Drug Information
General Information of This Drug
| Drug ID | DRG00075 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Ibuprofen | |||||
| Synonyms |
ibuprofen; 15687-27-1; 2-(4-Isobutylphenyl)propanoic acid; Motrin; Brufen; Advil; Nurofen; Dolgit; Liptan; Medipren; Nuprin; Anflagen; Buburone; Butylenin; Ibuprocin; Lamidon; Ebufac; Ibumetin; Mynosedin; Roidenin; Andran; Apsifen; Bluton; Epobron; Haltran; Nobfen; Nobgen; Trendar; Adran; PediaProfen; Rufen; 2-[4-(2-methylpropyl)phenyl]propanoic acid; Brufort; Ibuprohm; Inabrin; Nobfelon; Pantrop; Rebugen; Suspren; Tabalon; Burana; Anco; Urem; (RS)-Ibuprofen; Ibu-slo; Brufanic; Napacetin; Ibufen; Ibuprin; Profen; Ibren; Cap-Profen; Tab-Profen; Ibu-Tab; (+/-)-Ibuprofen; Novoprofen; Proartinal; Amersol; Dibufen; Dolgin; Dolocyl; Lidifen; Optifen; Ostofen; Panafen; Paxofen; Proflex; Quadrax; Uprofen; Carol; Midol; Rafen; Cobo; Ifen; 4-Isobutylhydratropic acid; (+-)-Ibuprofen; Artril 300; p-Isobutylhydratropic acid; duralbuprofen; Balkaprofen; Betaprofen; Butacortelone; Daiprophen; Ibuprofeno; Ibuprofenum; Jenaprofen; Algofen; Amelior; Amibufen; Antagil; Antalfene; Antarene; Antiflam; Artofen; Bruflam; Bufigen; Bukrefen; Buracaps; Citalgan; Combiflam; Dansida; Dentigoa; Dignoflex; Dolgirid; Dolmaral; Dolofen; Dolofort; Dologel; Dolomax; Doloren; Doltibil; Dorival; Duobrus; Dysdolen; Easifon; Exneural; Femadon; Femafen; Femapirin; Femidol; Fenspan; Fibraflex; Gelufene; Gynofug; Ibubest; Ibubeta; Ibucasen; Ibudolor; Ibuflamar; Ibugesic; Ibuhexal; Ibulagic; Ibuleve; Ibulgan; Ibumerck; Ibupirac; Junifen; Kratalgin; Lebrufen; Librofem; Malafene; Manypren; Mensoton; Neobrufen; Nerofen; Noalgil; Nobafon; Noritis; Novadol; Novogent; Nuprilan; Opturem; Oralfene; Ostarin; Paduden; Perofen; Ranofen; Relcofen; Rhinadvil; Sadefen; Salivia; Seclodin; Sednafen; Seklodin; Seskafen; Siyafen; Solpaflex; Sugafen; Suprafen; Syntofene; Tatanal; Unipron; Alaxan; Anafen; Artril; Bloom; Brofen; Bufeno; Bupron; Cesra; Cunil; Dalsy; Dolibu; Dolven; Duafen; Emflam; Eputex; Ergix; Faspic; Fendol; Gofen; Grefen; Ibudol; Ibufug; Ibugel; Ibugen; Ibular; Ibulav; Ibumed; Ibusal; Ibutid; Inflam; Inoven; Ipren; Irfen; Isodol; Kesan; Lopane; Melfen; Midol 200; Moment; Narfen; Ozonol; Provon; Rofen; Rufin; Rupan; Stelar; Tempil; Tofen; Tonal; Upfen; Zofen; Drin; Ibol; Pediatric Advil; Apo-Ibuprofen; Brufen Retard; Deep Relief; Dolo-Dolgit; Ibu-Attritin; Neo-Helvagit; Novo-Profen; Ibu-slow; Neo-Mindol; Dolofen-F; Donjust B; Nagifen-D; Dura-Ibu; Children's Advil; Motrin IB; Children's Motrin; Togal N; 4-Isobutyl-alpha-methylphenylacetic acid; ACHES-N-PAIN; Dularbuprofen; Caldolor; Ibu-Tab 200; Ibuprofene; Ibuprophen; Pedea; alpha-(4-Isobutylphenyl)propionic acid; Children's Ibuprofen; IP-82; Kontagripp Mono; Tabalon 400; Novo Dioxadol; Schmerz-Dolgit; Junior Strength Advil; 58560-75-1; Brufen 400; Emflam-200; DOLO PUREN; Hemagene Tailleur; Junior Strength Motrin; Act-3; Ak+C2278tren; Advil Liqui-Gels; Adex 200; Codral Period Pain; Junior Strength Ibuprofen; (+-)-Ibuprophen; Children's