Drug Information
General Information of This Drug
| Drug ID | DRG00026 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Maytansine | |||||
| Synonyms |
MAYTANSINE; Maitansine; Maytansine [USAN]; Maitansinum; Maitansina; NSC-153858; 35846-53-8; Maitansine [INN]; Maytansin; CHEBI:6701; NSC 153858; Maytansine (USAN); N-Acetyl-N-methyl-L-alanine(1S-(1R*,2S*,3R*,5R*,6R*,16E,18E,20S*,21R*))-11-chloro-21-hydroxy-12,20-dimethoxy-2,5,9,16-tetramethy-8,23-dioxo-4,24-dioxa-9,22-diazatetracyclo(19.3.1.1(sup 10,14).0(sup 3,5))hexacosa-10,12,14(26),16,18-pentaen-6-yl ester; 14083FR882; (3beta,4beta,5beta,10beta,11e,13e)-Maytansine; Maitansinum [INN-Latin]; NSC153858; Maitansina [INN-Spanish]; EINECS 252-754-7; BRN 5417399; UNII-14083FR882; L-Alanine, N-acetyl-N-methyl-, 11-chloro-21-hydroxy-12,20-dimethoxy-2,5,9,16-tetramethyl-8,23-dioxo-4,24-dioxa-9,22-diazatetracyclo(19.3.1.110,14.03,5)hexacosa-10,12,14(26),16,18-pentaen-6-yl ester, (1S-(1R*,2S*,3R*,5R*,6R*,16E,18E,20S*,21R*))-; MAYTANSINE [MI]; SCHEMBL61357; CHEMBL292702; DTXSID00879995; BDBM50480257; LMPK04000017; (4beta,5beta,11E,13E)-maytansine; HY-13674; CS-0007711; NS00057900; D04864; Q6720157; 3GT
Click to Show/Hide
|
|||||
| Target(s) | Microtubule (MT) | Target Info | ||||
| Structure |
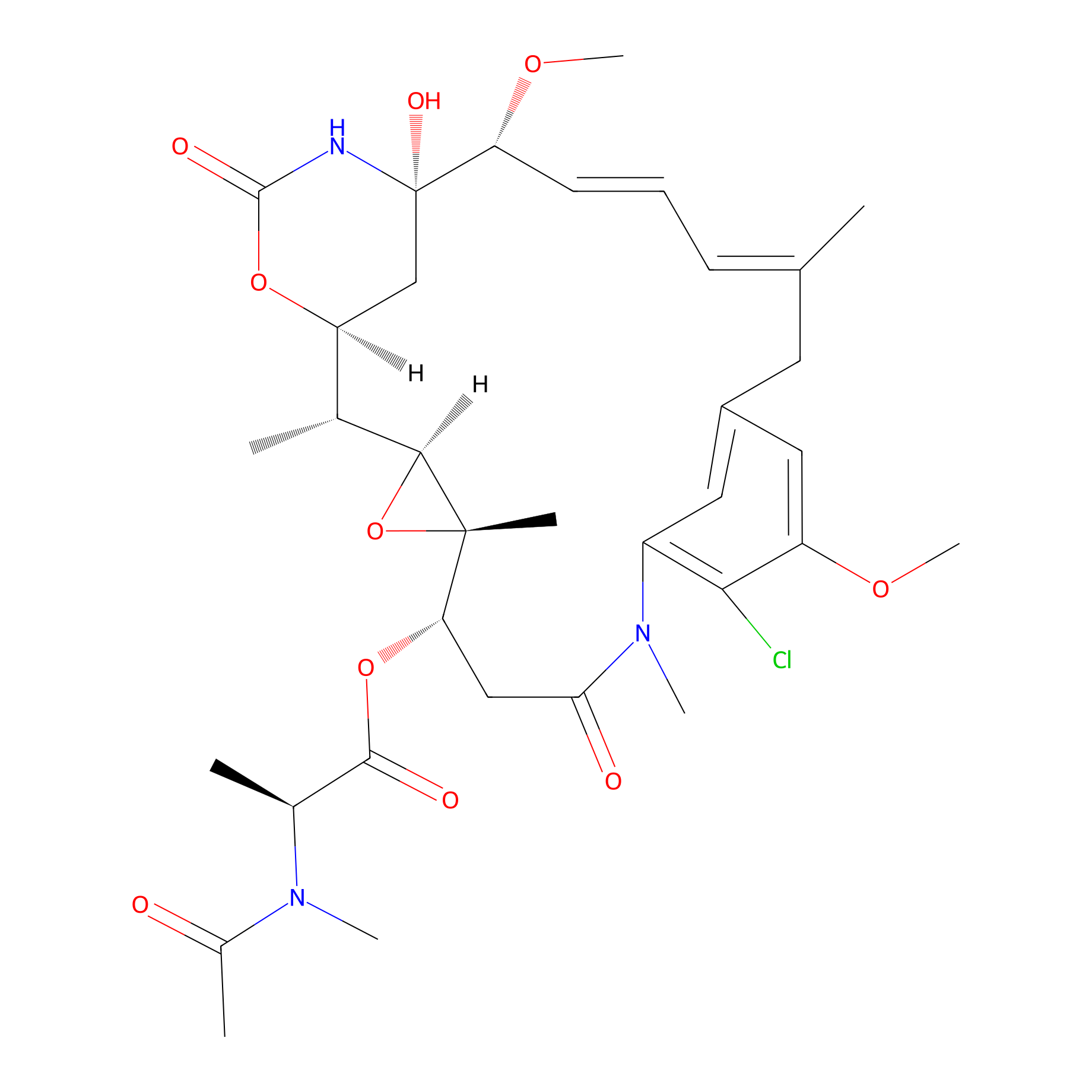
|
|||||
| Formula |
C34H46ClN3O10
|
|||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 692.2 | ||||
| Lipid-water partition coefficient (xlogp) | 2 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 10 | |||||
| Rotatable Bond Count (rotbonds) | 6 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC1C2CC(C(C=CC=C(CC3=CC(=C(C(=C3)OC)Cl)N(C(=O)CC(C4(C1O4)C)OC(=O)C(C)N(C)C(=O)C)C)C)OC)(NC(=O)O2)O
|
|||||
| InChI |
InChI=1S/C34H46ClN3O10/c1-18-11-10-12-26(45-9)34(43)17-25(46-32(42)36-34)19(2)30-33(5,48-30)27(47-31(41)20(3)37(6)21(4)39)16-28(40)38(7)23-14-22(13-18)15-24(44-8)29(23)35/h10-12,14-15,19-20,25-27,30,43H,13,16-17H2,1-9H3,(H,36,42)/b12-10+,18-11+/t19-,20+,25+,26-,27+,30+,33+,34+/m1/s1
