Drug Information
General Information of This Drug
| Drug ID | DRG00045 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Duocarmycin Sa | |||||
| Synonyms |
CHEMBL4443767
Click to Show/Hide
|
|||||
| Target(s) | Human Deoxyribonucleic acid (hDNA) | Target Info | ||||
| Structure |
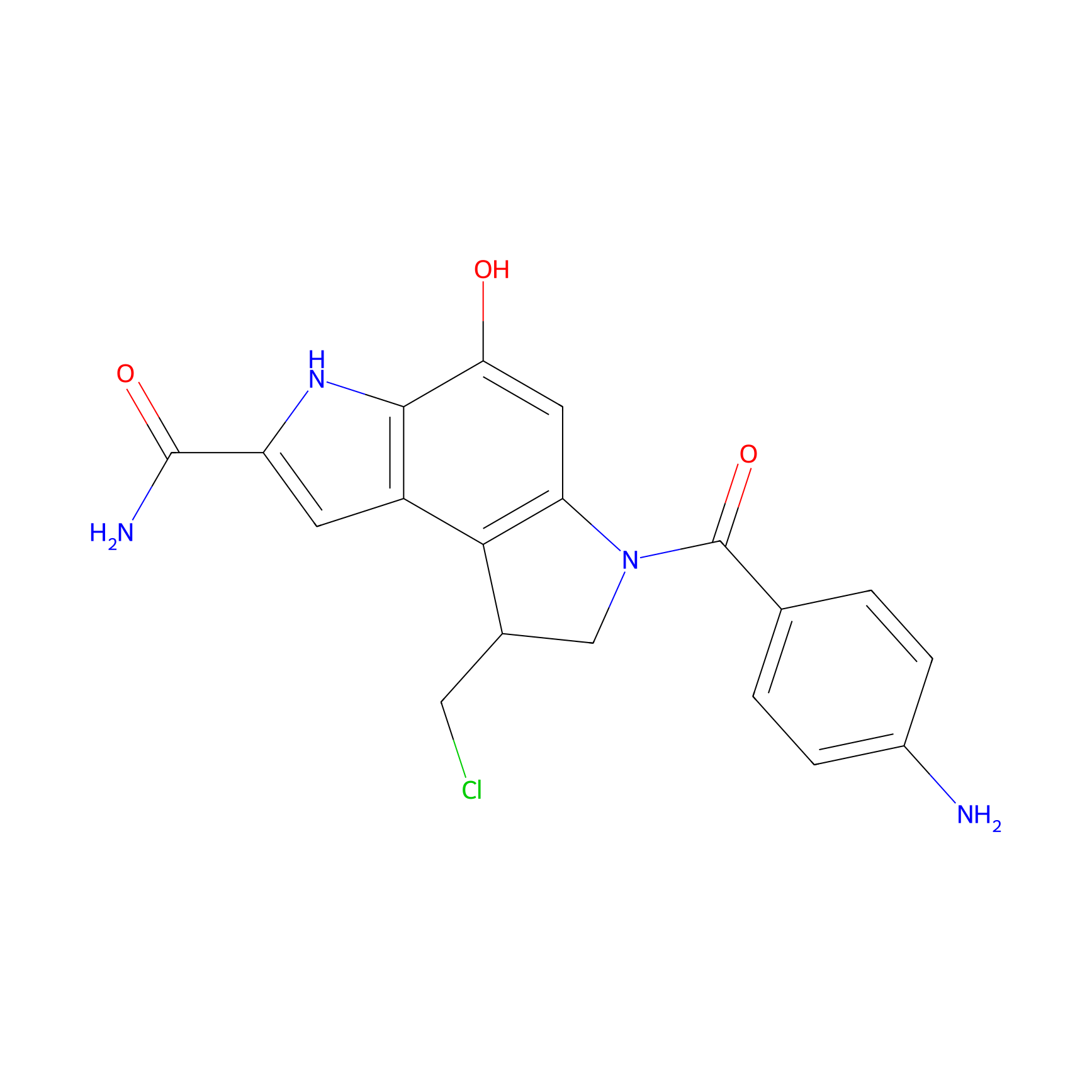
|
|||||
| Formula |
C19H17ClN4O3
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 384.8 | ||||
| Lipid-water partition coefficient (xlogp) | 1.7 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 4 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 4 | |||||
| Rotatable Bond Count (rotbonds) | 3 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C1C(C2=C3C=C(NC3=C(C=C2N1C(=O)C4=CC=C(C=C4)N)O)C(=O)N)CCl
|
|||||
| InChI |
InChI=1S/C19H17ClN4O3/c20-7-10-8-24(19(27)9-1-3-11(21)4-2-9)14-6-15(25)17-12(16(10)14)5-13(23-17)18(22)26/h1-6,10,23,25H,7-8,21H2,(H2,22,26)
|
|||||
| InChIKey |
DSGWQDAPTJQUBO-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
6-(4-aminobenzoyl)-8-(chloromethyl)-4-hydroxy-7,8-dihydro-3H-pyrrolo[3,2-e]indole-2-carboxamide
|
|||||
The activity data of This Drug
| Standard Type | Value | Administration times | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | 72 h | SK-BR-3 cell | CVCL_0033 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 9 nM | 72 h | H292 cell | CVCL_0455 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 28 nM | 72 h | MCF-7 cell | CVCL_0031 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 33 nM | 72 h | HL-60 cell | CVCL_0002 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 39 nM | 72 h | A-549 cell | CVCL_0023 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 57 nM | 72 h | HCT 116 cell | CVCL_0291 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 89 nM | 72 h | 16HBE14o- cell | CVCL_0112 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | 154 nM | 72 h | HT-1080 cell | CVCL_0317 | [1] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
TFα-peptide-duocarmycin conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Colon adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 402 nM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TF-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Colon carcinoma | HCT 116 cell | CVCL_0291 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 526 nM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Breast adenocarcinoma | SK-BR-3 cell | CVCL_0033 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 864 nM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TFα-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Acute myeloid leukemia | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 1831 nM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TF-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 8266 nM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Lung adenocarcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