Elixsure; RD 13621; Am-Fam 400; IB-100; Alivium; (+-)-p-Isobutylhydratropic acid; Advil migraine; U-18,573; Perrigo ibuprofen; Hydratropic acid, p-isobutyl-; (4-Isobutylphenyl)-alpha-methylacetic acid; VUFB 9649; p-Isobutyl-2-phenylpropionic acid; MOTRIN MIGRAINE PAIN; (+-)-2-(p-Isobutylphenyl)propionic acid; UCB 79171; alpha-(p-isobutylphenyl)propionic acid; CCRIS 3223; CHEBI:5855; rac Ibuprofen; CHILDREN'S ADVIL-FLAVORED; HSDB 3099; UNII-WK2XYI10QM; Ibuprofene [INN-French]; Ibuprofenum [INN-Latin]; Advil Migraine Liqui-Gels; Racemic ibuprofen; WK2XYI10QM; Ibuprofeno [INN-Spanish]; alpha-p-Isobutylphenylpropionic acid; EINECS 239-784-6; Midol Liquid Gels; (+-)-alpha-Methyl-4-(2-methylpropyl)benzeneacetic acid; Benzeneacetic acid, .alpha.-methyl-4-(2-methylpropyl)-; NSC 256857; NSC-256857; BRN 2049713; alpha-Methyl-4-(2-methylpropyl)benzeneacetic acid; Ibuprofen sodium; U 18573; U-18573; DTXSID5020732; Ibu; 2-(4-ISOBUTYLPHENYL)PROPIONIC ACID; M01AE01; (+/-)-p-Isobutylhydratropic acid; 2-(p-Isobutylphenyl)propionic acid; MFCD00010393; NSC256857; CHEMBL521; 2-(4-Isobutyl-phenyl)-propionic acid; Acide (isobutyl-4-phenyl)-2 propionique; Benzeneacetic acid, alpha-methyl-4-(2-methylpropyl)-; DTXCID90732; alpha-Methyl-4-(isobutyl)phenylacetic acid; MLS000069733; (+/-)-2-(4-Isobutylphenyl)Propanoic Acid; Dolofin; Esprenit; R.D. 13621; Fenbid; Zafen; EC 239-784-6; COMBUNOX COMPONENT IBUPROFEN; REPREXAIN COMPONENT IBUPROFEN; VICOPROFEN COMPONENT IBUPROFEN; Ibuprofen [USAN:USP:INN:BAN:JAN]; SINE-AID IB COMPONENT IBUPROFEN; .alpha.-(p-isobutylphenyl)propionic acid; NCGC00015529-09; .alpha.-(4-Isobutylphenyl)propionic acid; Acide (isobutyl-4 phenyl)-2 propionique; SMR000058184; (+/-)-2-(p-isobutylphenyl)propionic acid; 4-Isobutyl-.alpha.-methylphenylacetic acid; Ibuprofene (INN-French); Ibuprofenum (INN-Latin); ADVIL ALLERGY SINUS COMPONENT IBUPROFEN; Ibuprofeno (INN-Spanish); IBUPROFEN (MART.); IBUPROFEN [MART.]; IBUPROFEN (USP-RS); IBUPROFEN [USP-RS]; CHILDREN'S ADVIL COLD COMPONENT IBUPROFEN; ADVIL CONGESTION RELIEF COMPONENT IBUPROFEN; CHILDREN'S MOTRIN COLD COMPONENT IBUPROFEN; Advil, Children's; .alpha.-Methyl-4-(2-methylpropyl)benzeneacetic acid; IBUPROFEN (EP MONOGRAPH); IBUPROFEN (USP IMPURITY); IBUPROFEN [EP MONOGRAPH]; IBUPROFEN [USP IMPURITY]; IBUPROFEN (USP MONOGRAPH); IBUPROFEN [USP MONOGRAPH]; Midol IB Cramp Relief; Bayer Select Pain Relief; Ibuprofen (USAN:USP:INN:BAN:JAN); 2-(4-isobutylphenyl)propanoate; Motrin IB Gelcaps; rac Ibuprofen (1.0 mg/mL in Methanol); alpha-(4-Isobutylphenyl)propionate; CAS-15687-27-1; Motrin (TN); Advil (TN); Children's Elixsure IB; SR-01000000214; Actrogel; Cedaprin; Ibupril; IbuprofenJunior; Ibuprox; Probufen; Proprinal; Aktren; ibuprofeninfants; Ibutab; kidgets infants; MotrinInfants; DragonTabs; Ibux; Advil Liquigels; Childrens Advil; CounterActIB; Dolex Children; Dover Addaprin; Ibuprofen Mini; Ibuprofen Minis; Ibuprofen Oral; ibuprofen tablet; Infants Motrin; profen ibinfants; Uline Ibuprofen; Welby Ibuprofen; Xpect