Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00163
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
c[DKP-RGD]
|
|||||
| Structure |
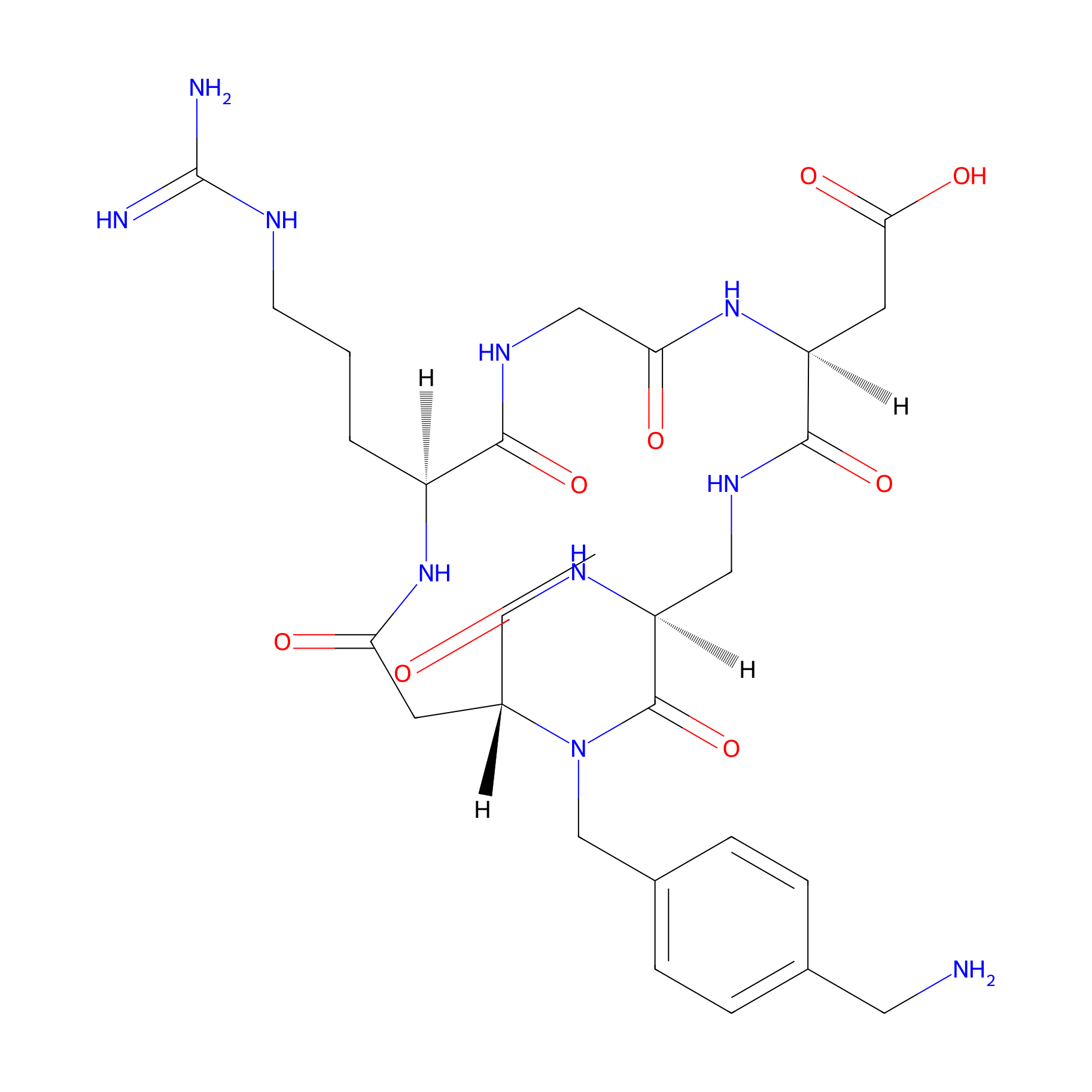
|
|||||
| Sequence |
RGD
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Indoleamine 2,3-dioxygenase 1 (IDO1)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C27H38N10O8
|
|||||
| Isosmiles |
N=C(N)NCCC[C@@H]1NC(=O)C[C@@H]2C(=O)N[C@@H](CNC(=O)[C@H](CC(=O)O)NC(=O)CNC1=O)C(=O)N2Cc1ccc(CN)cc1
|
|||||
| InChI |
InChI=1S/C27H38N10O8/c28-10-14-3-5-15(6-4-14)13-37-19-9-20(38)34-16(2-1-7-31-27(29)30)23(42)33-12-21(39)35-17(8-22(40)41)24(43)32-11-18(26(37)45)36-25(19)44/h3-6,16-19H,1-2,7-13,28H2,(H,32,43)(H,33,42)(H,34,38)(H,35,39)(H,36,44)(H,40,41)(H4,29,30,31)/t16-,17-,18-,19+/m0/s1
|
|||||
| InChIKey |
LDILHAXCSRSOSM-CADBVGFASA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
630.663
|
Polar area
|
291.03
|
||
|
Complexity
|
630.2874082
|
xlogp Value
|
-4.31543
|
|||
|
Heavy Count
|
45
|
Rot Bonds
|
9
|
|||
|
Hbond acc
|
9
|
Hbond Donor
|
10
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1], [2] | |||||
| IC50 | > 5 nM | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
IC50values were calculated as the concentration of compound required for 50% inhibition of biotinylated vitronectin binding
|
|||||
| Experimental Condition | αvβ5 receptor | |||||
| Peptide Activity Information 2 | [1], [2] | |||||
| IC50 | 26.4±3.7 nM | |||||
| Binding Affinity Assay |
IC50values were calculated as the concentration of compound required for 50% inhibition of biotinylated vitronectin binding
|
|||||
| Experimental Condition | αvβ3 receptor | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
NLG-RGD [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of Treg cells |
1%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L5 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | Regulatory CD4+ T cell | Homo sapiens | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of NK cells |
2%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L6 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | Natural killer cell | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of IFN-γ-producing CD8 T cells |
40%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L4 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | IFN-gamma-producing CD4 T cell | Homo sapiens | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of IFN-γ-producing CD4 T cells |
16%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L3 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | IFN-gamma-producing CD4 T cell | Homo sapiens | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of CD8 T cells |
