Drug Information
General Information of This Drug
| Drug ID | DRG00296 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Zidovudine | |||||
| Synonyms |
zidovudine; Azidothymidine; 30516-87-1; 3'-Azido-3'-deoxythymidine; Retrovir; AZT; Thymidine, 3'-azido-3'-deoxy-; Zidovudinum; Compound S; Zidovudina; ZIDOVUDINE [AZT]; zidovudin; Trizivir; BW A509U; Zidovudinum [Latin]; 3'-Deoxy-3'-azidothymidine; BWA509U; BW-A509U; ZDV; BW-A-509U; DRG-0004; Azidothymidine (AZT); Aztec; CCRIS 105; 3'-azt; HSDB 6515; 3'-Azidothymidine; UNII-4B9XT59T7S; NSC 602670; 4B9XT59T7S; DTXSID8020127; CHEBI:10110; 1-[(2R,4S,5S)-4-azido-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione; BW-A 509U; MFCD00006536; 1-[(2R,4S,5S)-4-azido-5-(hydroxymethyl)oxolan-2-yl]-5-methyl-1,2,3,4-tetrahydropyrimidine-2,4-dione; CHEMBL129; NSC-602670; Azitidin; 1-(3-Azido-2,3-dideoxy-beta-D-ribofuranosyl)thymine; DTXCID60127; MLS000028548; 1-((2R,4S,5S)-4-AZIDO-5-(HYDROXYMETHYL)TETRAHYDROFURAN-2-YL)-5-METHYLPYRIMIDINE-2,4(1H,3H)-DIONE; AZT Antiviral; COMBIVIR COMPONENT ZIDOVUDINE; TRIZIVIR COMPONENT ZIDOVUDINE; AZT, Antiviral; Zidovudinum (Latin); 1-(3-Azido-2,3-dideoxy-beta-D-ribofuranosyl)-5-methylpyrimidine-2,4-(1H,3H)-dione; AZT (Antiviral); Zidovudine [USAN:USP:INN:BAN:JAN]; NCGC00023945-05; SMR000058351; Zidovudina [Spanish]; ZIDOVUDINE (IARC); ZIDOVUDINE [IARC]; Antiviral AZT; ZIDOVUDINE (MART.); ZIDOVUDINE [MART.]; ZIDOVUDINE (USP-RS); ZIDOVUDINE [USP-RS]; Timazid; 399024-19-2; ZIDOVUDINE (EP IMPURITY); ZIDOVUDINE [EP IMPURITY]; ZIDOVUDINE (EP MONOGRAPH); ZIDOVUDINE [EP MONOGRAPH]; ZIDOVUDINE (USP MONOGRAPH); ZIDOVUDINE [USP MONOGRAPH]; Propolis+AZT; 3'-Azido-2',3'-Dideoxythymidine; Zidovudine (USAN:USP:INN:BAN:JAN); Retrovir(TM); AZT & Li & EPO; Retrovir (TN); 3' Azido 3' deoxythymidine; Cpd S; Intron A & AZT; Racemic Liposomal AZT; Liposomal AZT-SN-1; Liposomal AZT-SN-3; Zidovudine+PRO 140; PC-SOD+AZT; 3' Azido 2',3' Dideoxythymidine; AZT & srCD4; AZT & rIFN.alpha.2; AZT & rsT4; rIFN-beta seron & AZT; 3'-Azido-3'-deoxythymidine (AIDS); AZT & EPO; AZT & GM-CSF; AZT & HPA; AZT & sCD4; AZT & SST; zudovidine; Aziodothymidine; AZT & Li & GM-CSF; AZT+PRO 140; Met-SDF-1.beta. & AZT; AZT & Li & IL-1; AZT & Li & IL-6; AZT & IL-1; AZT & IL-2; AZT & IL-6; AZT & Interferon-.alpha.-2; AZT & Concanavalin A (ConA); AZT & Lymphoblastoid Interferon; AZT & PM-19; Met-SDF-1.beta. & Zidovudine; 4lhm; 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione (AZT); 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione [AZT]; AZT & rsCD4 & rIFN.alpha.A; 3'-azido-3'-deoxythymidine, AZT; DS-4152 & AZT; 1-((2R,4R,5S)-4-azido-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione; 1-[(2R,4S,5S)-4-azido-5-(hydroxymethyl)tetrahydrofuran-2-yl]-5-methyl-pyrimidine-2,4-dione; 5-methyl-1-[rac-(2R,4S,5S)-4-azido-5-(hydroxymethyl)tetrahydrofuran-2-yl]pyrimidine-2,4-dione; AZT & Colony-stimulating factor 2; AZT & NP (from PHCA or HSA); Zidovudine; 1-(3-Azido-2,3-dideoxy-ss-d-erythro-pentofuranosyl)-5-methylpyrimidine-2,4(1H,3H)-dione; Zidovudine (GR 63367X); BP Zidovudine and Lamivudine Impurity Standard; 3''-azido-thymidine; K7 [P Ti2 W10 O40]; Zidovudine & IFNL1; Zidovudine & IFNL2; Zidovudine & IFNL3; Zidovudine (Retrovir); COMPOUND-S; Spectrum_001348; AZT & CD4(178)-PE 40; AZT & IFN.alpha.; Zidovudine & IL-29; ZIDOVUDINE [MI]; Zidovudine & IL-28A; Zidovudine & IL-28B; ZIDOVUDINE [INN]; ZIDOVUDINE [JAN]; AZT & Interleukin 29; Opera_ID_1602; Prestwick3_000333; Spectrum2_000927; Spectrum3_001507; Spectrum4_000332; Spectrum5_001101; 3'azido-3'deoxythymidine; ZIDOVUDINE [HSDB]; ZIDOVUDINE [USAN]; 3'-Azido-3'-deoxythymidine & Erythropoietin; 3'-Azido-3'-deoxythymidine & Sho-Saiko-To; Azidothymidine; Zidovudine; Interferon AD + 3'-azido-3'-deoxythymidine; AZT & Interleukin 28A; AZT & Interleukin 28B; 3'-Azido-3'-deoxythymidine & Concanavalin A; 3'-Azido-3'-deoxythymidine & Interleukin-1; 