Ibuprofen; Ibuprofen Ib; Infants Advil; Medique Iprin; Flex-prin; CDT-ibuprofen; Childrens Motrin; Dolex Flex; Equate Ibuprofen; IbuprofenSoftgels; Leader Ibuprofen; MFCD00069290; Nurofen Meltlets; Rexall Ibuprofen; Zee Ibutab; Profen Ib; Aramark Ibuprofen; Assured Ibuprofen; Ibuprofen Tablets; IbuprofenChildrens; Infants Ibuprofen; Sunmark Ibuprofen; Topcare Ibuprofen; AeroTab Ibuprofen; CareOne Ibuprofen; IbuprofenDye Free; IbuprofenDye-Free; Fast Pain Relief; Rx Act Ibuprofen; (y)-Ibuprofen; Equaline Ibuprofen; Ibuprofen Migraine; Ibuprofen Softgels; Ibuprofen Thompson; profen IBchildrens; Redicare Ibuprofen; Ibuprofen 200mg; Ibuprofen,(S); (+) ibuprofen; Childrens Ibuprofen; Unishield Ibuprofen; Care One Ibuprofen; Circle K Ibuprofen; Dye Free Ibuprofen; Dye-Free Ibuprofen; IBU Tabs; Ibuprofen (Advil); MoreBrand Ibuprofen; Acide (isobutyl-4-phenyl)-2 propionique [French]; (?)-Ibuprofen; CAREALL Ibuprofen; Ibuprofen 200; Motrin IB Migraine; Dg Health Ibuprofen; Ibuprofen [USAN]; Moorebrand Ibuprofen; dye-free pain relief; Ibuprofen 200 mg; Medi-first Ibuprofen; Basic Care Ibuprofen; EPROBRON; Good Sense Ibuprofen; IBUOROFEN; IBUPAK; IBUPFROFEN; Otis Clapp Ultraprin; Sound Body Ibuprofen; Sunmark Ibuprofen Ib; Caring mill ibuprofen; Ibuprofen (NSAID); Up and Up Ibuprofen; p-Isobutylhydratropate; Health Mart Ibuprofen; Pain Relief Ibuprofen; Combunox (Salt/Mix); I buprofen 200 mg; IBUPROFEN CA; KOSHER MEDS; (A+/-)-Ibuprofen; betr remedies ibuprofen; Clear Choice Ibuprofen; Crane Safety Ibuprofen; Henry Schein Ibuprofen; Medique at Home Iprin; Members Mark Ibuprofen; sunmark ibuprofeninfants; Direct Safety Ibuprofen; Harris Teeter Ibuprofen; INFANT'S ADVIL; Spectrum_000849; leader infants ibuprofen; equate ibuprofenchildrens; IBUPROFEN [INN]; IBUPROFEN [JAN]; Opera_ID_554; care one ibuprofeninfants; IBUPROFEN [MI]; Preferred Plus Ibuprofen; rx act ibuprofenchildrens; Signature Care Ibuprofen; Ibuprofen, Caseys 4good; VALUMEDS IBUPROFEN; Childrens Ibuprofen fruit; Childrens Ibuprofen grape; IBUPROFEN [HSDB]; Ibuprofen Mini 200 mg; Sunmark ibuprofenchildrens; Topcare ibuprofenchildrens; Family Wellness Ibuprofen; First Aid Only Ibuprofen; Handy Solutions Ibuprofen; Health Mart Ibuprofen Ib; IBUPROFEN200 MG; Lil Drug Store Ibuprofen; Physicians Care Ibuprofen; Spectrum2_000129; Spectrum3_000465; Spectrum4_000015; Spectrum5_000862; Topcare Infants Ibuprofen; Welby Childrens Ibuprofen; Childrens Ibuprofen 100; IBUPROFEN [VANDF]; IBUPROFEN Tablet coated; Ibuprofen Tablets, coated; IbuprofenImmediate Release; Dye-Free Ibuprofen 200; Equaline ibuprofenchildrens; Medi-first Plus Ibuprofen; WLN: QVY&R DIY; careone childrens ibuprofen; CBMicro_005634; COMPRESSO IBU 66; Green Guard Ibupro Relief; IBUPROFEN 100; IBUPROFEN 250; Leader Childrens Ibuprofen; Care One ibuprofenchildrens; COMPRESSO IBU 66S; Epitope ID:139973; Good Sense ibuprofeninfants; p-isobutyl-hydratropic acid; Topcare Childrens Ibuprofen; I 4883; basic care infants ibuprofen; Cambridge id 5152402; family wellness ibuprofen