Drug Information
General Information of This Drug
| Drug ID | DRG00407 | |||||
|---|---|---|---|---|---|---|
| Drug Name | Combretastatin A4 | |||||
| Synonyms |
Combretastatin A4; 117048-59-6; Combretastatin A-4; Combrestatin A4; (Z)-2-METHOXY-5-(3,4,5-TRIMETHOXYSTYRYL)PHENOL; Combretastatin 4; Combretastatin-A4; CA-4; 2'-deoxycombretastatin A1; NSC-613729; 2-methoxy-5-[(Z)-2-(3,4,5-trimethoxyphenyl)ethenyl]phenol; Crc 87-09; CHEMBL67; MFCD03453309; 3,4,5-Trimethoxy-3'-hydroxy-4'-methoxystilbene; NSC-817373; 16U6OP69RQ; Phenol, 2-methoxy-5-[(1Z)-2-(3,4,5-trimethoxyphenyl)ethenyl]-; CA4; 2-Methoxy-5-[(1Z)-2-(3,4,5-trimethoxyphenyl)ethenyl]phenol; 2-methoxy-5-[(Z)-2-(3,4,5-trimethoxyphenyl)vinyl]phenol; 2-Methoxy-5-[(Z)-2-(3,4,5-trimethoxy-phenyl)-vinyl]-phenol; Phenol, 2-methoxy-5-(2-(3,4,5-trimethoxyphenyl)ethenyl)-, (Z)-; NSC 817373; Crc-98-09; UNII-16U6OP69RQ; combretastin A4; Combrestatin A-4; Combretastin A-4; combretastatin-A-4; Combretastastin A-4; NSC 609397; NSC 613729; 2-methoxy-5-(3,4,5-trimethoxystyryl)phenol; Z-Combretastatin A-4; 2-Methoxy-5-[2-(3,4,5-trimethoxy-phenyl)-vinyl]-phenol; SCHEMBL18945; combretastatin A-4, (CSA4); CA 4; GTPL8854; COMBRETASTATIN A-4 [MI]; CHEBI:181391; DTXSID101025983; GLXC-02861; HMS3650G11; AMY24892; COMBRETASTATIN A4 [WHO-DD]; EX-A4207; HY-N2146; BDBM50005480; s7783; AKOS015950727; AC-5280; CCG-265026; CS-6023; DB14680; DS-5860; Phenol,4,5-trimethoxyphenyl)ethenyl]-; ST-1986; C2520; Combretastatin A4, >=98% (HPLC), powder; NS00116331; 5-(3,4,5-trimethoxystyryl)-2-methoxyphenol; C20268; 3'-Hydroxy-3,4,4',5-tetramethoxy-cis-stilbene; EN300-1587417; (Z)-5-(3,4,5-trimethoxystyryl)-2-methoxyphenol; SR-01000946784; (Z)-3''-hydroxy-3,4,4'',5-tetramethoxystilbene; Q5150955; SR-01000946784-1; BRD-K61195623-001-01-4; Z2312943638; [3,4,5-trimethoxy-3'-hydroxy-4'-methoxy-(z)-stilbene]; 2-Methoxy-5-[(Z)-2-(3,4,5-trimethox y-phenyl)-vinyl]-phenol; (combretastin A-4)2-Methoxy-5-[2-(3,4,5-trimethoxy-phenyl)-vinyl]-phenol; 5-[(S)-2-Hydroxy-2-(3,4,5-trimethoxy-phenyl)-ethyl]-2-methoxy-phenol; (combretastatin A-4)2-Methoxy-5-[2-(3,4,5-trimethoxy-phenyl)-vinyl]-phenol; 1-(3,4,5-Trimethoxyphenyl)-2-(3'-hydroxy-4'-methoxyphenyl) ethane 3,4,5-trimethoxy-3'-hydroxy-4'-methoxystilbene
Click to Show/Hide
|
|||||
| Target(s) | Microtubule (MT) | Target Info | ||||
| Structure |
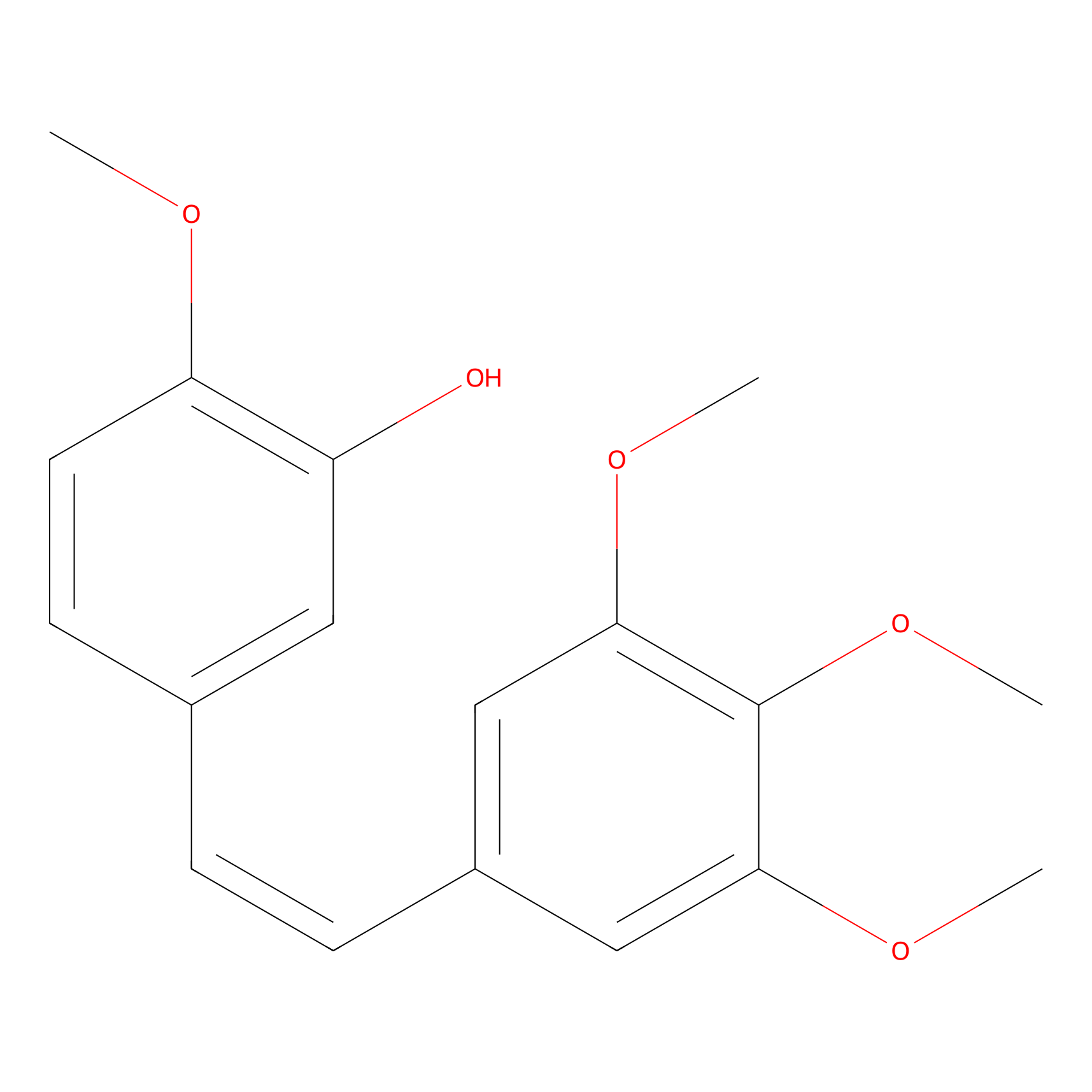
|
|||||
| Formula |
C18H20O5
|
|||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 316.3 | ||||
| Lipid-water partition coefficient (xlogp) | 3.7 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | |||||
| Rotatable Bond Count (rotbonds) | 6 | |||||
| PubChem CID | ||||||
| Canonical smiles |
COC1=C(C=C(C=C1)C=CC2=CC(=C(C(=C2)OC)OC)OC)O
|
|||||
| InChI |
InChI=1S/C18H20O5/c1-20-15-8-7-12(9-14(15)19)5-6-13-10-16(21-2)18(23-4)17(11-13)22-3/h5-11,19H,1-4H3/b6-5-
|
|||||
| InChIKey |
HVXBOLULGPECHP-WAYWQWQTSA-N
|
|||||
| IUPAC Name |
2-methoxy-5-[(Z)-2-(3,4,5-trimethoxyphenyl)ethenyl]phenol
|
|||||
The activity data of This Drug
| Standard Type | Value | Disease Model | Cell line | Cell line ID | Ref. | |
|---|---|---|---|---|---|---|
| Half Maximal Cytotoxicity Concentration (CC50) | 13.9 uM | Normal | HT22 cell | CVCL_0321 | [1] | |
| Half Maximal Cytotoxicity Concentration (CC50) | 38.3 uM | Hepatocellular carcinoma | Huh-7 cell | CVCL_0336 | [2] | |
| Half Maximal Cytotoxicity Concentration (CC50) | >300 uM | Normal | HEK-293T cell | CVCL_0063 | [3] | |
| Half Maximal Effective Concentration (EC50) | 3.45 nM | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | [4] | |
| Half Maximal Effective Concentration (EC50) | 3.5 nM | Skin squamous cell carcinoma | A431 cell | CVCL_0037 | [5] | |
| Half Maximal Effective Dosage (ED50) | 2.7 ug/mL | Lymphoma | P388 cell | CVCL_7222 | [6] | |
| Half Maximal Effective Dosage (ED50) | 0.03 pM | Cutaneous melanoma | SK-MEL-5 cell | CVCL_0527 | [7] | |
| Half Maximal Effective Dosage (ED50) | 12:00 PM | Colon cancer | HT29 cell | CVCL_A8EZ | [7] | |
| Half Maximal Effective Dosage (ED50) | 1.4 uM | Cutaneous melanoma | SK-MEL-5 cell | CVCL_0527 | [8] | |
| Half Maximal Effective Dosage (ED50) | 5.6 uM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [8] | |
| Half Maximal Growth Inhibition (GI50) | 0.26 ng/mL | Anaplastic thyroid carcinoma | SW-1736 cell | CVCL_3883 | [9] | |
| Half Maximal Growth Inhibition (GI50) | 0.34 ug/mL | Colon carcinoma | KM-20L2 cell | CVCL_D889 | [10] | |
| Half Maximal Growth Inhibition (GI50) | 0.39 ug/mL | Pancreatic ductal adenocarcinoma | BxPC-3 cell | CVCL_0186 | [11] | |
| Half Maximal Growth Inhibition (GI50) | 0.6 ng/mL | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [10] | |
| Half Maximal Growth Inhibition (GI50) | 0.65 ng/mL | Hypopharyngeal squamous cell carcinoma | FaDu cell | CVCL_1218 | [9] | |
| Half Maximal Growth Inhibition (GI50) | 0.8 ng/mL | Prostate carcinoma | DU145 cell | CVCL_0105 | [12] | |
