Drug Information
General Information of This Drug
| Drug ID | DRG00121 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Naproxen | |||||
| Synonyms |
NAPROXEN; 22204-53-1; (S)-Naproxen; Naprosyn; (+)-Naproxen; Naproxene; Equiproxen; (S)-2-(6-methoxynaphthalen-2-yl)propanoic acid; Aleve; (S)-(+)-Naproxen; d-Naproxen; Ec-Naprosyn; (S)-(+)-2-(6-Methoxy-2-naphthyl)propionic acid; (+)-(S)-Naproxen; Laraflex; Naprosyne; Naproxeno; Naproxenum; Nycopren; Calosen; Naixan; Reuxen; Bonyl; Axer; (2S)-2-(6-methoxynaphthalen-2-yl)propanoic acid; Novonaprox; Acusprain; Anexopen; Apronax; Artagen; Arthrisil; Artrixen; Artroxen; Atiflan; Bipronyl; Clinosyn; Danaprox; Flexipen; Genoxen; Lefaine; Leniartil; Nafasol; Nalyxan; Napflam; Naposin; Naprius; Veradol; Congex; Daprox; Fuxen; Napmel; Napren; Naxen; Naxyn; Xenar; Dysmenalgit N; Apo-Naproxen; naproxen(+); Floginax; Napratec; Naprontag; Naxopren; Pronaxen; Rheumaflex; Saritilron; Sinartrin; Soproxen; Anaprox; Flexen; Headlon; Napxen; Narocin; Noflam; Patxen; Prafena; Priaxen; Rahsen; Sinton; Sutolin; Sutony; Tohexen; Traumox; Velsay; Vinsen; Narma; Naxid; Roxen; Anax; Flanax Forte; Naprosyn LLE; Napren E; Proxen LE; Naxen F; Proxen LLE; U-Ritis; RS-3540; (S)-2-(6-Methoxy-2-naphthyl)propionic acid; (+)-2-(6-Methoxy-2-naphthyl)propionic acid; Naprosyn LLE Forte; Naproxi 250; Naproxi 500; (+)-2-(Methoxy-2-naphthyl)-propionic acid; (S)-6-Methoxy-alpha-methyl-2-naphthaleneacetic acid; Naxyn 250; Naxyn 500; Xenar-CR; (S)-2-(6-Methoxy-2-naphthyl)propanoic acid; d-2-(6-Methoxy-2-naphthyl)propionic acid; (+)-2-(Methoxy-2-naphthyl)-propionsaeure; CCRIS 5265; CHEBI:7476; Diocodal; Naprolag; Naprosine; Naprosy; Prexan; CG 3117; Apo-Napro-NA; (+)-(S)-6-Methoxy-alpha-methyl-2-naphthaleneacetic acid; HSDB 3369; Naproxene [INN-French]; Naproxenum [INN-Latin]; Naproxeno [INN-Spanish]; EINECS 244-838-7; UNII-57Y76R9ATQ; (S)-(+)-6-Methoxy-alpha-methyl-2-naphthaleneacetic acid; Dysmenalgit; NSC-750183; NSC-757239; Proxen; RS 3540; 57Y76R9ATQ; DTXSID4040686; PN400 COMPONENT NAPROXEN; d-2-(6'-Methoxy-2'-naphthyl)-propionsaeure; CHEMBL154; (+)-6-Methoxy-alpha-methyl-2-naphthaleneacetic acid; Propionic acid, 2-(6-methoxy-2-naphthyl)-, (+)-; (alphas)-6-methoxy-alpha-methyl-2-naphthaleneacetic acid; 2-Naphthaleneacetic acid, 6-methoxy-alpha-methyl-, (+)-; 2-Naphthaleneacetic acid, 6-methoxy-alpha-methyl-, (S)-; Duk; VIMOVO COMPONENT NAPROXEN; DTXCID2020686; Methoxypropiocin; (2S)-2-(6-methoxy-2-naphthyl)propanoic acid; EC 244-838-7; Apranax; Naprium; Naprux; MFCD00010500; Naproxen [USAN:USP:INN:BAN:JAN]; NSC 750183; NSC 757239; 2-Naphthaleneacetic acid, 6-methoxy-.alpha.