3'-Azido-3'-deoxythymidine & Interleukin-2; 3'-Azido-3'-deoxythymidine & Interleukin-6; ZIDOVUDINE [VANDF]; 3'-azido-3-deoxythimydine; 3'-azido3'-deoxythymidine; AZT & IFNL1; AZT & IFNL2; AZT & IFNL3; AZT & Interferon lambda-1; AZT & Interferon lambda-2; AZT & Interferon lambda-3; AZT (PHARMACEUTICAL); ZIDOVUDINE [WHO-DD]; ZIDOVUDINE [WHO-IP]; 3''-Deoxy-3-azidothymidine; BSPBio_000365; BSPBio_003153; KBioGR_000703; KBioSS_001828; MLS001055351; MLS001076358; MLS002153202; MLS002222249; Zidovudine & Interleukin 29; DivK1c_000524; SPECTRUM1502109; ZIDOVUDINE [EMA EPAR]; 3'-deoxy-3'-azido-thymidine; SPBio_000834; Zidovudine & Interleukin 28A; Zidovudine & Interleukin 28B; AZT & IL-28A; AZT & IL-28B; BPBio1_000403; GTPL4825; Zidovudine (JP17/USP/INN); 3'-Azido-3'-deoxythymidine & Lithium & Erythropoietin; 3'-Azido-3'-deoxythymidine & Lithium & Interleukin-1; 3'-Azido-3'-deoxythymidine & Lithium & Interleukin-6; 3'-Azido-3'-deoxythymidine & Lymphoblastoid Interferon; SCHEMBL14615088; SN-1-dipalmitoylglycerophospho-AZT (in a lipid vesicle); SN-3-dipalmitoylglycerophospho-AZT (in a lipid vesicle); ZIDOVUDINE [ORANGE BOOK]; AZT & IL-29; HMS501K06; KBio1_000524; KBio2_001828; KBio2_004396; KBio2_006964; KBio3_002653; J05AF01; racemic-dipalmitoylglycerophospho-AZT (in a lipid vesicle); 3''-Azido-3''-deoxy-thymidine; NINDS_000524; Zidovudine & Interferon lambda-1; Zidovudine & Interferon lambda-2; Zidovudine & Interferon lambda-3; HMS1921J20; HMS2090G11; HMS2092D06; HMS2096C07; HMS2234K17; HMS3259H17; HMS3713C07; Pharmakon1600-01502109; ZIDOVUDINUM [WHO-IP LATIN]; 3''azido-2''3''-dideoxythymidine; Tox21_110062; Tox21_110894; Tox21_202203; Tox21_300578; BBL033764; BDBM50002692; CCG-39924; NSC758185; STK801891; AKOS005622576; AKOS015842610; Tox21_110062_1; 3''-azido-2'',3''-dideoxythymidine; DB00495; NC00666; NSC-758185; ZIDOVUDINE COMPONENT OF COMBIVIR; ZIDOVUDINE COMPONENT OF TRIZIVIR; IDI1_000524; NCGC00014918-01; NCGC00023945-03; NCGC00023945-04; NCGC00023945-06; NCGC00023945-07; NCGC00023945-08; NCGC00023945-09; NCGC00023945-10; NCGC00023945-12; NCGC00023945-13; NCGC00023945-24; NCGC00023945-25; NCGC00178237-01; NCGC00178237-02; NCGC00254276-01; NCGC00259752-01; 1-((2R,4S,5S)-4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione; AS-13019; SBI-0051731.P002; DB-225998; FT-0601543; NS00000600; Propolis & Thymidine, 3'-azido-3'-deoxy-; S2579; SW198799-2; EN300-52518; 3'-Azido-3'-deoxythymidine, >=98% (HPLC); C07210; D00413; D88500; 3'-Azido-3'deoxythymidine & Interferon .alpha.; 3'-Azido-3'-deoxythymidine, >=99.0% (HPLC); A820413; Q198504; SR-01000000098; SR-05000001587; J-700147; SR-01000000098-3; SR-05000001587-1; BRD-K72903603-001-04-6; BRD-K72903603-001-14-5; LAMIVUDINE/ZIDOVUDINE TEVA COMPONENT ZIDOVUDINE; Z754931264; Zidovudine, European Pharmacopoeia (EP) Reference Standard; 3'-Azido-3'-deoxythymidine & Recombinant Interferon-.alpha.-2; Zidovudine, United States Pharmacopeia (USP) Reference Standard; 3'-Azido-3'-deoxythymidine & CD4-Pseudomonas exotoxin A hybrid; Beta interferon(rIFN-beta seron) & 3'-Azido-3'-deoxythymidine(AZT); Lecithinized superoxide dismutase & Thymidine, 3'-azido-3'-deoxy-; 3'-Azido-2',3'-dideoxythymidine & sCD4(soluble recombinant protein); Thymidine, 3'-azido-3'-deoxy- & PRO 140 (Anti-CCR5 monoclonal antibody); Zidovudine, Pharmaceutical Secondary Standard; Certified Reference Material; (AZT) 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione; (AZT)1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione; 1-(3-Azido-2,3-dideoxy-beta-D-ribofuranosy)-5-methylpyridimine-2,4-(1H,3H)-dione; 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione; 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione(AZT); 3'-Azido-3'-deoxythymidine & Granulocyte-macrophage colony-stimulating factor; 3'-Azido-3'-deoxythymidine & Lithium & Granulocyte-macrophage colony-stimulating factor; 3'-Azido-3'deoxythymidine & Recombinant