| Half Maximal Growth Inhibition (GI50) | <1 ug/mL | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | [6] | |
| Half Maximal Growth Inhibition (GI50) | <1 ug/mL | Glioblastoma | SF-295 cell | CVCL_1690 | [6] | |
| Half Maximal Growth Inhibition (GI50) | >1 ng/mL | Glioblastoma | SF-295 cell | CVCL_1690 | [10] | |
| Half Maximal Growth Inhibition (GI50) | <1 ng/mL | Astrocytoma | SF268 cell | CVCL_1689 | [13] | |
| Half Maximal Growth Inhibition (GI50) | 2 ug/mL | Cutaneous melanoma | SK-MEL-5 cell | CVCL_0527 | [6] | |
| Half Maximal Growth Inhibition (GI50) | 2.6 ug/mL | Neuroblastoma | SK-N-SH cell | CVCL_0531 | [6] | |
| Half Maximal Growth Inhibition (GI50) | 7.1 ug/mL | Anaplastic thyroid carcinoma | SW-1736 cell | CVCL_3883 | [6] | |
| Half Maximal Growth Inhibition (GI50) | 61 ng/mL | Colon carcinoma | KM-20L2 cell | CVCL_D889 | [13] | |
| Half Maximal Growth Inhibition (GI50) | <0.1 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [14] | |
| Half Maximal Growth Inhibition (GI50) | 1 nM | Prostate carcinoma | PC-3 cell | CVCL_0035 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 2 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 2 nM | Renal carcinoma | RXF 393 cell | CVCL_1673 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 2 nM | Non-small cell lung carcinoma | NCI-H522 cell | CVCL_1567 | [16] | |
| Half Maximal Growth Inhibition (GI50) | 2 nM | Non-small cell lung carcinoma | HOP-62 cell | CVCL_1285 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 2 nM | Melanoma | M14 cell | CVCL_1395 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 2 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [17] | |
| Half Maximal Growth Inhibition (GI50) | 2.65 nM | Non-small cell lung carcinoma | HOP-62 cell | CVCL_1285 | [18] | |
| Half Maximal Growth Inhibition (GI50) | 2.78 nM | Melanoma | M14 cell | CVCL_1395 | [18] | |
| Half Maximal Growth Inhibition (GI50) | 3 nM | Plasma cell myeloma | RPMI-8226 cell | CVCL_7353 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 3 nM | Cutaneous melanoma | SK-MEL-5 cell | CVCL_0527 | [16] | |
| Half Maximal Growth Inhibition (GI50) | 3 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 3 nM | Colon adenocarcinoma | SW620 cell | CVCL_0547 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 3.2 nM | Lymphoma | P388 cell | CVCL_7222 | [20] | |
| Half Maximal Growth Inhibition (GI50) | 4 nM | Astrocytoma | U-251MG cell | CVCL_0021 | [16] | |
| Half Maximal Growth Inhibition (GI50) | 4.55 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [21] | |
| Half Maximal Growth Inhibition (GI50) | 5 nM | Melanoma | UACC-62 cell | CVCL_1780 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 5.51 nM | Melanoma | UACC-62 cell | CVCL_1780 | [18] | |
| Half Maximal Growth Inhibition (GI50) | 5.98 nM | Renal cell carcinoma | SN12C cell | CVCL_1705 | [18] | |
| Half Maximal Growth Inhibition (GI50) | 6 nM | Renal cell carcinoma | SN12C cell | CVCL_1705 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 6 nM | Renal carcinoma | A498 cell | CVCL_1056 | [16] | |
| Half Maximal Growth Inhibition (GI50) | 6 nM | Renal adenocarcinoma | ACHN cell | CVCL_1067 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 6 nM | Melanoma | UACC-62 cell | CVCL_1780 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 6 nM | Astrocytoma | SF268 cell | CVCL_1689 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 6 nM | Astrocytoma | U-251MG cell | CVCL_0021 | [14] | |
| Half Maximal Growth Inhibition (GI50) | 7 nM | Minimally invasive lung adenocarcinoma | NCI-H322M cell | CVCL_1557 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 7 nM | Lung squamous cell carcinoma | NCI-H520 cell | CVCL_1566 | [14] | |
| Half Maximal Growth Inhibition (GI50) | 7 nM | Glioblastoma | SNB-75 cell | CVCL_1706 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 8 nM | Glioblastoma | SNB-75 cell | CVCL_1706 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 8 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [22] | |
| Half Maximal Growth Inhibition (GI50) | 10 nM | Prostate carcinoma | DU145 cell | CVCL_0105 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 10 nM | Melanoma | UACC-62 cell | CVCL_1780 | [23] | |
| Half Maximal Growth Inhibition (GI50) | <10 nM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [24] | |
| Half Maximal Growth Inhibition (GI50) | 10 nM | Cutaneous melanoma | SK-MEL-5 cell | CVCL_0527 | [23] | |
| Half Maximal Growth Inhibition (GI50) | <10 nM | Colon adenocarcinoma | HCT 15 cell | CVCL_0292 | [25] | |
| Half Maximal Growth Inhibition (GI50) | 10 nM | Breast ductal carcinoma | BT-549 cell | CVCL_1092 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 10 nM | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | [26] | |
| Half Maximal Growth Inhibition (GI50) | 15 nM | Ovarian endometrioid adenocarcinoma | IGROV-1 cell | CVCL_1304 | [19] | |