-methyl-, (S)-; Naproxene (INN-French); Naproxenum (INN-Latin); 2-Naphthaleneacetic acid, 6-methoxy-.alpha.-methyl-, (.alpha.S)-; CAS-22204-53-1; Naproxeno (INN-Spanish); NAPROXEN (MART.); NAPROXEN [MART.]; NAPROXEN (USP-RS); NAPROXEN [USP-RS]; (S)-2-(6-Methoxy-naphthalen-2-yl)-propionic acid; NAPROXEN (EP MONOGRAPH); NAPROXEN [EP MONOGRAPH]; (S)-6-Methoxy-.alpha.-methyl-2-naphthaleneacetic acid; NAPROXEN (USP MONOGRAPH); NAPROXEN [USP MONOGRAPH]; Equiproxen (Veterinary); Naproxen (USAN:USP:INN:BAN:JAN); S-NAPROXEN; Panoxen; Proxine; (S)-6-Methoxy-alpha-methyl-2-naphthaleneacetate; Mnpa; Naproxen oral; SR-01000075977; DL Naproxen; Equiproxen Granules; Naprosyn (TN); Naproxen,(S); NPS; Prestwick_349; (+)-2-(Methoxy-2-naphthyl)-propionsaeure [German]; d-2-(6'-Methoxy-2'-naphthyl)-propionsaeure [German]; Naproxendelayed release; EC-NAPROXEN; 6-methoxy-alpha-methyl-2-naphthaleneacetic acid; Spectrum_000977; (R)-6-Methoxy-a-methyl-2-naphthaleneacetic acid; 4or0; 4po0; NAPROXEN [HSDB]; NAPROXEN [USAN]; (S)-(+)-Naproxene; NAPROXEN [INN]; NAPROXEN [JAN]; Naproxen Delayed Release; Naproxen Oral Suspension; NAPROXEN [MI]; NAPROXEN [VANDF]; Prestwick0_000791; Prestwick1_000791; Prestwick2_000791; Spectrum2_001043; Spectrum3_000514; Spectrum4_000069; Spectrum5_001327; (+)NAPROXEN; Equiproxen 10% Solution; Naproxen2-(6-Methoxy-naphthalen-2-yl)-propionic acid; Epitope ID:139974; NAPROXEN [WHO-DD]; UPCMLD-DP001; 2-(6-Methoxy-naphthalen-2-yl)-propionic acid(Naproxen); SCHEMBL3046; Lopac0_000792; BSPBio_002067; KBioGR_000597; KBioSS_001457; BIDD:GT0062; DivK1c_000242; NAPROXEN [GREEN BOOK]; SPECTRUM1500425; Naproxen (JP17/USP/INN); Naproxen, 1mg/ml in Methanol; SPBio_000966; SPBio_002861; NAPROXEN [ORANGE BOOK]; GTPL5230; UPCMLD-DP001:001; CMWTZPSULFXXJA-VIFPVBQESA-; HMS500M04; KBio1_000242; KBio2_001457; KBio2_004025; KBio2_006593; KBio3_001567; G02CC02; M01AE02; M02AA12; Naproxen 1.0 mg/ml in Methanol; NINDS_000242; HMS1920P13; HMS2089N21; HMS2091H12; HMS3649M13; HMS3886C15; Pharmakon1600-01500425; NAPROXEN COMPONENT OF VIMOVO; Tox21_301953; BDBM50339185; CCG-40130; NSC750183; NSC757239; AKOS005267223; AC-1363; DB00788; ME-0100; SDCCGSBI-0050769.P005; IDI1_000242; Naproxen 100 microg/mL in Acetonitrile; NCGC00016759-01; NCGC00016759-02; NCGC00016759-03; NCGC00016759-29; NCGC00016759-38; NCGC00021127-01; NCGC00161591-01; NCGC00255562-01; HY-15030; (+)-2-(Methoxy-2-naphthyl)-propionsaure; Naproxen, meets USP testing specifications; SBI-0050769.P004; Sallus Pain Relief Collection With Naproxen; AM20060551; M1021; NS00098611; S5177; Naproxen, VETRANAL(TM), analytical