|
|||||
| InChIKey |
WKPWGQKGSOKKOO-RSFHAFMBSA-N
|
|||||
| IUPAC Name |
[(1S,2R,3S,5S,6S,16E,18E,20R,21S)-11-chloro-21-hydroxy-12,20-dimethoxy-2,5,9,16-tetramethyl-8,23-dioxo-4,24-dioxa-9,22-diazatetracyclo[19.3.1.110,14.03,5]hexacosa-10,12,14(26),16,18-pentaen-6-yl] (2S)-2-[acetyl(methyl)amino]propanoate
|
|||||
The activity data of This Drug
| Standard Type | Value | Administration times | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | 0.026 µM | 48 h | HCT 116 cell | CVCL_0291 | [1] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
BT1718 [Phase 2]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Grade 3 increased GGT | 8.30% | |||
| Patients Enrolled |
24 patients with various types of solid tumors.
|
||||
| Administration Dosage | 9.6 mg/m2 BIW | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
24 patients were enrolled with various types of solid tumors across both dose escalation cohorts (see table). BIW RP2D was determined as 7.2 mg/m2. The 2 patients with DLTs at 9.6 mg/m2 BIW experienced grade 3 increased GGT or fatigue. QW dose escalation continues at 20 mg/m2. Mean number of cycles received = 2.3 months (N = 24), with no objective responses observed to date in this unselected population.
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Fatigue | 8.30% | |||
| Patients Enrolled |
24 patients with various types of solid tumors.
|
||||
| Administration Dosage | 9.6 mg/m2 BIW | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
24 patients were enrolled with various types of solid tumors across both dose escalation cohorts (see table). BIW RP2D was determined as 7.2 mg/m2. The 2 patients with DLTs at 9.6 mg/m2 BIW experienced grade 3 increased GGT or fatigue. QW dose escalation continues at 20 mg/m2. Mean number of cycles received = 2.3 months (N = 24), with no objective responses observed to date in this unselected population.