| Half Maximal Growth Inhibition (GI50) | 16 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [16] | |
| Half Maximal Growth Inhibition (GI50) | 18 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [16] | |
| Half Maximal Growth Inhibition (GI50) | 19 nM | Renal carcinoma | UO-31 cell | CVCL_1911 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 19 nM | Melanoma | Malme-3M cell | CVCL_1438 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 26 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [27] | |
| Half Maximal Growth Inhibition (GI50) | 31.6 nM | Amelanotic melanoma | MDA-MB-435 cell | CVCL_0417 | [28] | |
| Half Maximal Growth Inhibition (GI50) | 31.62 nM | Prostate carcinoma | PC-3 cell | CVCL_0035 | [29] | |
| Half Maximal Growth Inhibition (GI50) | 31.62 nM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 31.62 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 31.62 nM | Colon adenocarcinoma | HCT 15 cell | CVCL_0292 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 31.62 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 39.81 nM | Prostate carcinoma | PC-3 cell | CVCL_0035 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 39.81 nM | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 39.81 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 39.81 nM | Clear cell renal cell carcinoma | Caki-1 cell | CVCL_0234 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 50.12 nM | Colon adenocarcinoma | KM12 cell | CVCL_1331 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 63 nM | Colon adenocarcinoma | HCC 2998 cell | CVCL_1266 | [15] | |
| Half Maximal Growth Inhibition (GI50) | 63.1 nM | High grade ovarian serous adenocarcinoma | NCI-ADR-RES cell | CVCL_1452 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 84 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [30] | |
| Half Maximal Growth Inhibition (GI50) | 84.1 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [31] | |
| Half Maximal Growth Inhibition (GI50) | 100 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [29] | |
| Half Maximal Growth Inhibition (GI50) | 125.89 nM | Astrocytoma | SF268 cell | CVCL_1689 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 158.49 nM | Plasma cell myeloma | RPMI-8226 cell | CVCL_7353 | [29] | |
| Half Maximal Growth Inhibition (GI50) | 199.53 nM | Renal carcinoma | UO-31 cell | CVCL_1911 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 200 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [1] | |
| Half Maximal Growth Inhibition (GI50) | 398.11 nM | Non-small cell lung carcinoma | EKVX cell | CVCL_1195 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 630.96 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [23] | |
| Half Maximal Growth Inhibition (GI50) | 631 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [28] | |
| Half Maximal Growth Inhibition (GI50) | 794.33 nM | Glioblastoma | SNB-75 cell | CVCL_1706 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 1000 nM | Renal carcinoma | RXF 393 cell | CVCL_1673 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 2.51189 uM | Normal | COLO205 cell | CVCL_F402 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 3.16228 uM | Cutaneous melanoma | SK-MEL-28 cell | CVCL_0526 | [23] | |
| Half Maximal Growth Inhibition (GI50) | 31.6 uM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [22] | |
| Half Maximal Inhibitory Concentration (IC50) | <1 ug/mL | Endocervical adenocarcinoma | L132 cell | CVCL_1908 | [32] | |
| Half Maximal Inhibitory Concentration (IC50) | 5 ug/mL | Colon cancer | HT29 cell | CVCL_A8EZ | [32] | |
| Half Maximal Inhibitory Concentration (IC50) | 57 ug/mL | Prostate carcinoma | DU145 cell | CVCL_0105 | [32] | |
| Half Maximal Inhibitory Concentration (IC50) | 28.2 pM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [33] | |
| Half Maximal Inhibitory Concentration (IC50) | 79.4 pM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [33] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.1 nM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [34] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.24 nM | Invasive breast carcinoma | ZR-75-1 cell | CVCL_0588 | [35] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.8 nM | T acute lymphoblastic leukemia | Jurkat cell | CVCL_0065 | [36] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.9 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [37] | |
| Half Maximal Inhibitory Concentration (IC50) | 0.94 nM | Lung adenocarcinoma | Calu-6 cell | CVCL_0236 | [38] | |
| Half Maximal Inhibitory Concentration (IC50) | 1 nM | Melanoma | B16 cell | CVCL_F936 | [37] | |
| Half Maximal Inhibitory Concentration (IC50) | 1 nM | Glioma | A10 cell | CVCL_0130 | [39] | |
| Half Maximal Inhibitory Concentration (IC50) | 1 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [40] | |