standard; (2S)-2-(6-Methoxynaphth-2-yl)propanoic acid; (S)-2-(6-methoxy-2-naphthyl)-propionic acid; (S)-2-(6-methoxynaphthalen-2-yl)propanoicacid; C01517; d-2-(6'-methoxy-2'-naphthyl)-propionic acid; D00118; EN300-139795; O10203; (2S)-2-(6-methoxy(2-naphthyl))propanoic acid; AB00052049-04; AB00052049_05; ()-2-(METHOXY-2-NAPHTHYL)-PROPIONIC ACID; ()-2-(METHOXY-2-NAPHTHYL)-PROPIONSAEURE; (+)(S)2-(6-methoxy-2-naphthyl)-propionic acid; 2-(6-Methoxy-2-naphthyl)propanoic acid , (+)-; SR-01000003110; Q-201447; Q1215575; SR-01000003110-5; SR-01000075977-3; SR-01000075977-4; (+)-6-Methoxy-.alpha.-methyl-2-napthaleneacetic acid; BRD-K59197931-001-02-9; BRD-K59197931-001-03-7; BRD-K59197931-236-09-6; SR-01000075977-10; (+)-6-Methoxy-.alpha.-methyl-2-naphthaleneacetic acid; (S)-(+)-2-(6-methoxy-naphthalen-2-yl)-propionic acid; (S)-6-Methoxy-.alpha.-methyl-2-naphthalene acetic acid; 2-naphthaleneacetic acid, 6-methoxy-alpha-methyl, (S)-; 2-naphthleneacetic acid, 6-methoxy-alpha-methyl-, (+)-; Naproxen, British Pharmacopoeia (BP) Reference Standard; Naproxen, European Pharmacopoeia (EP) Reference Standard; PROPIONIC ACID, 2-(METHOXY-2-NAPHTHYL)-, ()-; Z1695772815; (S)-(+)-6-Methoxy-alpha-methyl-2-naphthaleneacetic acid, 98%; 2-Naphthaleneacetic acid, 6-methoxy-.alpha.-methyl-, (+)-; Naproxen, United States Pharmacopeia (USP) Reference Standard; (+)-(S)-6-METHOXY-.ALPHA.-METHYL-2-NAPHTHALENEACETIC ACID; Naproxen, Pharmaceutical Secondary Standard; Certified Reference Material; (S)-(+)-2-(6-Methoxy-2-naphthyl)propionic Acid;(S)-(+)-6-Methoxy-alpha-methyl-2-naphthaleneacetic Acid; 131991-52-1; InChI=1/C14H14O3/c1-9(14(15)16)10-3-4-12-8-13(17-2)6-5-11(12)7-10/h3-9H,1-2H3,(H,15,16)/t9-/m0/s1
Click to Show/Hide
|
|||||
| Target(s) | Prostaglandin G/H synthase 1 (COX-1) | Target Info | ||||
| Structure |
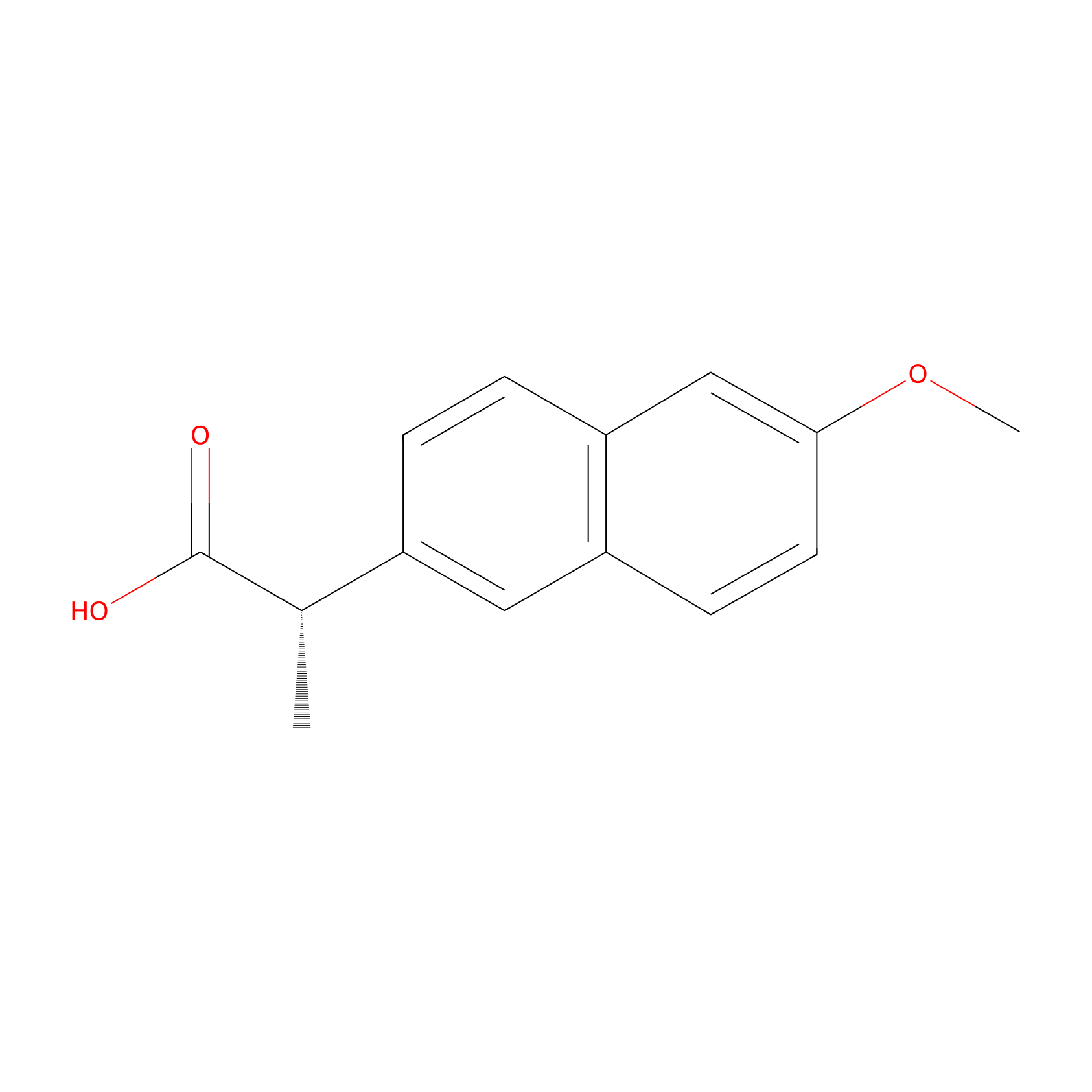
|
|||||
| Formula |
C14H14O3
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 230.26 | ||||
| Lipid-water partition coefficient (xlogp) | 3.3 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | |||||
| Rotatable Bond Count (rotbonds) | 3 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC(C1=CC2=C(C=C1)C=C(C=C2)OC)C(=O)O
|
|||||
| InChI |
InChI=1S/C14H14O3/c1-9(14(15)16)10-3-4-12-8-13(17-2)6-5-11(12)7-10/h3-9H,1-2H3,(H,15,16)/t9-/m0/s1
|
|||||
| InChIKey |
CMWTZPSULFXXJA-VIFPVBQESA-N
|
|||||
| IUPAC Name |
(2S)-2-(6-methoxynaphthalen-2-yl)propanoic acid
|
|||||
The activity data of This Drug
| Standard Type | Value | Disease Model | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Inhibitory Concentration (IC50) | 2.385 mM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [1] | |