The cancer phenotype is commonly associated with aberrant glycosylation patterns. One glycan that is directly linked to cancer is the Thomsen-Friedenreich antigen (TF or CD176). It is a disaccharide composed of a galactose β1-3 N-acetylgalactosamine, O-linked to a glycoprotein through serine or threonine residues and commonly written as Galβ1-3GalNAc--O-Ser/Thr. The TF is therapeutically attractive due to its cryptic nature in normal cells and exposure in embryonic and cancer cells. The expression of the TF has been demonstrated in 90% of primary human carcinomas, including in the lung, the breast, and the pancreas. Additionally, cancer initiating cells or cancer stem cells in the lung, liver, and colon express the TF. The peptide sequence HGRFILPWWYAFSPS (TF-peptide) is known to bind tightly to the TF (Kd = 1.2 M) and has been demonstrated to inhibit processes directly involved in TF accessibility.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Lung adenocarcinoma | A-549 cell | CVCL_0023 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Lung mucoepidermoid carcinoma | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Lung mucoepidermoid carcinoma | H292 cell | CVCL_0455 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Normal | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | > 100 µM | |||
| Evaluation Method | MTS cell proliferation assay | ||||
| Administration Time | 72 h | ||||
| MOA of PDC |
As a payload for PDCs (and ADCs), analogues of the duocarmycins are attractive. Duocarmycin SA and yatakemycin rank among the most potent natural cytotoxins discovered. The cyclopropyl and prodrug seco forms are both naturally occurring and equipotent in most circumstances. Studies of the binding-driven bonding model of their interaction with DNA suggest that their utility will be enhanced when targeted to tumor cells. In fact, SYD985, an ADC that utilizes a peptide linker for a duocarmycin analogue to trastuzumab, has recently been progressed to phase III clinical trial.
Click to Show/Hide
|
||||
| Description |
All cell lines demonstrated sensitivity toward the DSA-PABA payload 5 in the nanomolar range. The cleaved peptide 6, and the benzyl protected control peptide 4, had no appreciable effect on cell proliferation (>100 uM), suggesting that any observed cytotoxic activity is due to the DSA warhead. Interestingly, PDC 7, without the cathepsin B cleavable sequence, also had no appreciable effect on cell proliferation (>100 uM), despite possessing the active DSA DNA alkylating moiety. Perhaps this PDC is either not being taken up into cells or not being broken down by proteases and peptidases in the cell. In both cases, the warhead may not be reaching the nucleus, the site of action of the duocarmycins. Cell lines that demonstrated appreciable levels of TF were sensitive to PDC 3. Interestingly, HCT116, which had relatively high TF expression and cathepsin B activity, demonstrated the greatest sensitivity to PDC 3, approaching similar potency to the payload 5. Excitingly, A549, H292, and 16HBE14o, which showed no detectable TF expression, appeared unaffected by PDC 3 up to 100 uM, suggesting that TF expression is required for the efficacy of PDC 3.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | 16HBE14o- cell | CVCL_0112 | ||
References