| Half Maximal Inhibitory Concentration (IC50) | 1 nM | B acute lymphoblastic leukemia |
RS4
11 cell |
CVCL_0093 | [41] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.2 nM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [42] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.3 nM | High grade ovarian serous adenocarcinoma | OVCAR-8 cell | CVCL_1629 | [39] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.4 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [43] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.5 nM | Bone marrow neuroblastoma | SH-SY5Y cell | CVCL_0019 | [44] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.52 nM | Colon adenocarcinoma | SW620 cell | CVCL_0547 | [45] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.6 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [46] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.7 nM | Colon adenocarcinoma | HCT 15 cell | CVCL_0292 | [47] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.9 nM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [48] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.9 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [49] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.9 nM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [50] | |
| Half Maximal Inhibitory Concentration (IC50) | 2 nM | Melanoma | B16 cell | CVCL_F936 | [51] | |
| Half Maximal Inhibitory Concentration (IC50) | 2 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [52] | |
| Half Maximal Inhibitory Concentration (IC50) | 2 nM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [53] | |
| Half Maximal Inhibitory Concentration (IC50) | 2 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [54] | |
| Half Maximal Inhibitory Concentration (IC50) | 2 nM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [55] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.1 nM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [56] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.19 nM | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | [52] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.2 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [57] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.3 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [58] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.5 nM | Lung small cell carcinoma | H69AR cell | CVCL_3513 | [59] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.5 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [60] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.5 nM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [61] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.7 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [37] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.7 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [59] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.8 nM | Prostate carcinoma | PC-3 cell | CVCL_0035 | [62] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.9 nM | Colon adenocarcinoma | LoVo cell | CVCL_0399 | [38] | |
| Half Maximal Inhibitory Concentration (IC50) | 3 nM | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | [63] | |
| Half Maximal Inhibitory Concentration (IC50) | 3 nM | Normal | HBL-100 cell | CVCL_4362 | [64] | |
| Half Maximal Inhibitory Concentration (IC50) | 3 nM | Lymphoblastic leukemia | L1210 cell | CVCL_0382 | [65] | |
| Half Maximal Inhibitory Concentration (IC50) | 3 nM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [66] | |
| Half Maximal Inhibitory Concentration (IC50) | 3 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [67] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.1 nM | Lymphoma | P388 cell | CVCL_7222 | [43] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.1 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [68] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.162 nM | Lymphoma | P388 cell | CVCL_7222 | [69] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.2 nM | Colon adenocarcinoma | HCT 15 cell | CVCL_0292 | [70] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.3 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [71] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.4 nM | Amelanotic melanoma | MDA-MB-435 cell | CVCL_0417 | [72] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.5 nM | High grade ovarian serous adenocarcinoma | NCI-ADR-RES cell | CVCL_1452 | [73] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.5 nM | Breast ductal carcinoma | BT-549 cell | CVCL_1092 | [38] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.6 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [58] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.8 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [74] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.8 nM | Hepatocellular carcinoma | SMMC-7721 cell | CVCL_0534 | [75] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.9 nM | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | [76] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [63] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [77] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | High grade ovarian serous adenocarcinoma | OVCAR-8 cell | CVCL_1629 | [73] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [41] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [78] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 nM | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | [79] | |