| Half Maximal Inhibitory Concentration (IC50) | >50 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [2] | |
| Half Maximal Inhibitory Concentration (IC50) | >50 uM | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | [2] | |
| Half Maximal Inhibitory Concentration (IC50) | >400 uM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [3] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
Linear arginineglycineaspartic acid (RGD) - Naproxen conjugate 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 28 ± 2.3% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 41 ± 2.0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
Linear arginineglycineaspartic acid (RGD) - Naproxen conjugate 4 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 12.00% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 57 ± 0.4% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
Linear asparagineglycinearginine (NGR) 1 - Naproxen conjugate 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Cell inhibition rate | 32 ± 4.1% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 48 ± 1.1% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous cystadenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 53 ± 1.4% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
Linear asparagineglycinearginine (NGR) 1 - Naproxen conjugate 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Invasive breast carcinoma | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [4] | ||||
| Efficacy Data | Cell inhibition rate | 0% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human fibroblast cells | Homo sapiens | ||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian endometrioid adenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 31 ± 2.1% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Cell inhibition rate | 66 ± 0.5% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Ovarian serous cystadenocarcinoma | ||||
| Efficacy Data | Cell inhibition rate | 68 ± 0.9% | |||
| Evaluation Method | MTT assay | ||||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 μM | ||||
| MOA of PDC |
The two tripeptide sequences, arginine-glycine-aspartic acid (RGD) and asparagine-glycine-arginine (NGR) motifs have been identified based on phage display studies and they have been used widely in the field of targeted drug delivery. RGD is a well-known peptide sequence for targeting integrin receptors and can bind to avβ3 and avβ5 integrin receptors subunits, which are overexpressed in the angiogenesis process of cancer cells. Because av integrin