Click to Show/Hide
|
||||
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 12.50% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | Twice a week for 12 days | ||||
| Administration Dosage | 1 mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 72 days.
|
||||
| In Vivo Model | Mice implanted with MT1-positive HT-1080 cells | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 12.70% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | Three times a week for 12 days | ||||
| Administration Dosage | 1 mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 75 days.
|
||||
| In Vivo Model | Mice implanted with MT1-positive HT-1080 cells | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 18% | |||
| Administration Time | 14 days | ||||
| Administration Dosage | 1 mg/kg qw | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 or BT1718) showed rapid and complete tumour clearance (EBC-1 cells), while BDCs containing more stabilised linkers showed comparatively reduced efficacy (fig. 5) suggesting that target internalisation is not the sole mechanism of action for BDC efficacy and that extracellular cleavage and release of toxin within the local tumour environment likely also contribute. Only the most labile BDC (BT17BDC17) caused any significant toxicity (17% 9.7 body weight loss); all others were well tolerated (<10% body weight loss at 10 mg/kg tiw). Optimal therapeutic index was achieved with BT1718. Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10 mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
Click to Show/Hide
|
||||
| In Vivo Model | Cell-derived xenograftmodel inmiceimplanted with MT1-positive HT-1080 cells. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 27% | |||
| Administration Time | 14 days | ||||
| Administration Dosage | 1 mg/kg biw | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 or BT1718) showed rapid and complete tumour clearance (EBC-1 cells), while BDCs containing more stabilised linkers showed comparatively reduced efficacy (fig. 5) suggesting that target internalisation is not the sole mechanism of action for BDC efficacy and that extracellular cleavage and release of toxin within the local tumour environment likely also contribute. Only the most labile BDC (BT17BDC17) caused any significant toxicity (17% 9.7 body weight loss); all others were well tolerated (<10% body weight loss at 10 mg/kg tiw). Optimal therapeutic index was achieved with BT1718. Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10 mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
Click to Show/Hide
|
||||
| In Vivo Model | Cell-derived xenograftmodel inmiceimplanted with MT1-positive HT-1080 cells. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 85.40% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | Three times a week for 12 days | ||||
| Administration Dosage | 3 mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 74 days.
|
||||
| In Vivo Model | Mice implanted with MT1-positive HT-1080 cells | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 88% | |||
| Administration Time | 14 days | ||||
| Administration Dosage | 3 mg/kg qw | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 or BT1718) showed rapid and complete tumour clearance (EBC-1 cells), while BDCs containing more stabilised linkers showed comparatively reduced efficacy (fig. 5) suggesting that target internalisation is not the sole mechanism of action for BDC efficacy and that extracellular cleavage and release of toxin within the local tumour environment likely also contribute. Only the most labile BDC (BT17BDC17) caused any significant toxicity (17% 9.7 body weight loss); all others were well tolerated (<10% body weight loss at 10 mg/kg tiw). Optimal therapeutic index was achieved with BT1718. Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10 mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
Click to Show/Hide
|
||||
| In Vivo Model | Cell-derived xenograftmodel inmiceimplanted with MT1-positive HT-1080 cells. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Lung squamous cell carcinoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Administration Time | 14 days | ||||
| Administration Dosage | 10mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 or BT1718) showed rapid and complete tumour clearance (EBC-1 cells), while BDCs containing more stabilised linkers showed comparatively reduced efficacy (fig. 5) suggesting that target internalisation is not the sole mechanism of action for BDC efficacy and that extracellular cleavage and release of toxin within the local tumour environment likely also contribute. Only the most labile BDC (BT17BDC17) caused any significant toxicity (17% 9.7 body weight loss); all others were well tolerated (<10% body weight loss at 10 mg/kg tiw). Optimal therapeutic index was achieved with BT1718. Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10 mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
Click to Show/Hide
|
||||
| In Vivo Model | Cell-derived xenograftmodel inmiceimplanted with MT1-positive EBC-1 cells. | ||||
| In Vitro Model | Lung squamous cell carcinoma | EBC-1 cell | CVCL_2891 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Administration Time | 14 days | ||||
| Administration Dosage | 10 mg/kg biw | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 or BT1718) showed rapid and complete tumour clearance (EBC-1 cells), while BDCs containing more stabilised linkers showed comparatively reduced efficacy (fig. 5) suggesting that target internalisation is not the sole mechanism of action for BDC efficacy and that extracellular cleavage and release of toxin within the local tumour environment likely also contribute. Only the most labile BDC (BT17BDC17) caused any significant toxicity (17% 9.7 body weight loss); all others were well tolerated (<10% body weight loss at 10 mg/kg tiw). Optimal therapeutic index was achieved with BT1718. Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10 mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
Click to Show/Hide
|
||||
| In Vivo Model | Cell-derived xenograftmodel inmiceimplanted with MT1-positive HT-1080 cells. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Administration Time | 14 days | ||||
| Administration Dosage | 3 mg/kg biw | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 or BT1718) showed rapid and complete tumour clearance (EBC-1 cells), while BDCs containing more stabilised linkers showed comparatively reduced efficacy (fig. 5) suggesting that target internalisation is not the sole mechanism of action for BDC efficacy and that extracellular cleavage and release of toxin within the local tumour environment likely also contribute. Only the most labile BDC (BT17BDC17) caused any significant toxicity (17% 9.7 body weight loss); all others were well tolerated (<10% body weight loss at 10 mg/kg tiw). Optimal therapeutic index was achieved with BT1718. Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10 mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
Click to Show/Hide
|
||||
| In Vivo Model | Cell-derived xenograftmodel inmiceimplanted with MT1-positive HT-1080 cells. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Administration Time | 14 days | ||||
| Administration Dosage | 10 mg/kg qw | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 or BT1718) showed rapid and complete tumour clearance (EBC-1 cells), while BDCs containing more stabilised linkers showed comparatively reduced efficacy (fig. 5) suggesting that target internalisation is not the sole mechanism of action for BDC efficacy and that extracellular cleavage and release of toxin within the local tumour environment likely also contribute. Only the most labile BDC (BT17BDC17) caused any significant toxicity (17% 9.7 body weight loss); all others were well tolerated (<10% body weight loss at 10 mg/kg tiw). Optimal therapeutic index was achieved with BT1718. Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10 mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
Click to Show/Hide
|
||||
| In Vivo Model | Cell-derived xenograftmodel inmiceimplanted with MT1-positive HT-1080 cells. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | 12 days | ||||
| Administration Dosage | 10 mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Treatment with BDCs containing the most labile linkers (BT17BDC17 orBT1718) showed rapid and complete tumour clearance (EBC-1 cells).
