Drug Information
General Information of This Drug
| Drug ID | DRG00015 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Bendamustine | |||||
| Synonyms |
Bendamustine; 16506-27-7; 4-(5-(Bis(2-chloroethyl)amino)-1-methyl-1H-benzo[d]imidazol-2-yl)butanoic acid; Bendamustina; Bendamustinum; Bendamustine [INN]; 4-[5-[bis(2-chloroethyl)amino]-1-methylbenzimidazol-2-yl]butanoic acid; Bendamustine free base; Bendamustinum [Latin]; UNII-9266D9P3PQ; DTXSID2046888; HSDB 7763; 9266D9P3PQ; Treanda (TN); SDX 105; DTXCID0026888; NIOSH/DD6304600; 5-(Bis(2-chloroethyl)amino)-1-methyl-2-benzimidazolebutyric acid; 16506-27-7 (free base); 1H-Benzimidazole-2-butanoic acid, 5-(bis(2-chloroethyl)amino)-1-methyl-; Bendamustine (INN); Bendamustinum (Latin); NCGC00181170-01; 2-Benzimidazolebutyric acid, 5-(bis(2-chloroethyl)amino)-1-methyl-; DD63046000; omega-(1-Methyl-5-bis(beta-chloroethyl)aminobenzimidazole-(2)-butyric acid; bendamustin; Bendamustina [Spanish]; 5-(Bis(2-chloroethyl)amino)-1-methyl-1H-benzimidazole-2-butanoic acid; 4-(5-(BIS(2-CHLOROETHYL)AMINO)-1-METHYL-1H-BENZIMIDAZOLE-2-YL)BUTANOIC ACID; 4-{5-[bis(2-chloroethyl)amino]-1-methyl-1H-1,3-benzodiazol-2-yl}butanoic acid; CAS-16506-27-7; Bendamustine [INN:BAN]; gamma-; 1H-benzimidazole-2-butanoic acid, 5-[bis(2-chloroethyl)amino]-1-methyl-; SDX-105 free base; BENDAMUSTINE [MI]; BENDAMUSTINE [HSDB]; BENDAMUSTINE [VANDF]; SCHEMBL26319; BENDAMUSTINE [WHO-DD]; CHEMBL487253; GTPL7478; L01AA09; CHEBI:135515; BDBM173621; BCP04111; Tox21_112771; MFCD00866481; s5939; AKOS022181343; Tox21_112771_1; BCP9000389; DB06769; SDCCGSBI-0633739.P001; NCGC00181170-02; NCGC00181170-03; NCGC00181170-10; AC-22488; AS-73546; HY-13567; BCP0726000100; CS-0007192; FT-0742187; NS00007783; D07501; AB01273966-01; AB01273966-02; AB01273966_03; US9096627, CY190602; EN300-26189677; Q425745; J-010179; BRD-K17068645-003-02-6; 4-{5-[bis(2-chloroethyl)amino]-1-methyl-1H-benzimidazol-2-yl}butanoic acid; Bendamustine; 2-Benzimidazolebutyric acid, 5-[bis(2-chloroethyl)amino]-1-methyl- (8CI); 4-[5-[Bis(2-chloroethyl)amino]-1-methylbenzimidazol-2-yl]butanoic acid; Bendamustine; ?-[1-Methyl-5-[bis(ss-chloroethyl)amino]-2-benzimidazolyl]butyric acid; InChI=1/C16H21Cl2N3O2/c1-20-14-6-5-12(21(9-7-17)10-8-18)11-13(14)19-15(20)3-2-4-16(22)23/h5-6,11H,2-4,7-10H2,1H3,(H,22,23
Click to Show/Hide
|
|||||
| Target(s) | Human Deoxyribonucleic acid (hDNA) | Target Info | ||||
| Structure |
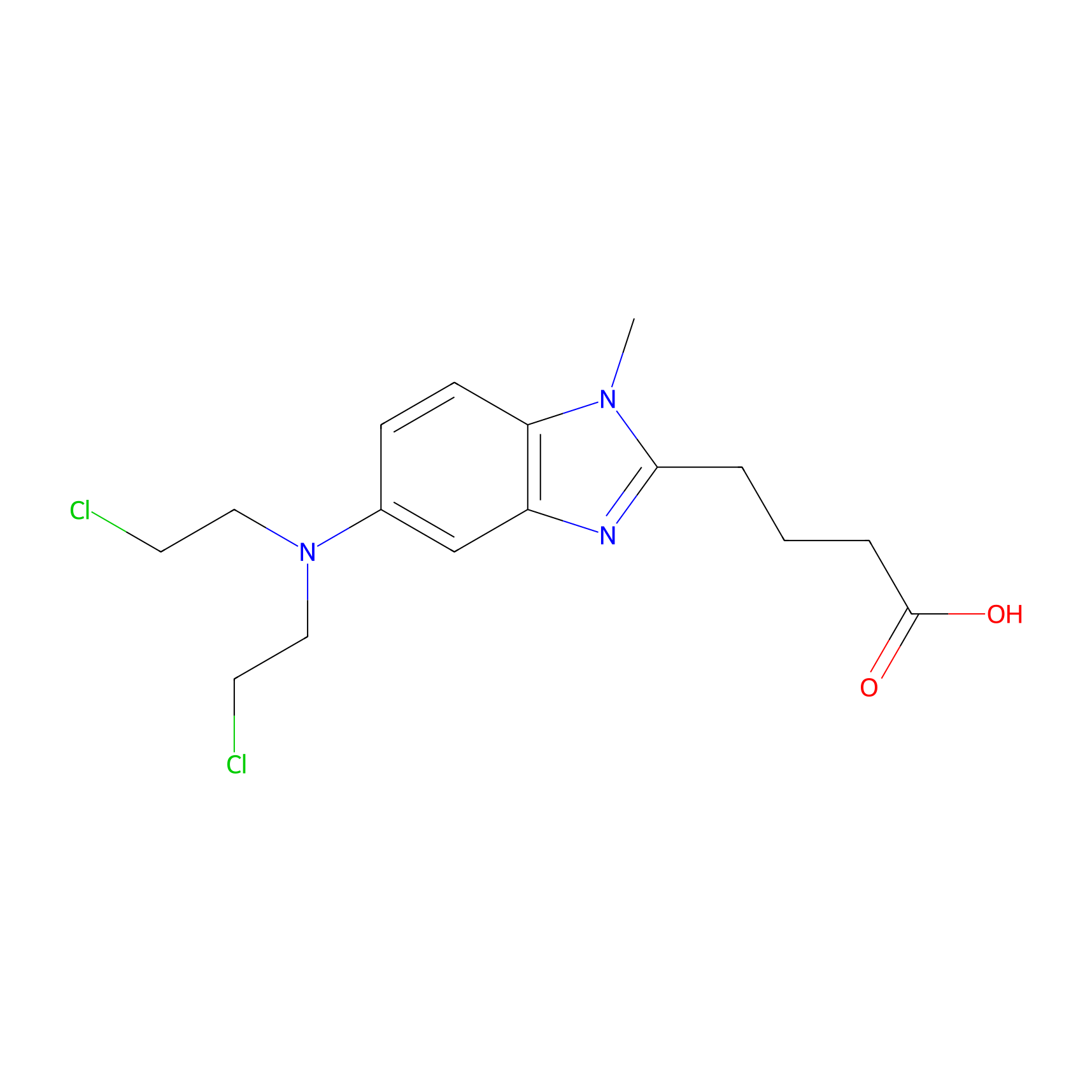
|
|||||
| Formula |
C16H21Cl2N3O2
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 358.3 | ||||
| Lipid-water partition coefficient (xlogp) | 2.9 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 4 | |||||
