Drug Information
General Information of This Drug
| Drug ID | DRG00040 | |||||
|---|---|---|---|---|---|---|
| Drug Name | NLG919 | |||||
| Synonyms |
NLG919; 1402836-58-1; 1-cyclohexyl-2-(5H-imidazo[5,1-a]isoindol-5-yl)ethanol; NLG919 (GDC-0919); 1-Cyclohexyl-2-(5H-imidazo-[5,1-a]isoindol-5-yl)ethanol; CHEMBL3629569; NLG919(GDC-0919,Navoximod); MFCD26142661; 5H-Imidazo[5,1-a]isoindole-5-ethanol, alpha-cyclohexyl-; 1-Cyclohexyl-2-(5H-imidazo[5,1-a]isoindol-5-yl)ethan-1-ol; 1-CYCLOHEXYL-2-{5H-IMIDAZO[4,3-A]ISOINDOL-5-YL}ETHANOL; YTRRAUACYORZLX-UHFFFAOYSA-N; GTPL9019; SCHEMBL13280897; DTXSID20735206; EX-A574; HMS3653M05; AMY12328; BCP09887; BDBM50126144; s7111; AKOS025287054; CCG-267268; CS-3512; SB16495; NCGC00386274-04; AC-30124; AS-35112; DA-45347; HY-13983; SY046802; FT-0700192; SW219857-1; A853907; J-690162; Q27087945; 1-cyclohexyl-2-[5H-imidazo[4,3-a]isoindol-5-yl]ethan-1-ol; 5-(2-Cyclohexyl-2-hydroxyethyl)-5H-imidazo[5,1-a]isoindole; 3-(2-Amino-ethyl)-5-[3-(4-butoxyl-phenyl)-propylidene]-thiazolidine-2,4-dione hydrochloride; a-Cyclohexyl-5H-imidazo[5,1-a]isoindole-5-ethanol; 1-Cyclohexyl-2-(5H-imidazo[5,1-a]isoindol-5-yl)ethanol; NLG919; alpha-Cyclohexyl-5H-imidazo[5,1-a]isoindole-5-ethanol;1-cyclohexyl-2-(5H-imidazo[5,1-a]isoindol-5-yl)ethanol
Click to Show/Hide
|
|||||
| Target(s) | Indoleamine 2,3-dioxygenase 1 (IDO1) | Target Info | ||||
| Structure |
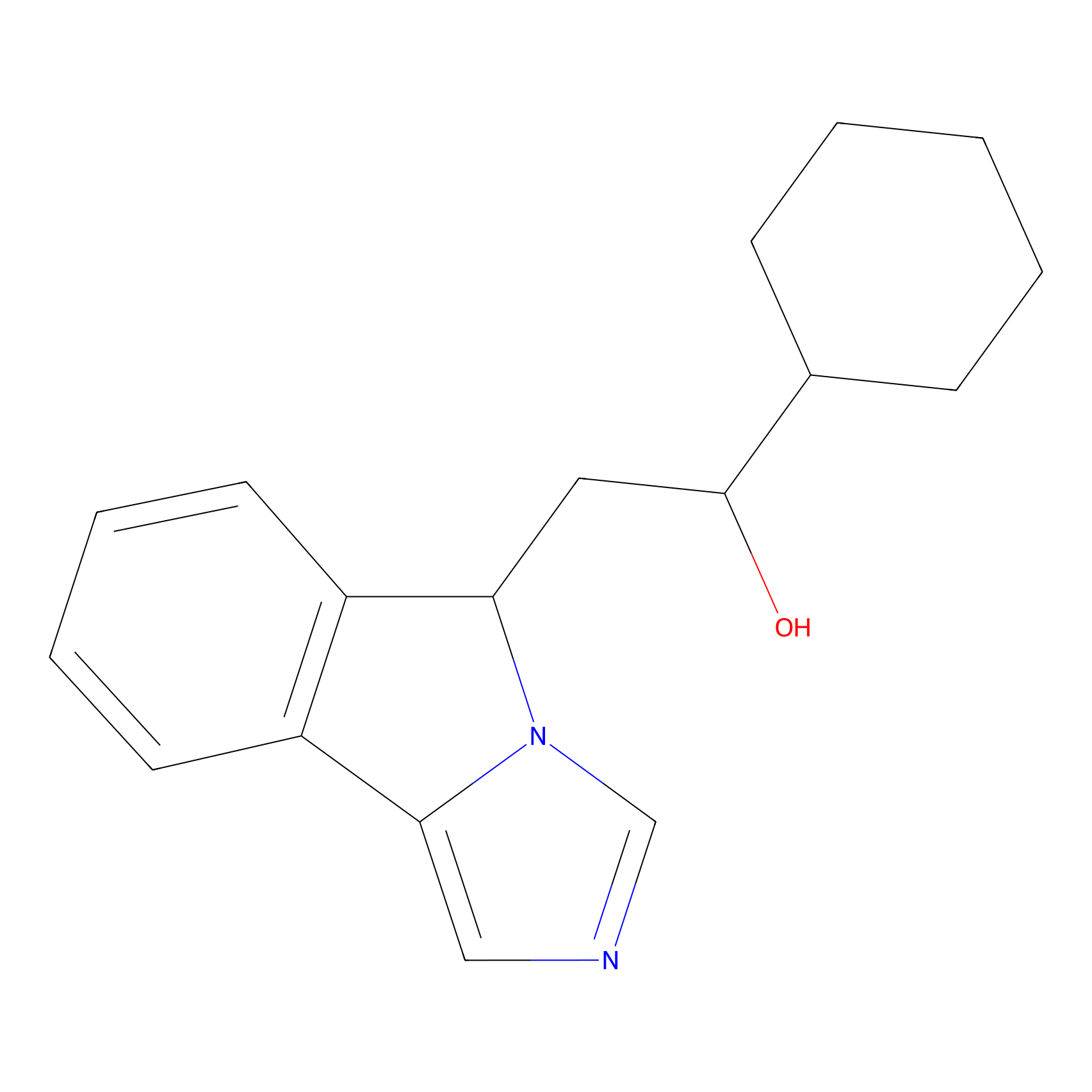
|
|||||
| Formula |
C18H22N2O
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 282.4 | ||||
| Lipid-water partition coefficient (xlogp) | 3.5 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 2 | |||||
| Rotatable Bond Count (rotbonds) | 3 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C1CCC(CC1)C(CC2C3=CC=CC=C3C4=CN=CN24)O
|
|||||
| InChI |
InChI=1S/C18H22N2O/c21-18(13-6-2-1-3-7-13)10-16-14-8-4-5-9-15(14)17-11-19-12-20(16)17/h4-5,8-9,11-13,16,18,21H,1-3,6-7,10H2
|
|||||
| InChIKey |
YTRRAUACYORZLX-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
1-cyclohexyl-2-(5H-imidazo[5,1-a]isoindol-5-yl)ethanol
|
|||||
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
NLG-RGD [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of Treg cells | 1% | |||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L5 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | Regulatory CD4+ T cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of NK cells | 2% | |||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L6 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | Natural killer cell | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of IFN-γ-producing CD8 T cells | 40% | |||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L4 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | IFN-gamma-producing CD4 T cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of IFN-γ-producing CD4 T cells | 16% | |||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L3 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | IFN-gamma-producing CD4 T cell | Homo sapiens | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of CD8 T cells | 34% | |||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L2 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | CD8 T cell | Homo sapiens | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of CD4 T cells | 32% | |||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L1 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | CD4 T cell | Homo sapiens | ||
References