| Half Maximal Inhibitory Concentration (IC50) | 4.09 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [80] | |
| Half Maximal Inhibitory Concentration (IC50) | 4.1 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [81] | |
| Half Maximal Inhibitory Concentration (IC50) | 4.3 nM | Melanoma | M21 cell | CVCL_D031 | [82] | |
| Half Maximal Inhibitory Concentration (IC50) | 4.5 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [83] | |
| Half Maximal Inhibitory Concentration (IC50) | 4.7 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [84] | |
| Half Maximal Inhibitory Concentration (IC50) | 5 nM | T acute lymphoblastic leukemia | Jurkat cell | CVCL_0065 | [85] | |
| Half Maximal Inhibitory Concentration (IC50) | 5 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [86] | |
| Half Maximal Inhibitory Concentration (IC50) | 5 nM | Amelanotic melanoma | MDA-MB-435 cell | CVCL_0417 | [87] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.1 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [88] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.5 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [38] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.6 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [82] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.6 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [87] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.7 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [83] | |
| Half Maximal Inhibitory Concentration (IC50) | 6 nM | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | [89] | |
| Half Maximal Inhibitory Concentration (IC50) | 6 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [90] | |
| Half Maximal Inhibitory Concentration (IC50) | 6 nM | Hepatocellular carcinoma | SNU-423 cell | CVCL_0366 | [79] | |
| Half Maximal Inhibitory Concentration (IC50) | 6 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [91] | |
| Half Maximal Inhibitory Concentration (IC50) | 6.05 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [80] | |
| Half Maximal Inhibitory Concentration (IC50) | 7 nM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [92] | |
| Half Maximal Inhibitory Concentration (IC50) | 7 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [76] | |
| Half Maximal Inhibitory Concentration (IC50) | 7 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [93] | |
| Half Maximal Inhibitory Concentration (IC50) | 7.2 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [94] | |
| Half Maximal Inhibitory Concentration (IC50) | 8 nM | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | [79] | |
| Half Maximal Inhibitory Concentration (IC50) | 8 nM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [95] | |
| Half Maximal Inhibitory Concentration (IC50) | 8 nM | Breast adenocarcinoma | MDA-MB-231 cell | CVCL_0062 | [96] | |
| Half Maximal Inhibitory Concentration (IC50) | 9 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [93] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.3 nM | T-lymphoblastic leukemia | MOLT-3 cell | CVCL_0624 | [48] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.3 nM | Prostate carcinoma | DU145 cell | CVCL_0105 | [62] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.5 nM | Human papillomavirus-related endocervical adenocarcinoma | KB cell | CVCL_0372 | [92] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.7 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [97] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.8 nM | Colon carcinoma | RKO cell | CVCL_0504 | [76] | |
| Half Maximal Inhibitory Concentration (IC50) | 10 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [98] | |
| Half Maximal Inhibitory Concentration (IC50) | 10 nM | Hepatoma | Bel-7402 cell | CVCL_5492 | [62] | |
| Half Maximal Inhibitory Concentration (IC50) | 10 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [96] | |
| Half Maximal Inhibitory Concentration (IC50) | 11 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [99] | |
| Half Maximal Inhibitory Concentration (IC50) | 11 nM | Hepatoma | Bel-7402 cell | CVCL_5492 | [100] | |
| Half Maximal Inhibitory Concentration (IC50) | 12 nM | Ovarian endometrioid adenocarcinoma | A2780 cell | CVCL_0134 | [76] | |