Soluble CD4 & Recombinant Interferon.alpha.A; 4-(4-Azido-5-hydroxy-tetrahydro-furan-2-yl)-5-methyl-3H-pyrazine-2,6-dione; Met-Stromal Cell-derived Factor-1.beta. (Human) & 3'-Azido-3'-deoxythymidine; 1-((2R,4S,5S)-4-(diazoamino)-5-(hydroxymethyl)-tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione; 1-((2R,4S,5S)-4-azido-5-(hydroxymethyl)-tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1H,3H)-dione; 1-((2R,5S)-4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione; 1-((2S,4R,5R)-4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione; 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione (AzddThd, AZT); 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione (N3ddThd); 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione(3''-azido-2'',3''-dideoxythymidine); 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione(azidothymidine, AZT); 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-2-yl)-5-methyl-1H-pyrimidine-2,4-dione(Zidovudine, AZT); 3-((2S,3S,5R)-2-(hydroxymethyl)-5-(5-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)tetrahydrofuran-3-yl)triaz-1-en-2-ium-1-ide
Click to Show/Hide
|
|||||
| Structure |
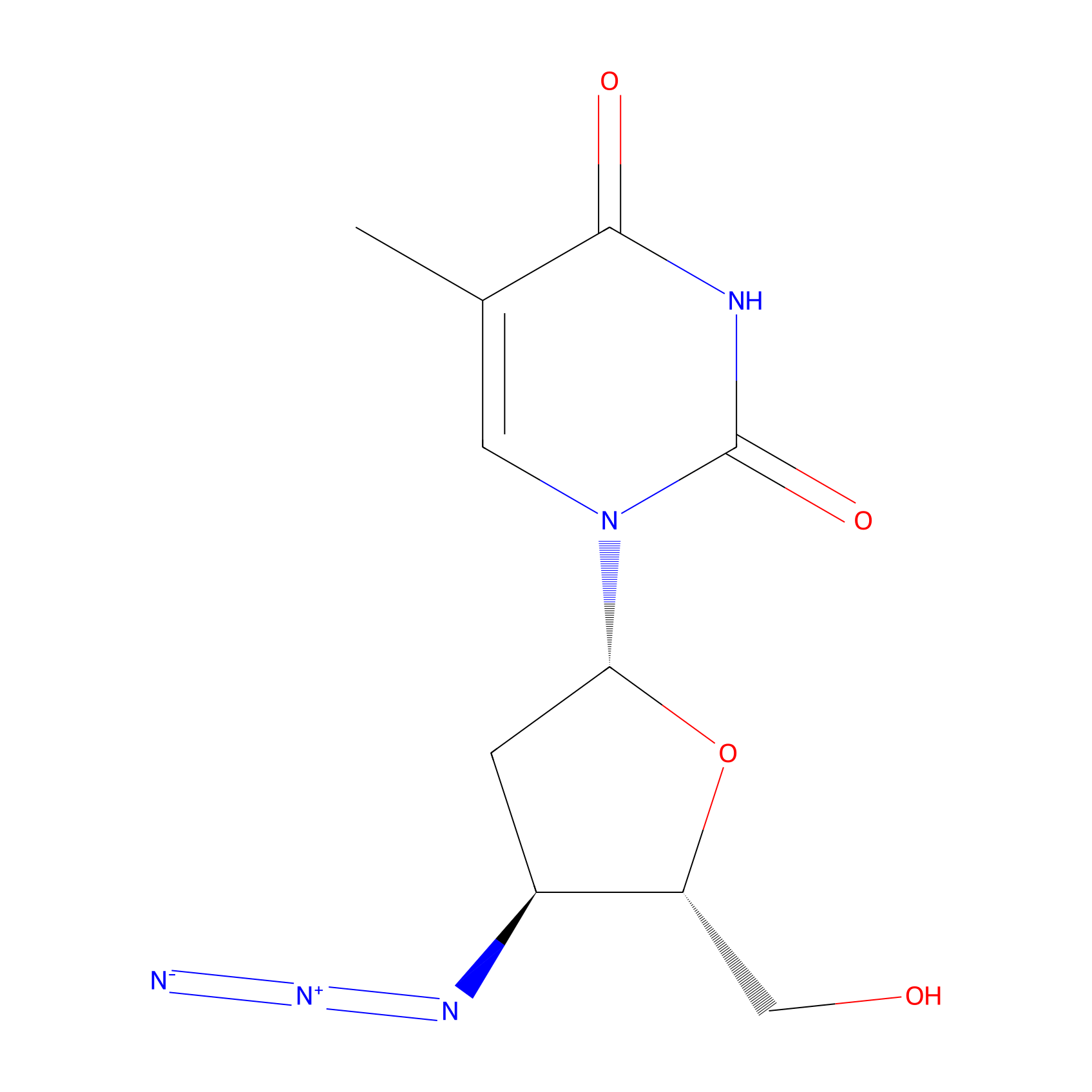
|
|||||
| Formula |
C10H13N5O4
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 267.24 | ||||
| Lipid-water partition coefficient (xlogp) | 0 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | |||||
| Rotatable Bond Count (rotbonds) | 3 | |||||
| PubChem CID | ||||||
| Canonical smiles |
CC1=CN(C(=O)NC1=O)C2CC(C(O2)CO)N=[N+]=[N-]
|
|||||
| InChI |
InChI=1S/C10H13N5O4/c1-5-3-15(10(18)12-9(5)17)8-2-6(13-14-11)7(4-16)19-8/h3,6-8,16H,2,4H2,1H3,(H,12,17,18)/t6-,7+,8+/m0/s1
|
|||||
| InChIKey |
HBOMLICNUCNMMY-XLPZGREQSA-N
|
|||||
| IUPAC Name |
1-[(2R,4S,5S)-4-azido-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione
|
|||||
The activity data of This Drug
| Standard Type | Value | Disease Model | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Cytotoxicity Concentration (CC50) | 1.5 ug/mL | Normal | MT4 cell | CVCL_2632 | [1] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 1.64 ug/mL | Normal | MT4 cell | CVCL_2632 | [2] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 2.01 ug/mL | Normal | MT4 cell | CVCL_2632 | [3] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 2.581 ug/mL | Normal | MT4 cell | CVCL_2632 | [4] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 4.5521 mM | Leukemia | C8166 cell | CVCL_1099 | [5] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >5.092 mM | Leukemia | C8166 cell | CVCL_1099 | [6] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 5.4014 mM | Leukemia | C8166 cell | CVCL_1099 | [7] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 5.60171 mM | Leukemia | C8166 cell | CVCL_1099 | [8] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 14.3 mM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [9] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 35.6 ug/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [10] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 65.4 ug/mL | Normal | MT4 cell | CVCL_2632 | [11] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >100 ug/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [1] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >200 ug/mL | Leukemia | C8166 cell | CVCL_1099 | [12] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >500 nM | Normal | MT4 cell | CVCL_2632 | [13] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >1000 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [14] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >1000 nM | Normal | MT4 cell | CVCL_2632 | [14] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >3.74 uM | Normal | MT4 cell | CVCL_2632 | [15] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 6 uM | Normal | MT4 cell | CVCL_2632 | [16] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >7.48 