ib; IBUPROFEN [WHO-DD]; IBUPROFEN [WHO-IP]; SCHEMBL3001; Equaline Childrens Ibuprofen; Ibuprofen Capsules 200 mg; Kirkland Signature Ibuprofen; 24/7 Life IBUPROFEN; Berkley and Jensen Ibuprofen; McKesson Ibuprofen 200 mg; 365 everday value ibuprofen; Childrens Ibuprofen bubblegum; Dye Free Childrens Ibuprofen; Green Guard Ibuprofen Relief; Isobutylpropanoicphenolic acid; Lopac0_000691; BSPBio_002170; Concentrated ibuprofen infants; Good Sense IbuprofenChildrens; Help I Have An Aching Body; IBUPROFEN [EMA EPAR]; KBioGR_000389; KBioSS_001329; 365 everyday value ibuprofen; APROFENREGULAR STRENGTH; DG Health childrens ibuprofen; Ibuprofen tablet, film-coated; MLS001146965; p-Isobutyl-2-phenylpropionate; Pain Relief Anti Inflammatory; sunmark childrens ibuprofen ib; BIDD:GT0050; COMPRESSO IBU 85 PAR; DivK1c_000887; Harris Teeter IbuprofenInfants; SPECTRUM1500347; Basic Care Childrens Ibuprofen; caring mill childrens ibuprofen; core values childrens ibuprofen; Easy Care First Aid Ibuprofen; Good Sense Childrens Ibuprofen; Ibuprofen, >=98% (GC); Preferred Plus Ibuprofen 200; Quality Choice Ibuprofen 200; Sound Body Childrens Ibuprofen; SPBio_000178; Up and Up Childrens Ibuprofen; Ibuprofen (JP17/USP/INN); Ibuprofen, 1mg/ml in Methanol; Pain Reliever / Fever Reducer; alpha-p-Isobutylphenylpropionate; GTPL2713; Health Mart Childrens Ibuprofen; Kirkland Signature Ibuprofen Ib; Motrin IB Gelcaps (Salt/Mix); Pain Relief-Ibuprofen 200 mg; parents choice infants ibuprofen; IBUPROFEN [ORANGE BOOK]; Ibuprofen Tablets USP, 200mg; 2-p-isobutylphenylpropionic acid; Good Neighbor Pharmacy Ibuprofen; Signature Care Infants Ibuprofen; Toxicology Saliva Collection Kit; HEFN
Click to Show/Hide
|
|||||
| Target(s) | Prostaglandin G/H synthase 2 (PTGS2) | Target Info | ||||
| Structure |
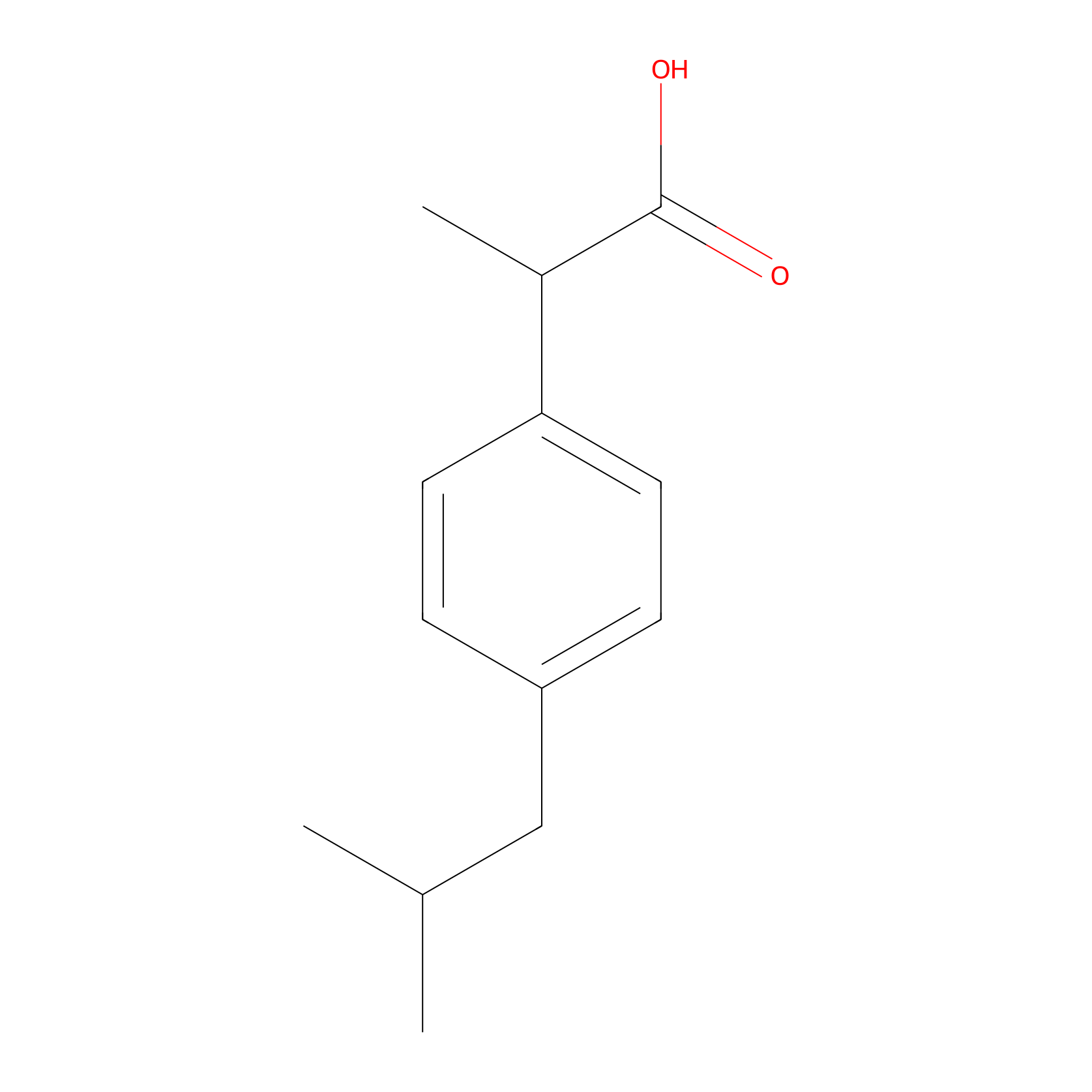
|
|||||
| Formula |
C13H18O2
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 206.28 | ||||
| Lipid-water partition coefficient (xlogp) | 3.5 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 2 | |||||
| Rotatable Bond Count (rotbonds) | 4 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC(C)CC1=CC=C(C=C1)C(C)C(=O)O
|
|||||
| InChI |
InChI=1S/C13H18O2/c1-9(2)8-11-4-6-12(7-5-11)10(3)13(14)15/h4-7,9-10H,8H2,1-3H3,(H,14,15)
|
|||||
| InChIKey |
HEFNNWSXXWATRW-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
2-[4-(2-methylpropyl)phenyl]propanoic acid
|
|||||
The activity data of This Drug
| Standard Type | Value | Disease Model | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | >50 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | >50 uM | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 uM | HPV-related endocervical adenocarcinoma | Bel7402/5-FU cell | CVCL_5493 | [2] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 uM | Hepatoma | Bel-7402 cell | CVCL_5492 | [2] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 uM | Cervical carcinoma | L02 cell | CVCL_6926 | [2] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
IBF-HYD-GFFpY [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 38.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 400 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 42.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 400 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 75.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 200μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 80.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 200μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 82.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 200μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 85.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 90.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 90.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 90.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 95.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | del | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 95.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 400 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 97.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 25μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 97.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 98.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 25μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 25μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 16 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 μM | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 17 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | del | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 18 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 24 h | ||||
| Administration Dosage | del | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
Linear arginineglycineaspartic acid (RGD) - Ibuprofen conjugate 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 11 ± 4.1% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 45 ± 2.0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
Linear arginineglycineaspartic acid (RGD) - Ibuprofen conjugate 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 28 ± 3.01% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 35 ± 2.1% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
Linear asparagineglycinearginine (NGR) 2 - Ibuprofen conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Cell inhibition rate | 36 ± 3.2% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 44 ± 2.4% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous cystadenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 46 ± 0.7% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
XNRG - Ibuprofen conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 40 ± 3.6% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous cystadenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 59 ± 1.1% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Cell inhibition rate | 61 ± 0.8% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
References