| Half Maximal Inhibitory Concentration (IC50) | 12 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [62] | |
| Half Maximal Inhibitory Concentration (IC50) | 12 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [101] | |
| Half Maximal Inhibitory Concentration (IC50) | 13 nM | Uterine sarcoma | MES-SA cell | CVCL_1404 | [102] | |
| Half Maximal Inhibitory Concentration (IC50) | 13 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [103] | |
| Half Maximal Inhibitory Concentration (IC50) | 13 nM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [104] | |
| Half Maximal Inhibitory Concentration (IC50) | 14 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [76] | |
| Half Maximal Inhibitory Concentration (IC50) | 14.2 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [105] | |
| Half Maximal Inhibitory Concentration (IC50) | 15 nM | Ileocecal adenocarcinoma | HCT-8 cell | CVCL_2478 | [106] | |
| Half Maximal Inhibitory Concentration (IC50) | 20 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [107] | |
| Half Maximal Inhibitory Concentration (IC50) | 20 nM | Burkitt lymphoma | CA46 cell | CVCL_1101 | [108] | |
| Half Maximal Inhibitory Concentration (IC50) | 21.6 nM | Bladder carcinoma | RT-112 cell | CVCL_1670 | [54] | |
| Half Maximal Inhibitory Concentration (IC50) | 22 nM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [81] | |
| Half Maximal Inhibitory Concentration (IC50) | 23.43 nM | Colon adenocarcinoma | T84 cell | CVCL_0555 | [45] | |
| Half Maximal Inhibitory Concentration (IC50) | 25 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [109] | |
| Half Maximal Inhibitory Concentration (IC50) | 29 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [46] | |
| Half Maximal Inhibitory Concentration (IC50) | 32 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [110] | |
| Half Maximal Inhibitory Concentration (IC50) | 32 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [55] | |
| Half Maximal Inhibitory Concentration (IC50) | 37 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [111] | |
| Half Maximal Inhibitory Concentration (IC50) | 41 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [112] | |
| Half Maximal Inhibitory Concentration (IC50) | 42 nM | Malignant neoplasms of the mouse mammary gland | FM3A cell | CVCL_3869 | [65] | |
| Half Maximal Inhibitory Concentration (IC50) | 42 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [113] | |
| Half Maximal Inhibitory Concentration (IC50) | 46 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [114] | |
| Half Maximal Inhibitory Concentration (IC50) | <50 nM | Pancreatic ductal adenocarcinoma | BxPC-3 cell | CVCL_0186 | [95] | |
| Half Maximal Inhibitory Concentration (IC50) | 50 nM | Mouse melanoma | B16-F10 cell | CVCL_0159 | [115] | |
| Half Maximal Inhibitory Concentration (IC50) | 52 nM | Breast carcinoma | MX1 cell | CVCL_4774 | [95] | |
| Half Maximal Inhibitory Concentration (IC50) | 71 nM | Breast carcinoma | MX1 cell | CVCL_4774 | [95] | |
| Half Maximal Inhibitory Concentration (IC50) | 80 nM | Lung large cell carcinoma | NCI-H460 cell | CVCL_0459 | [116] | |
| Half Maximal Inhibitory Concentration (IC50) | 82 nM | Gastric adenocarcinoma | MKN45 cell | CVCL_0434 | [102] | |
| Half Maximal Inhibitory Concentration (IC50) | 87.2 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [61] | |
| Half Maximal Inhibitory Concentration (IC50) | 94 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [53] | |
| Half Maximal Inhibitory Concentration (IC50) | 94.3 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [105] | |
| Half Maximal Inhibitory Concentration (IC50) | 120 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [117] | |
| Half Maximal Inhibitory Concentration (IC50) | 124 nM | Small cell lung carcinoma | DMS-79 cell | CVCL_1178 | [62] | |
| Half Maximal Inhibitory Concentration (IC50) | 160 nM | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | [118] | |
| Half Maximal Inhibitory Concentration (IC50) | 160 nM | Lymphoma | P388 cell | CVCL_7222 | [119] | |
| Half Maximal Inhibitory Concentration (IC50) | 160 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [120] | |
| Half Maximal Inhibitory Concentration (IC50) | 170 nM | Renal adenocarcinoma | ACHN cell | CVCL_1067 | [121] | |
| Half Maximal Inhibitory Concentration (IC50) | 180 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [122] | |
| Half Maximal Inhibitory Concentration (IC50) | 180 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [121] | |
| Half Maximal Inhibitory Concentration (IC50) | 190 nM | Normal | HaCaT cell | CVCL_0038 | [123] | |
| Half Maximal Inhibitory Concentration (IC50) | 190 nM | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | [124] | |
| Half Maximal Inhibitory Concentration (IC50) | 200 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [33] | |
| Half Maximal Inhibitory Concentration (IC50) | 210 nM | Amelanotic melanoma | A-375 cell | CVCL_0132 | [121] | |