uM | Normal | MT4 cell | CVCL_2632 | [17] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 7.8 uM | Normal | MT4 cell | CVCL_2632 | [18] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >10 uM | Normal | MT-2 cell | CVCL_2631 | [19] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 12 uM | Normal | MT4 cell | CVCL_2632 | [20] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >20 uM | Normal | MT4 cell | CVCL_2632 | [21] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 20 uM | Normal | MT4 cell | CVCL_2632 | [22] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 35.6 uM | Normal | MT4 cell | CVCL_2632 | [23] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 53 uM | Normal | Vero cell | CVCL_0059 | [24] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 60 uM | Normal | MT4 cell | CVCL_2632 | [25] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 61 uM | Normal | MT4 cell | CVCL_2632 | [26] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 75 uM | Normal | MT4 cell | CVCL_2632 | [27] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >80 uM | Normal | MT4 cell | CVCL_2632 | [28] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >93.55 uM | Normal | MT4 cell | CVCL_2632 | [29] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 93.55 uM | Normal | MT4 cell | CVCL_2632 | [30] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >100 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [31] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >100 uM | Normal | MT4 cell | CVCL_2632 | [32] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 123 uM | Normal | MT-2 cell | CVCL_2631 | [33] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 126 uM | Normal | Vero cell | CVCL_0059 | [34] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 200 uM | Sezary syndrome | H9 cell | CVCL_1240 | [35] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >200 uM | Normal | MT-2 cell | CVCL_2631 | [36] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >250 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [37] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >500 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [16] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 500 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [35] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >500 uM | T acute lymphoblastic leukemia | Jurkat cell | CVCL_0065 | [35] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >749 uM | Leukemia | C8166 cell | CVCL_1099 | [38] | |
| Half Maximal Effective Concentration (EC50) | 0.5 ng/mL | Normal | MT4 cell | CVCL_2632 | [39] | |
| Half Maximal Effective Concentration (EC50) | 0.7 ng/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [1] | |
| Half Maximal Effective Concentration (EC50) | 0.7 ng/mL | Normal | MT4 cell | CVCL_2632 | [1] | |
| Half Maximal Effective Concentration (EC50) | 2 ng/mL | Normal | MT4 cell | CVCL_2632 | [40] | |
| Half Maximal Effective Concentration (EC50) | 10 ng/mL | Sezary syndrome | H9 cell | CVCL_1240 | [41] | |
| Half Maximal Effective Concentration (EC50) | 10 ng/mL | Normal | MT4 cell | CVCL_2632 | [2] | |
| Half Maximal Effective Concentration (EC50) | 12 ng/mL | Sezary syndrome | H9 cell | CVCL_1240 | [42] | |
| Half Maximal Effective Concentration (EC50) | 0.7 nM | Normal | MT4 cell | CVCL_2632 | [43] | |
| Half Maximal Effective Concentration (EC50) | <1 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [44] | |
| Half Maximal Effective Concentration (EC50) | 2 nM | Normal | MT4 cell | CVCL_2632 | [16] | |
| Half Maximal Effective Concentration (EC50) | 2.8 nM | Normal | MT4 cell | CVCL_2632 | [18] | |
| Half Maximal Effective Concentration (EC50) | 3 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [16] | |
| Half Maximal Effective Concentration (EC50) | 3 nM | Normal | MT4 cell | CVCL_2632 | [16] | |