34%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L2 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | CD8 T cell | Homo sapiens | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Percentage of CD4 T cells |
32%
|
|||
| Administration Time | 30 min | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | Quantification of tumor-infiltrating immune cells assay | ||||
| MOA of PDC |
Significantly, this nanoinhibitor boosts the antitumor immune response of PD-L1 blockade by increasing immune effector cells and reducing immunosuppressive cells.
|
||||
| Description |
These findings support that aPD-L1 plus NLG-RGD NI efficiently elicits antitumor immunity by enhancing the activation and survival of immune effector cells and attenuating the accumulation immunosuppressive Treg cells.
|
||||
| In Vivo Model | Pan02 tumor model. | ||||
| In Vitro Model | Normal | CD4 T cell | Homo sapiens | ||
Dau=Aoa-GFLG-c[DKP-RGD] [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
2.5 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
5.4 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
14.7 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
49.4 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
c[DKP-RGD]-PEG4-Aoa=dau [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
3 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
9.2 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
21.7 μM
|
|||
| Administration Time | 72 h | ||||
| Description |
All dau-loaded conjugates showed high toxicity in all cell lines tested with EC50 values in the lower micromolar range.
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
56.9 μM
|
|||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87 cell | CVCL_3429 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Colon cancer | HT29 cell | CVCL_A8EZ | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) | > 150 μM | |||
| Administration Time | 15 min | ||||
| Description |
More specifically, compounds1band3bdemonstrated significantly higher activity against U87 cells compared to MCF-7 and HT-29 cells (Figure6). Importantly,1bwas more active than3bdemonstrating that the attached CPP is indeed necessary to increase the overall cellular uptake and thus cytotoxic activity of the conjugates. Of note is also that2bwas less efficient than1bbut marginally more active than3b, although with no selectivity. For the two conjugates bearing the GFLG motif, it was seen that3cis still significantly more active in U87 cells expressing integrin receptors. By contrast,1ckept its selectivity against HT-29 cells, but was pronouncedly more active in MCF-7 cells compared to1b.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
References
