Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00252
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
NLG-RGD
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
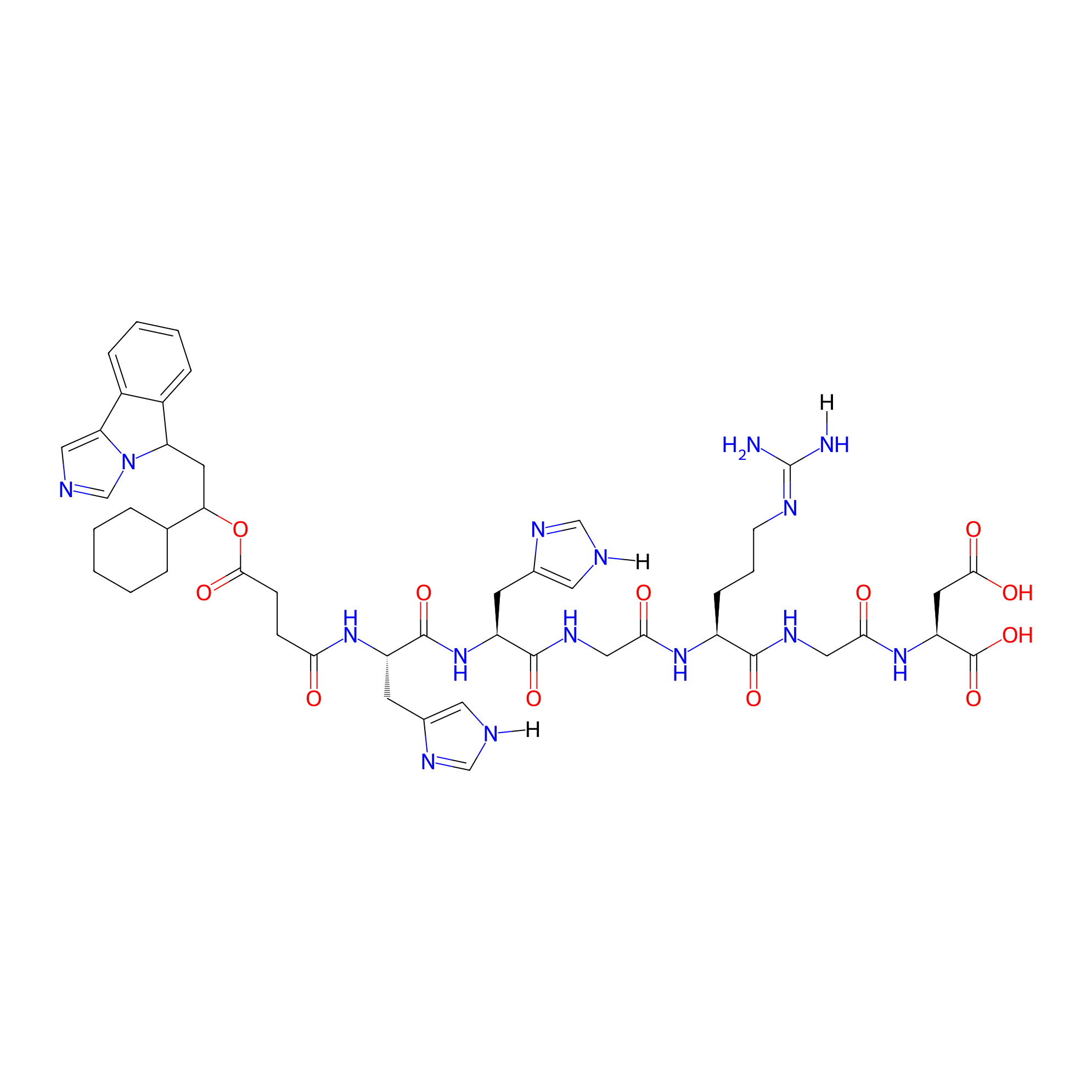
|
|||||
| Peptide Name |
c[DKP-RGD]
|
Peptide Info | ||||
| Receptor Name |
Indoleamine 2,3-dioxygenase 1 (IDO1)
|
Receptor Info | ||||
| Drug Name |
NLG919
|
Drug Info | ||||
| Therapeutic Target |
Indoleamine 2,3-dioxygenase 1 (IDO1)
|
Target Info | ||||
| Linker Name |
Succinic Acid
|
Linker Info | ||||
| Peptide Modified Type |
Modification by dosage
|
|||||
| Modified Segment |
Self-assemble
|
|||||
| Formula |
C48H63N15O12
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1042.125 | ||||
| Lipid-water partition coefficient (xlogp) | -1.1686 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 12 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 15 | |||||
| Rotatable Bond Count (rotbonds) | 29 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of CD4 T cells |
32%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L1 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | CD4 T cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of CD8 T cells |
34%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L2 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | CD8 T cell | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of IFN-γ-producing CD4 T cells |
16%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L3 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | IFN-gamma-producing CD4 T cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of IFN-γ-producing CD8 T cells |
40%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L4 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | IFN-gamma-producing CD4 T cell | Homo sapiens | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of NK cells |
2%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L6 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | Natural killer cell | Homo sapiens | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of Treg cells |
1%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L5 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | Regulatory CD4+ T cell | Homo sapiens | ||
References