| Half Maximal Effective Concentration (EC50) | 4 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [45] | |
| Half Maximal Effective Concentration (EC50) | 4.5 nM | Normal | MT4 cell | CVCL_2632 | [46] | |
| Half Maximal Effective Concentration (EC50) | 5 nM | Normal | MT4 cell | CVCL_2632 | [47] | |
| Half Maximal Effective Concentration (EC50) | 6 nM | Normal | MT4 cell | CVCL_2632 | [46] | |
| Half Maximal Effective Concentration (EC50) | 9 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [48] | |
| Half Maximal Effective Concentration (EC50) | 10 nM | Normal | MT4 cell | CVCL_2632 | [49] | |
| Half Maximal Effective Concentration (EC50) | 12 nM | Normal | MT4 cell | CVCL_2632 | [50] | |
| Half Maximal Effective Concentration (EC50) | 13 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [48] | |
| Half Maximal Effective Concentration (EC50) | 14 nM | Normal | MT-2 cell | CVCL_2631 | [51] | |
| Half Maximal Effective Concentration (EC50) | 15 nM | Sezary syndrome | H9 cell | CVCL_1240 | [52] | |
| Half Maximal Effective Concentration (EC50) | 15 nM | Normal | MT4 cell | CVCL_2632 | [14] | |
| Half Maximal Effective Concentration (EC50) | 16 nM | Normal | MT4 cell | CVCL_2632 | [22] | |
| Half Maximal Effective Concentration (EC50) | 20 nM | Leukemia | C8166 cell | CVCL_1099 | [53] | |
| Half Maximal Effective Concentration (EC50) | 37 nM | Normal | MT-2 cell | CVCL_2631 | [51] | |
| Half Maximal Effective Concentration (EC50) | 40 nM | Normal | MT4 cell | CVCL_2632 | [21] | |
| Half Maximal Effective Concentration (EC50) | 45 nM | Sezary syndrome | H9 cell | CVCL_1240 | [54] | |
| Half Maximal Effective Concentration (EC50) | 85 nM | Normal | MT4 cell | CVCL_2632 | [55] | |
| Half Maximal Effective Concentration (EC50) | 180 nM | Normal | MT-2 cell | CVCL_2631 | [56] | |
| Half Maximal Effective Concentration (EC50) | 300 nM | Normal | MT4 cell | CVCL_2632 | [18] | |
| Half Maximal Effective Concentration (EC50) | 700 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [57] | |
| Half Maximal Effective Concentration (EC50) | >20 uM | Sezary syndrome | H9 cell | CVCL_1240 | [28] | |
| Half Maximal Effective Concentration (EC50) | 25 uM | Acute monocytic leukemia | U1 cell | CVCL_M769 | [44] | |
| Half Maximal Effective Concentration (EC50) | >50 uM | Normal | Vero cell | CVCL_0059 | [58] | |
| Half Maximal Effective Concentration (EC50) | >100 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [59] | |
| Half Maximal Effective Concentration (EC50) | >100 uM | Normal | Vero cell | CVCL_0059 | [60] | |
| Half Maximal Effective Dosage (ED50) | 4 nM | Normal | MT4 cell | CVCL_2632 | [61] | |
| Half Maximal Effective Dosage (ED50) | 6 nM | Normal | MT4 cell | CVCL_2632 | [62] | |
| Half Maximal Effective Dosage (ED50) | 70 nM | Normal | MT-2 cell | CVCL_2631 | [33] | |
| Half Maximal Infective Dose (ID50) | 0.53 ug/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [63] | |
| Half Maximal Infective Dose (ID50) | 30 ng/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [63] | |
| Half Maximal Infective Dose (ID50) | 80 ng/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [63] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.87 mM | Normal | MT-2 cell | CVCL_2631 | [64] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.873 mM | Sezary syndrome | H9 cell | CVCL_1240 | [54] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.875 mM | Sezary syndrome | H9 cell | CVCL_1240 | [65] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.9 mM | Sezary syndrome | H9 cell | CVCL_1240 | [66] | |
| Half Maximal Inhibitory Concentration (IC50) | 3 ng/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [10] | |
| Half Maximal Inhibitory Concentration (IC50) | 20 ng/mL | Normal | MT4 cell | CVCL_2632 | [67] | |
| Half Maximal Inhibitory Concentration (IC50) | <30 ng/mL | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [68] | |