|
||||
| In Vivo Model | Mice implanted with MT1-positive EBC-1 cells | ||||
| In Vitro Model | Lung squamous cell carcinoma | EBC-1 cell | CVCL_2891 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | Twice a week for 12 days | ||||
| Administration Dosage | 10 mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 70 days.
|
||||
| In Vivo Model | Mice implanted with MT1-positive HT-1080 cells | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | Twice a week for 12 days | ||||
| Administration Dosage | 3 mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 71 days.
|
||||
| In Vivo Model | Mice implanted with MT1-positive HT-1080 cells | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) | 100% | |||
| Evaluation Method | Tumor volume detection | ||||
| Administration Time | Three times a week for 12 days | ||||
| Administration Dosage | 10 mg/kg | ||||
| MOA of PDC |
BT1718 is a novel first in class bicyclic targeting peptide that selectively binds MT1 MMP (MMP-14) and is linked to the maytansinoid tubulin inhibitor DM1 by a cleavable disulfide linker. Bicycle Toxin Conjugates have a low molecular weight compared to other conjugated toxin approaches, enabling rapid tumour penetration and a short systemic half-life (up to 40 minutes in non-human primates) potentially reducing toxicity. The target MT1-MMP (MT1) is a surface metalloproteinase involved in tissue remodelling through proteolysis of extracellular matrix components: Highly expressed in tumours with unmet medical need, such as triple negative breast cancer, non small cell lung cancer and ovarian cancer. Strong link with cell invasion and metastasis. High tumour MT1 expression is correlated with poor outcomes in multiple tumour types. High adjacent stromal expression and low expression in adult normal tissue.
Click to Show/Hide
|
||||
| Description |
Testing of BT1718 in different dosing regimes in an additional model (HT-1080 cells) also demonstrated excellent tumour regression, with 10mg/kg biw leading to complete tumour clearance in all 3 animals within 23 days and no re-growth out to 73 days.
|
||||
| In Vivo Model | Mice implanted with MT1-positive HT-1080 cells | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
LWJ-M30 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Colorectal cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.047 µM | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| MOA of PDC |
Given these findings, we employed tissue-specific drug delivery approaches PDC to overcome those adverse effects. Several TfR-targeted peptides sequence with different affinity have been reported, so we synthesized a series of TfR-targeted peptide-DM1 conjugates for screening the effects of TfR-targeted drug candidates. It is speculated that the conjugates can specifically target tumor with high expression of TfR and can be uptake more than that of the normal cells. We succeeded found LWJ-M30, a conjugate of DM1 and B6, had a significant therapeutic effect after preliminary screening. In order to further reveal the improved therapeutic effects and mechanism of the LWJ-M30 on cancer with high TfR expression, in this study, we investigated the role of the LWJ-M30 in the targeted therapy for colorectal cancer in vitro and in vivo and explored the therapeutic mechanism.
Click to Show/Hide
|
||||
| Description |
To determine the activity of the compounds, we analyzed the effects of them on cell growth and migration in HCT116 cells. We found that LWJ-M30 displayed significant inhibition of cell proliferation and clone formation ability. The IC50 of LWJ-M30 was 0.047 uM which was close to DM1 (0.026 uM). According to the results of wound-healing assay and transwell migration assay, LWJ-M30 also dramatically showed a inhibitory effect on cells migration. These results suggested that LWJ-M30 could inhibit the cells proliferation and migration.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
References
