Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02054
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
IBF-HYD-GFFpY
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
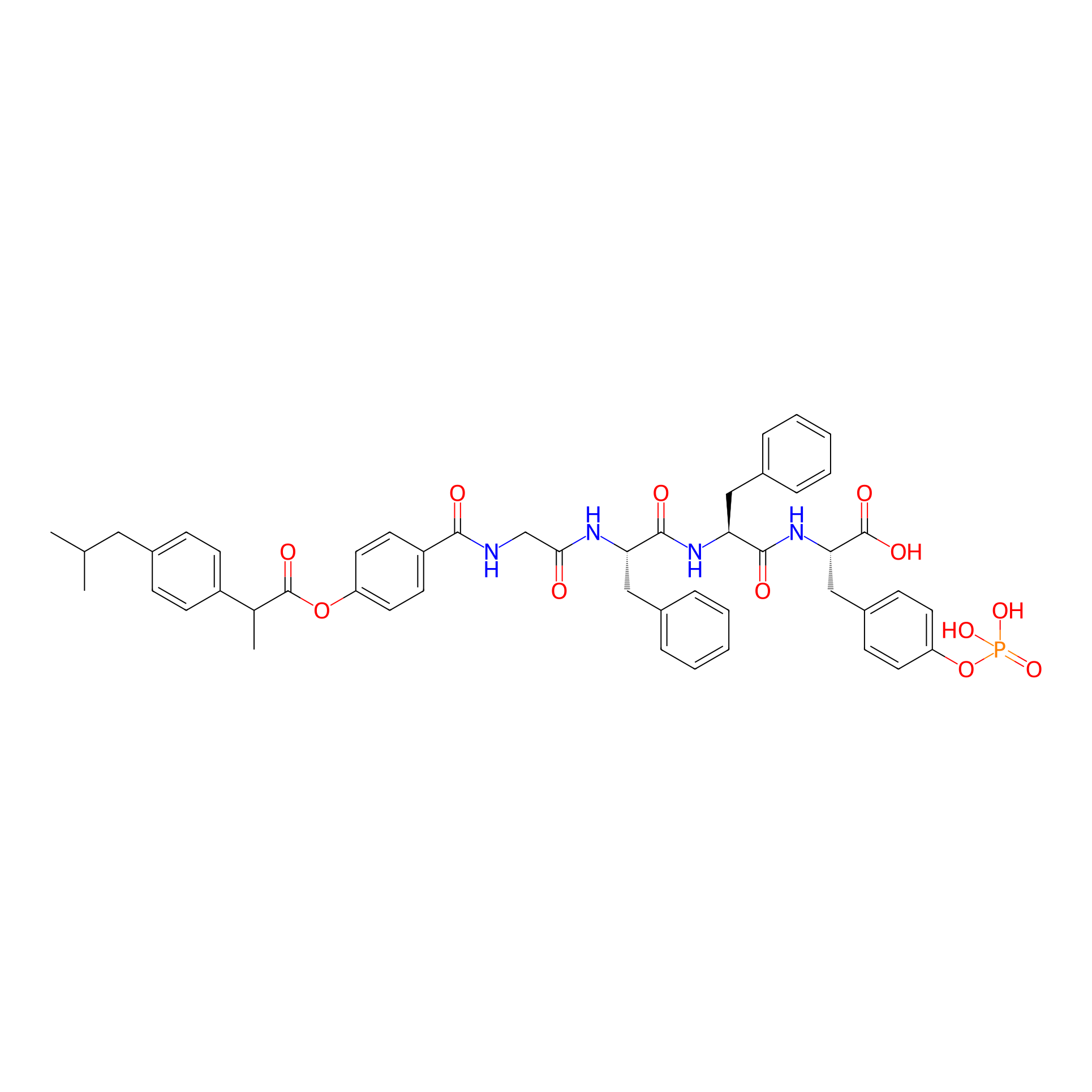
|
|||||
| Peptide Name |
GFFpY
|
Peptide Info | ||||
| Drug Name |
Ibuprofen
|
Drug Info | ||||
| Therapeutic Target |
Prostaglandin G/H synthase 2 (PTGS2)
|
Target Info | ||||
| Linker Name |
4-Hydroxybenzoic acid
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical molecules
|
|||||
| Modified Segment |
Phosphoric acid
|
|||||
| Formula |
C49H53N4O12P
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 920.953 | ||||
| Lipid-water partition coefficient (xlogp) | 5.0626 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 7 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 9 | |||||
| Rotatable Bond Count (rotbonds) | 22 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
38.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 400 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
42.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 400 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
75.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 200μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
80.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 200μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
82.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 200μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
85.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
90.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
90.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
90.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 100 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
95.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | del | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
95.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 400 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
97.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 25μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
97.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
98.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 25μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
100.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 25μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 16 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
100.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 50 μM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Monocytic-macrophage leukemia | RAW264.7 cell | CVCL_0493 | ||
| Experiment 17 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
100.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | del | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 83332 | ||
| Experiment 18 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Uveitis | ||||
| Efficacy Data | Cell viability |
100.00%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | del | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
In vivo self-assembly of small molecules offers an excellent opportunity for targeted and long-term accumulation of a therapeutic agent at the lesion site. Here we demonstrate the strategy of enzyme-instructed self-assembly (EISA) by designing a phosphorylated peptide-drug (IBF-HYD-GFFpY) precursor through the ester bond to release active drugs at the target site. Meanwhile, the in vivo assembly can be achieved by the catalysis of alkaline phosphatase (ALP) in the tear fluid for ocular drug delivery efficiently. The in vitro enzymatic experiments indicate that the dephosphorylation of IBF-HYD-GFFpY occurs firstly with the yield of IBF-HYD-GFFY which subsequently self-assembles into the supramolecular hydrogel to afford sustained drug release over 96 h. In the treatment of lipopolysaccharide (LPS)-activated Raw 264.7 macrophages, IBF-HYD-GFFpY exerts the more potent anti-inflammatory efficacy than that of free ibuprofen (IBF) at the concentration of 200 μM. Moreover, the aqueous solution of IBF-HYD-GFFpY via topical instillation hardly causes ocular irritation, and displays longer precorneal retention compared to the conventional eye drop formulation. In addition, in the in vivo study, a rabbit model of endotoxin-induced uveitis (EIU) evidences the comparable therapeutic efficacy of IBF-HYD-GFFpY eye drops with that of clinically used 0.1 wt% diclofenac (DIC) sodium eye drops by the reduction of macrophage and leukocyte influx. This work, in situ EISA in the tear microenvironment directing in vivo self-assembly of small molecules, may guide a powerful approach for developing enzymatic self-assembled molecules as an efficient delivery system of ocular drugs.
Click to Show/Hide
|
||||
| Description |
We assessed the in vitro cytotoxicity of IBF-HYD-GFFpY against Raw 264.7 macrophages, L-929 cells and HCEC cells by MTT assay. Fig. 3A clearly shows that both of IBF and IBF-HYD-GFFpY caused little cytotoxicity against all cell lines at concentrations below 200 μM. When the concentration of IBF-HYD-GFFpY was up to 400 μM, the viabilities of HCEC cells and Raw 264.7 macrophages declined significantly, indicating that the proposed IBF-HYD-GFFpY conjugate might cause apparent cytotoxicities towards HCEC cells and Raw 264.7 macrophages at concentrations above 200 μM.
Click to Show/Hide
|
||||
| In Vitro Model | Mycobacterium tuberculosis infection | Mycobacterium tuberculosis | 1773 | ||
References