| Half Maximal Inhibitory Concentration (IC50) | 500 ug/mL | Sezary syndrome | H9 cell | CVCL_1240 | [42] | |
| Half Maximal Inhibitory Concentration (IC50) | 5 nM | Normal | MT4 cell | CVCL_2632 | [35] | |
| Half Maximal Inhibitory Concentration (IC50) | 6 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 10 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [35] | |
| Half Maximal Inhibitory Concentration (IC50) | 10 nM | T acute lymphoblastic leukemia | Jurkat cell | CVCL_0065 | [35] | |
| Half Maximal Inhibitory Concentration (IC50) | 14 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 17 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 20 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 30 nM | Sezary syndrome | H9 cell | CVCL_1240 | [70] | |
| Half Maximal Inhibitory Concentration (IC50) | 40 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 46 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 60 nM | Normal | MT4 cell | CVCL_2632 | [71] | |
| Half Maximal Inhibitory Concentration (IC50) | 80 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 87 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 nM | Sezary syndrome | H9 cell | CVCL_1240 | [72] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.77 uM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [73] | |
| Half Maximal Inhibitory Concentration (IC50) | 10 uM | Normal | MT-2 cell | CVCL_2631 | [70] | |
| Half Maximal Inhibitory Concentration (IC50) | 11.86 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [74] | |
| Half Maximal Inhibitory Concentration (IC50) | 13 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [75] | |
| Half Maximal Inhibitory Concentration (IC50) | 14.3 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [76] | |
| Half Maximal Inhibitory Concentration (IC50) | 20 uM | T acute lymphoblastic leukemia | CEM cl13 cell | CVCL_V186 | [77] | |
| Half Maximal Inhibitory Concentration (IC50) | 26 uM | Normal | Vero cell | CVCL_0059 | [60] | |
| Half Maximal Inhibitory Concentration (IC50) | 28 uM | Normal | Vero cell | CVCL_0059 | [78] | |
| Half Maximal Inhibitory Concentration (IC50) | 29 uM | Normal | Vero cell | CVCL_0059 | [79] | |
| Half Maximal Inhibitory Concentration (IC50) | 30.9 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [79] | |
| Half Maximal Inhibitory Concentration (IC50) | 31 uM | Normal | MT-2 cell | CVCL_2631 | [70] | |
| Half Maximal Inhibitory Concentration (IC50) | 35.6 uM | Normal | MT4 cell | CVCL_2632 | [80] | |
| Half Maximal Inhibitory Concentration (IC50) | 53 uM | Normal | MT4 cell | CVCL_2632 | [46] | |
| Half Maximal Inhibitory Concentration (IC50) | 62.8 uM | T acute lymphoblastic leukemia | MOLT-4 cell | CVCL_0013 | [81] | |
| Half Maximal Inhibitory Concentration (IC50) | 96 uM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [46] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [31] | |
| Half Maximal Inhibitory Concentration (IC50) | 100 uM | Normal | MT4 cell | CVCL_2632 | [47] | |
| Half Maximal Inhibitory Concentration (IC50) | >100 uM | Leukemia | C8166 cell | CVCL_1099 | [82] | |
| Half Maximal Inhibitory Concentration (IC50) | 119.84 uM | Normal | MT-2 cell | CVCL_2631 | [51] | |
| Half Maximal Inhibitory Concentration (IC50) | 128.82 uM | Bladder carcinoma | 5637 cell | CVCL_0126 | [83] | |
| Half Maximal Inhibitory Concentration (IC50) | 150 uM | Sezary syndrome | H9 cell | CVCL_1240 | [84] | |
| Half Maximal Inhibitory Concentration (IC50) | 191 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [85] | |
| Half Maximal Inhibitory Concentration (IC50) | >375 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [86] | |
| Half Maximal Inhibitory Concentration (IC50) | 500 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [87] | |
| Half Maximal Inhibitory Concentration (IC50) | >970 uM | Normal | MT-2 cell | CVCL_2631 | [56] | |
Full Information of The Activity Data of The PDC(s) Related to This Drug
