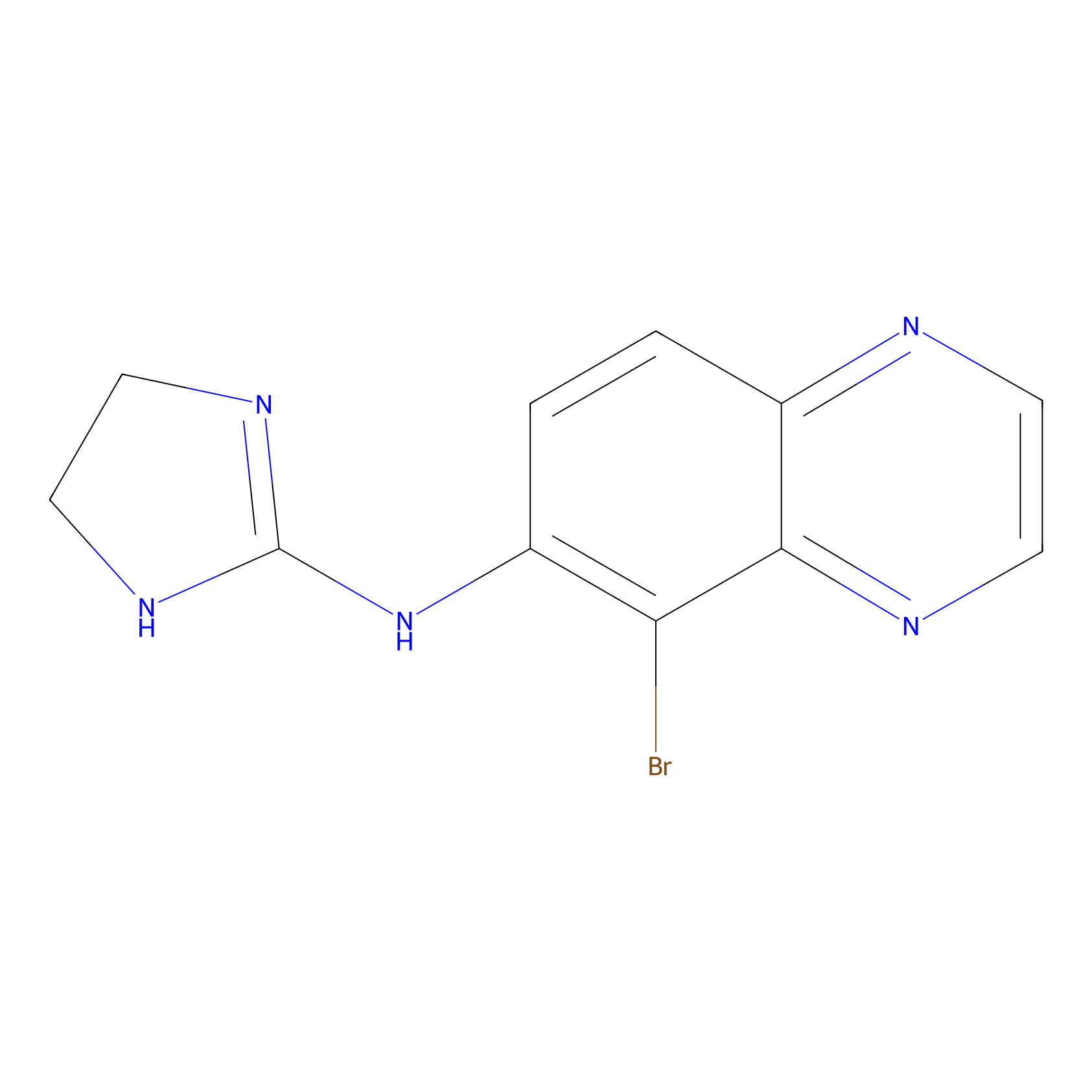
|
|||||
| Formula |
C11H10BrN5
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 292.13 | ||||
| Lipid-water partition coefficient (xlogp) | 0.6 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||
| Rotatable Bond Count (rotbonds) | 2 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C1CN=C(N1)NC2=C(C3=NC=CN=C3C=C2)Br
|
|||||
| InChI |
InChI=1S/C11H10BrN5/c12-9-7(17-11-15-5-6-16-11)1-2-8-10(9)14-4-3-13-8/h1-4H,5-6H2,(H2,15,16,17)
|
|||||
| InChIKey |
XYLJNLCSTIOKRM-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
5-bromo-N-(4,5-dihydro-1H-imidazol-2-yl)quinoxalin-6-amine
|
|||||
The activity data of This Drug
| Standard Type | Value | Administration times | Administration dosage | Vivo model | Ref. | |
|---|---|---|---|---|---|---|
| Duration of lower intraocular pressure | 8h | N.A. | N.A. | Dutch Belted rabbits (23kg) model. | [1] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
HR97-brimonidine [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Glaucoma | ||||
| Efficacy Data | Duration of lower intraocular pressure | 18 Days | |||
| Evaluation Method | Hand-held rebound tonometer icareTONOVET assay | ||||
| MOA of PDC |
Sustained drug delivery strategies have many potential benefits for treating a range of diseases, particularly chronic diseases that require treatment for years. For many chronic ocular diseases, patient adherence to eye drop dosing regimens and the need for frequent intraocular injections are significant barriers to effective disease management. Here, we utilize peptide engineering to impart melanin binding properties to peptide-drug conjugates to act as a sustained-release depot in the eye. We develop a super learning-based methodology to engineer multifunctional peptides that efficiently enter cells, bind to melanin, and have low cytotoxicity. When the lead multifunctional peptide (HR97) is conjugated to brimonidine, an intraocular pressure lowering drug that is prescribed for three times per day topical dosing, intraocular pressure reduction is observed for up to 18 days after a single intracameral injection in rabbits. Further, the cumulative intraocular pressure lowering effect increases ~17-fold compared to free brimonidine injection. Engineered multifunctional peptide-drug conjugates are a promising approach for providing sustained therapeutic delivery in the eye and beyond.
Click to Show/Hide
|
||||
| Description |
The HR97-brimonidine conjugate provided up to 18 days of IOP lowering with a single ICM injection in normotensive rabbits, which contrasts with the 8 h-effect provided by a brimonidine eye drop.
|
||||
| In Vivo Model | Dutch belted rabbits (2-3 kg) model. | ||||
References