| Rotatable Bond Count (rotbonds) | 9 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CN1C2=C(C=C(C=C2)N(CCCl)CCCl)N=C1CCCC(=O)O
|
|||||
| InChI |
InChI=1S/C16H21Cl2N3O2/c1-20-14-6-5-12(21(9-7-17)10-8-18)11-13(14)19-15(20)3-2-4-16(22)23/h5-6,11H,2-4,7-10H2,1H3,(H,22,23)
|
|||||
| InChIKey |
YTKUWDBFDASYHO-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
4-[5-[bis(2-chloroethyl)amino]-1-methylbenzimidazol-2-yl]butanoic acid
|
|||||
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
P6-bendamustine [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition | 28.00% | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Description |
Conjugation of the drugs to P4 affected their efficacy toward A20 cells. For chlorambucil and melphalan, conjugation reduced the cytotoxic effect and this was significant for chlorambucil at 25 μM (p = 0.0013). On the other hand, conjugation significantly improved the cytotoxic effect of bendamustine at 25 (p = 0.043) and 50 μM (p = 0.048). The efficacies of all P6-conjugates were significantly lower than those of P4-conjugates at concentrations above 10 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
P4-Bend-PEG-AuNP [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition | 30.00% | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition | 75.00% | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 50 µM | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition | 80.00% | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 µM | ||||
| Description |
All three P4-PDC-coated gold nanoparticles pre-incubated for 24 or 48 h induced statistically similar cytotoxicity in A20 to that induced by freshly prepared PDC4 and to coated particles without pre-incubation (the latter data not shown).
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
P4-bendamustine [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | A20 growth inhibition | 70.00% | |||
| Evaluation Method | XTT assay | ||||
| Administration Time | 72 h | ||||
| Administration Dosage | 50 µM | ||||
| Description |
Conjugation of the drugs to P4 affected their efficacy toward A20 cells. For chlorambucil and melphalan, conjugation reduced the cytotoxic effect and this was significant for chlorambucil at 25 μM (p = 0.0013). On the other hand, conjugation significantly improved the cytotoxic effect of bendamustine at 25 (p = 0.043) and 50 μM (p = 0.048). The efficacies of all P6-conjugates were significantly lower than those of P4-conjugates at concentrations above 10 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mouse reticulum cell sarcoma | A20 cell | CVCL_1940 | ||
References