Z-Phe-Pro-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 5 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 6 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Fmoc-Phe-Pro-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 8 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 27 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Qnc-Phe-Pro-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 14 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 27 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 16 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 16 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Boc-Phe-Pro-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 15 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 18 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tripeptide 84 - Azidothymidine conjugate 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 20 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 110 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tripeptide 84 - Azidothymidine conjugate 1 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 29 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 320 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Fmoc-Asn-Phe-Pro-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 30 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 59 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Boc-Asn-Phe-Pro-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 35 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 82 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Boc-Phe-Pro-Ile-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 42 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 120 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 25 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 25 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tripeptide 82 - Azidothymidine conjugate 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 45 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 65 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Qnc-Asn-Phe-Pro-AZT [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 64 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 94 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 30 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 30 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tetrapeptide 40 - Azidothymidine conjugate 2 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 76 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 320 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tetrapeptide 40 - Azidothymidine conjugate 8 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 100 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 270 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | 9 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tripeptide 84 - Azidothymidine conjugate 3 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 120 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 140 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tetrapeptide 40 - Azidothymidine conjugate 4 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 120 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 530 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 10 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
Tetrapeptide 40 - Azidothymidine conjugate 7 [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 140 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | 440 nM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) | > 10 | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | 84 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MT4/HIV-1 cell | CVCL_RW54 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK+ cell | CVCL_0207 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [88] | ||||