| Half Maximal Inhibitory Concentration (IC50) | 240 nM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [120] | |
| Half Maximal Inhibitory Concentration (IC50) | 250 nM | Pancreatic adenocarcinoma | SW1990 cell | CVCL_1723 | [46] | |
| Half Maximal Inhibitory Concentration (IC50) | 260 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [56] | |
| Half Maximal Inhibitory Concentration (IC50) | 280 nM | Prostate carcinoma | DU145 cell | CVCL_0105 | [125] | |
| Half Maximal Inhibitory Concentration (IC50) | 330 nM | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | [120] | |
| Half Maximal Inhibitory Concentration (IC50) | 370 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [126] | |
| Half Maximal Inhibitory Concentration (IC50) | 400 nM | Prostate carcinoma | PC-3 cell | CVCL_0035 | [39] | |
| Half Maximal Inhibitory Concentration (IC50) | 410 nM | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | [127] | |
| Half Maximal Inhibitory Concentration (IC50) | 420 nM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [114] | |
| Half Maximal Inhibitory Concentration (IC50) | 430 nM | Hepatocellular carcinoma | Huh-7 cell | CVCL_0336 | [109] | |
| Half Maximal Inhibitory Concentration (IC50) | 540 nM | Astrocytoma | U-251MG cell | CVCL_0021 | [128] | |
| Half Maximal Inhibitory Concentration (IC50) | 560 nM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [129] | |
| Half Maximal Inhibitory Concentration (IC50) | 720 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [130] | |
| Half Maximal Inhibitory Concentration (IC50) | >1000 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [59] | |
| Half Maximal Inhibitory Concentration (IC50) | 1000 nM | Colon cancer | HT29 cell | CVCL_A8EZ | [90] | |
| Half Maximal Inhibitory Concentration (IC50) | 1000 nM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [131] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.3 uM | High grade ovarian serous adenocarcinoma | OVCAR-8 cell | CVCL_1629 | [132] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.718 uM | Hepatoma | Bel-7402 cell | CVCL_5492 | [46] | |
| Half Maximal Inhibitory Concentration (IC50) | 1.8 uM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [132] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.41 uM | Hepatoblastoma | Hep-G2 cell | CVCL_0027 | [129] | |
| Half Maximal Inhibitory Concentration (IC50) | 2.5 uM | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | [132] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.1 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [122] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.2 uM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [43] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.22 uM | Hepatocellular carcinoma | SMMC-7721 cell | CVCL_0534 | [114] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.27 uM | Cervical carcinoma | L02 cell | CVCL_6926 | [133] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.4 uM | Normal | HMEC-1 cell | CVCL_0307 | [134] | |
| Half Maximal Inhibitory Concentration (IC50) | 3.6 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [34] | |
| Half Maximal Inhibitory Concentration (IC50) | 4 uM | Acute myeloid leukemia | HL-60 cell | CVCL_0002 | [134] | |
| Half Maximal Inhibitory Concentration (IC50) | 4.165 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [77] | |
| Half Maximal Inhibitory Concentration (IC50) | 5.99 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [135] | |
| Half Maximal Inhibitory Concentration (IC50) | 6.1 uM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [136] | |
| Half Maximal Inhibitory Concentration (IC50) | 8.87 uM | Colon adenocarcinoma | HCT 15 cell | CVCL_0292 | [125] | |
| Half Maximal Inhibitory Concentration (IC50) | 9.12 uM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [125] | |
| Half Maximal Inhibitory Concentration (IC50) | >10 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [39] | |
| Half Maximal Inhibitory Concentration (IC50) | >10 uM | Gastric carcinoma | NUGC-3 cell | CVCL_1612 | [102] | |
| Half Maximal Inhibitory Concentration (IC50) | >10 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [39] | |
| Half Maximal Inhibitory Concentration (IC50) | 11 uM | Normal | HEK293 cell | CVCL_0045 | [115] | |
| Half Maximal Inhibitory Concentration (IC50) | >20 uM | Lung adenocarcinoma | A-549 cell | CVCL_0023 | [123] | |
| Half Maximal Inhibitory Concentration (IC50) | 25 uM | Normal | HEK293 cell | CVCL_0045 | [134] | |
| Tumor Growth Inhibition value (TGI) | 79.43 nM | Amelanotic melanoma | MDA-MB-435 cell | CVCL_0417 | [23] | |
| Tumor Growth Inhibition value (TGI) | 158.49 nM | Ovarian serous adenocarcinoma | OVCAR-3 cell | CVCL_0465 | [23] | |
| Tumor Growth Inhibition value (TGI) | 199.53 nM | Colon carcinoma | HCT 116 cell | CVCL_0291 | [23] | |
