Drug Information
General Information of This Drug
| Drug ID | DRG00032 | |||||
|---|---|---|---|---|---|---|
| Drug Name | N1-(7-chloroquinolin-4-yl)butane-1,4-diamine | |||||
| Synonyms |
53186-45-1; 4-(4-Aminobutyl)amino-7-chloroquinoline; N1-(7-CHLOROQUINOLIN-4-YL)BUTANE-1,4-DIAMINE; 1,4-Butanediamine, n1-(7-chloro-4-quinolinyl)-; N'-(7-chloroquinolin-4-yl)butane-1,4-diamine; MFCD07700284; N-(4-aminobutyl)-7-chloroquinolin-4-amine; N-(7-chloroquinolin-4-yl)butane-1,4-diamine; CHEMBL236491; SCHEMBL1147513; DTXSID50472238; FIVQKRXTOFYZCQ-UHFFFAOYSA-N; AKOS010116787; SB71980; AS-53416; CS-0188349; N-(7-chloro-4-quinolyl)-1,4-diaminobutane; N'-(7-chloro-4-quinolyl)butane-1,4-diamine; P16457; N1-(7-Chloro-4-quinolinyl)-1,4-butanediamine
Click to Show/Hide
|
|||||
| Structure |
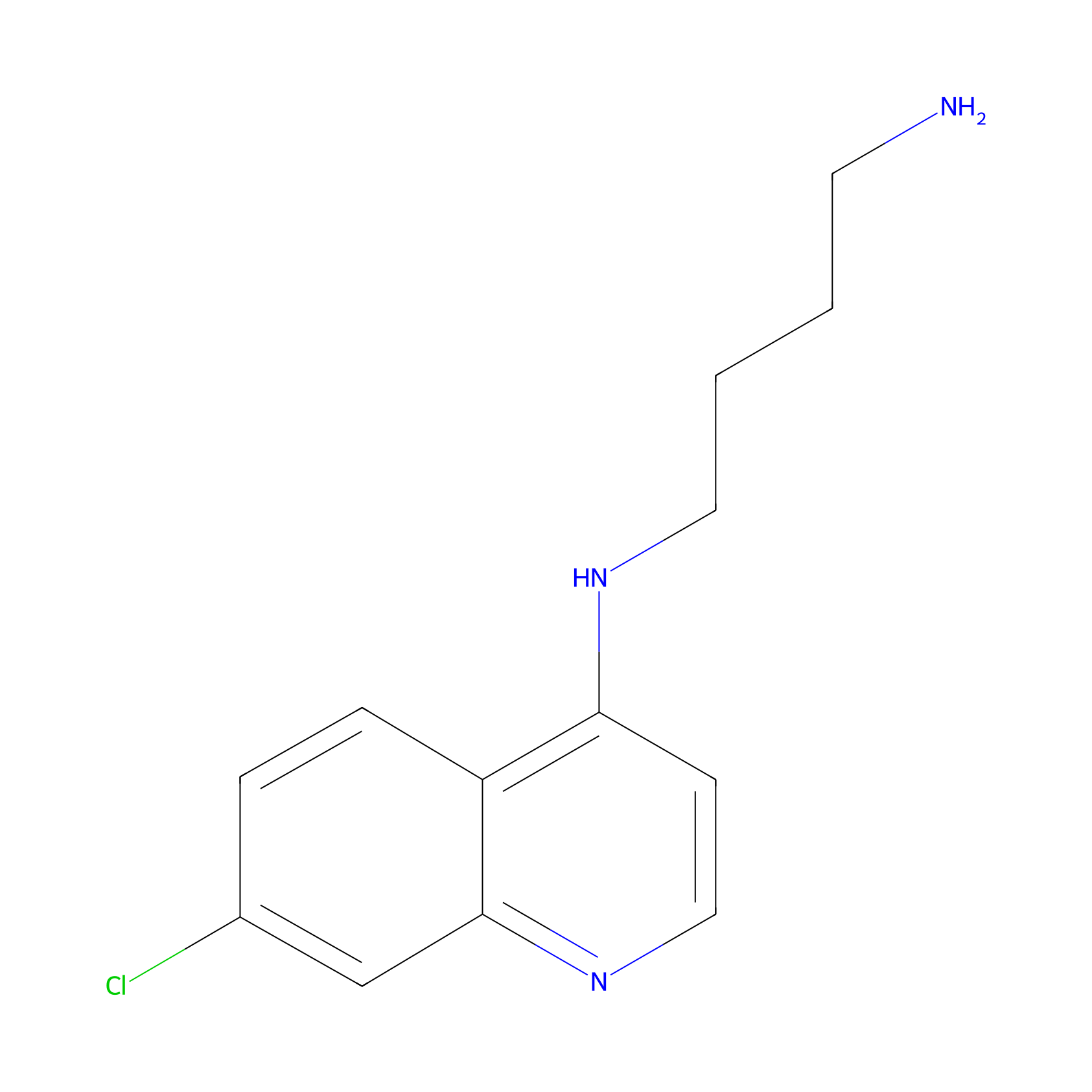
|
|||||
| Formula |
C13H16ClN3
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 249.74 | ||||
| Lipid-water partition coefficient (xlogp) | 2.7 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||
| Rotatable Bond Count (rotbonds) | 5 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C1=CC2=C(C=CN=C2C=C1Cl)NCCCCN
|
|||||
| InChI |
InChI=1S/C13H16ClN3/c14-10-3-4-11-12(16-7-2-1-6-15)5-8-17-13(11)9-10/h3-5,8-9H,1-2,6-7,15H2,(H,16,17)
|
|||||
| InChIKey |
FIVQKRXTOFYZCQ-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
N'-(7-chloroquinolin-4-yl)butane-1,4-diamine
|
|||||
Full Information of The Activity Data of The PDC(s) Related to This Drug
Cq-C4-TP10 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.8 ± 0.1 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| In Vivo Model | Plasmodium falciparum 3D7. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.5 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C10-TP10 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 0.8 ± 0.3 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| In Vivo Model | Plasmodium falciparum 3D7. | ||||
Cq-S-S-TP10 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.0 ± 0.3 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| In Vivo Model | Plasmodium falciparum 3D7. | ||||
Cq-TR-TP10 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1.2 ± 0.2 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| In Vivo Model | Plasmodium falciparum 3D7. | ||||
Cq-C4-Transportan [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 5.2 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C4-PasTAT [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8.5 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C4-DPT-sh1 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C4-DPT-sh2 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C4-IDR-1018 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C4-TAT [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C4-R9 [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
Cq-C4-Penetratin [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malaria | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 10 µM | |||
| MOA of PDC |
The significant increase in the hemolytic activity of TP10 upon conjugation to the 4-aminoquinoline suggests that drug cargo prevents an otherwise active CPP carrier from exerting the desired cell penetrating/antiplasmodial action safely, as it produces conjugates that exert membranolytic activity.
|
||||
| Description |
Only three of the Cq-C4-CPP conjugates, namely, 5a, 5b, and 5g, displayed IC50 values below 10 μM, with TP10- and Transportan-derived conjugates 5a (IC50 = 1.52 μM) and 5b (IC50 = 5.20 μM) being the most active.
|
||||
| In Vivo Model | Plasmodium falciparum W2. | ||||
References