| Indication | Human immunodeficiency virus infection | ||||
| Efficacy Data | Half maximal cytotoxicity concentration (CC50) | > 100 μM | |||
| Administration Time | 5 days | ||||
| MOA of PDC |
We report on the synthesis of a series of peptideAZT (2A, 2B, 5A, 5B) and peptide-(AZT-MP) conjugates incorporating peptide sequences found in a nonapeptide known to be a HIV-PR substrate. We also selected the AZT-MP phosphoramidate peptide-conjugates containing an additional alanine residue directly linked to the phosphorus atom. Indeed, among the various aminoacid phosphoramidates of ddN analogues known so far, the alanine derivative was shown to be one of the most efficient ddN-MP delivery system. All these conjugates contain the scissile Phe-Pro motif. In the case of the AZT prodrugs, the AZT and peptide moieties were connected through an ester bond. To ensure a greater stability, particularly toward aminopeptidases, the N-terminal amino group of the peptide was masked by different protecting groups. In the case of the AZT-MP prodrugs, the various peptide moieties were connected via their N-terminal amino group to the AZT-phenoxy-phosphodiester group. We report also on (i) the stability of the peptide-AZT and peptide(AZT-MP) prodrugs when incubated in a physiological medium that does or does not contain 10% fetal calf serum, and in CEM cell lysates in order to mimic the behaviour of these compounds inside the cells, (ii) their ability to inhibit the HIV-PR activity and their susceptibility to be hydrolysed by PR, and (iii) their in vitro anti-HIV activities and cytotoxicities, which were evaluated in acutely-infected and uninfected MT4 and CEM cells, respectively. Moreover, their antiviral activities were also investigated in a thymidine kinase-deficient (TK- ) CEM cell line in order to gain further insight into their mechanism of action.
Click to Show/Hide
|
||||
| Description |
The in vitro anti-HIV activity and cytotoxicity of the peptide conjugates were determined both in infected and non-infected CEM-SS and MT-4 cells against HIV-1 (LAI and IIIB strain, respectively) according to published procedures. These assays were also performed on infected and non-infected TK- CEM cells.The data are collected in Tables 1 and 2 together along with data for AZT. Ester conjugates when incubated with the cells for 5 days were found to inhibit HIV replication at IC50 ranging from 6 to 140 nM in TK+ CEM-SS and from 5 to 530 nM in MT-4. Under these assay conditions, AZT inhibited HIV replication at IC50 of 14 nM (CEM-SS) and 16 nM (MT-4). As with their parent drug, none of the conjugates inhibited HIV replication in TK- CEM cells. Moreover, except for compound 5Bb, none of them was found to be cytotoxic on the three cell types. The antiviral activity level measured for these ester conjugates appeared to be correlated, to some extent, to their hydrolysis t1/2 value and hence, to the amount of AZT released during the 5 days of the experiment. Indeed, the prodrug series displaying the higher t1/2 value range, that is, the Ile-AZT series, correspond to those for which a lower HIV inhibition level was obtained. Conversely, the set of prodrug that gave a higher anti-HIV inhibition, that is, the Pro-AZT series, were those that were hydrolysed more rapidly. Moreover, the faster their hydrolysis, the closer their antiviral activity to that of free AZT. This is particularly relevant for the antiviral activities measured on MT-4 cells. However, it is noticeable that compounds Z-Phe-ProAZT (2Ac) and Qnc-Phe-Pro-Ile-AZT (2Bd), which are the most stable compounds of the series, constitute exception to this rule. Indeed, these derivatives exhibited, despite a quite high stability, a surprizingly high anti-HIV activity in CEM-SS and also in MT-4 cells for 2Ac.
Click to Show/Hide
|
||||
| In Vitro Model | HIV infection | HIV-1 LAI infected CEM/TK- cell | CVCL_0207 | ||
References