is overexpressed on the surface of cancer cells, an integrin ligand can be used as a targeting system for cancer treatment. RGD peptide conjugated with cytotoxic agents (RGD-drug conjugates) is likely to exhibit a tumor-targeting and thus antiangiogenic synergetic effect. During the last few years, a number of RGD-cytotoxic drugs have been developed and showed promising activities in vitro and in vivo. Conjugation of camptothecin with RGD is an example for improving the therapeutic index of an antitumoral drug.[20c] Synthesis of dimeric RGD peptide-paclitaxel conjugate is another successful example of targeted drug delivery. Other motif that has been used for tumor targeting is NGR tripeptide. This sequence can bind to CD13 that is specially overexpressed in tumor blood vessels and is involved in angiogenesis, invasion, and metastasis. Because RGD is a peptide tag which targets the process of angiogenesis and NGR also targets tumor blood vessels, we decided to synthesize the conjugated forms of two famous NSAIDs, naproxen, and ibuprofen, with these two tripeptides. To investigate the impact of possible steric hindrance due to the attachment of the drug to the peptide, a linear six-carbon segment (hexanoic acid) was also used as a spacer.
Click to Show/Hide
|
||||
| Description |
NGR conjugate forms of both ibuprofen and naproxen showed improved activity when they were tested against SKOV-3 cell line which is positive for APN/CD13. Interestingly, both ibuprofen and naproxen show increased activity against this cell line when a six-carbon spacer is used for their conjugation to NGR. This is probably due to the less steric hindrance for NGR interaction with its binding protein on the cell surface. Ibuprofen-spacer-NGR and naproxen-spacer-NGR showed the same pattern of increased activity against HT-1080 tumor cells which this cell line show high expression of CD13. Surprisingly, NGR conjugates of both drugs without spacer did not show improved activity compared to the nonconjugated forms against this cell line. Therefore, it could be speculated that HT-1080 cells are more sensitive to the steric hindrance for interaction between NGR and its binding protein. None of the conjugates of ibuprofen and naproxen with or without spacer showed significantly improved activity against A2780 (as a tumor cell with normal RGD-binding protein) and OVCAR3. Therefore, it could be inferred again that the RGD motif is not qualified as a targeting tool for ibuprofen and naproxen.
Click to Show/Hide
|
||||
| In Vitro Model | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | ||
References