| Tumor Growth Inhibition value (TGI) | 251.19 nM | Melanoma | M14 cell | CVCL_1395 | [23] | |
| Tumor Growth Inhibition value (TGI) | 794.33 nM | Colon adenocarcinoma | KM12 cell | CVCL_1331 | [23] | |
| Tumor Growth Inhibition value (TGI) | 1.25893 uM | Glioblastoma | SF-295 cell | CVCL_1690 | [23] | |
| Tumor Growth Inhibition value (TGI) | 1.25893 uM | Colon adenocarcinoma | HCC 2998 cell | CVCL_1266 | [23] | |
| Tumor Growth Inhibition value (TGI) | 1.99526 uM | Chronic myeloid leukemia | K562 cell | CVCL_0004 | [23] | |
| Tumor Growth Inhibition value (TGI) | 1.99526 uM | Breast ductal carcinoma | BT-549 cell | CVCL_1092 | [23] | |
| Tumor Growth Inhibition value (TGI) | 2.51189 uM | Non-small cell lung carcinoma | HOP-62 cell | CVCL_1285 | [23] | |
| Tumor Growth Inhibition value (TGI) | 3.98107 uM | T acute lymphoblastic leukemia | MOLT-4 cell | CVCL_0013 | [23] | |
| Tumor Growth Inhibition value (TGI) | 3.98107 uM | Non-small cell lung carcinoma | NCI-H522 cell | CVCL_1567 | [23] | |
| Tumor Growth Inhibition value (TGI) | 5.01187 uM | T acute lymphoblastic leukemia | CCRF-CEM cell | CVCL_0207 | [23] | |
| Tumor Growth Inhibition value (TGI) | 6.30957 uM | Colon cancer | HT29 cell | CVCL_A8EZ | [23] | |
| Tumor Growth Inhibition value (TGI) | 12.58925 uM | Astrocytoma | U-251MG cell | CVCL_0021 | [23] | |
| Tumor Growth Inhibition value (TGI) | 39.81072 uM | High grade ovarian serous adenocarcinoma | OVCAR-8 cell | CVCL_1629 | [23] | |
| Tumor Growth Inhibition value (TGI) | 50.11872 uM | Renal carcinoma | UO-31 cell | CVCL_1911 | [23] | |
| Tumor Growth Inhibition value (TGI) | 50.11872 uM | Cutaneous melanoma | SK-MEL-28 cell | CVCL_0526 | [23] | |
| Tumor Growth Inhibition value (TGI) | 63.09573 uM | Ovarian endometrioid adenocarcinoma | IGROV-1 cell | CVCL_1304 | [23] | |
| Tumor Growth Inhibition value (TGI) | 63.09573 uM | Melanoma | UACC-62 cell | CVCL_1780 | [23] | |
| Tumor Growth Inhibition value (TGI) | 79.43282 uM | Renal carcinoma | RXF 393 cell | CVCL_1673 | [23] | |
| Tumor Growth Inhibition value (TGI) | 79.43282 uM | Non-small cell lung carcinoma | EKVX cell | CVCL_1195 | [23] | |
| Tumor Growth Inhibition value (TGI) | 79.43282 uM | Minimally invasive lung adenocarcinoma | NCI-H322M cell | CVCL_1557 | [23] | |
| Tumor Growth Inhibition value (TGI) | 79.43282 uM | Melanoma | Malme-3M cell | CVCL_1438 | [23] | |
| Tumor Growth Inhibition value (TGI) | 79.43282 uM | Leukemia | SR cell | CVCL_1711 | [23] | |
| Tumor Growth Inhibition value (TGI) | 79.43282 uM | Colon adenocarcinoma | SW620 cell | CVCL_0547 | [23] | |
| Tumor Growth Inhibition value (TGI) | 100 uM | Prostate carcinoma | DU145 cell | CVCL_0105 | [23] | |
| Tumor Growth Inhibition value (TGI) | 100 uM | Prostate carcinoma | PC-3 cell | CVCL_0035 | [23] | |
| Tumor Growth Inhibition value (TGI) | 100 uM | Ovarian serous cystadenocarcinoma | SK-OV-3 cell | CVCL_0532 | [23] | |
| Tumor Growth Inhibition value (TGI) | 100 uM | Invasive breast carcinoma | T-47D cell | CVCL_0553 | [23] | |
Each Peptide-drug Conjugate Related to This Drug
Full Information of The Activity Data of The PDC(s) Related to This Drug
NT4-CA4 ester [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [137] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 0.00% | |||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [137] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 20.00% | |||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [137] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 100.00% | |||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Colon adenocarcinoma | HT-29 cell | CVCL_0320 | ||
NT4-CA4 ether [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [137] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 0.00% | |||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Colon adenocarcinoma | HT-29 cell | CVCL_0320 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [137] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 0.00% | |||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [137] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Cell viability | 55.00% | |||
| Administration Time | 6 days | ||||
| Administration Dosage | 0.03 mM | ||||
| MOA of PDC |
We have been studying protease-resistant branched peptides as tumor targeting agents by using tetra-branched peptides (NT4) containing the human regulatory peptide neurotensin (NT) sequence. Neurotensin receptors are overexpressed in several human malignancies, such as colon, pancreatic, prostate and small-cell lung cancer. We have been using NT4 conjugated to different functional units for tumor imaging and therapy, and found that NT4 conjugated to methotrexate produced 60% reduction of tumor growth in xenografted mice. Results obtained with NT4 indicated that branched peptides are promising novel multifunctional targeting molecules, which might allow cancer detection and therapy by means of the same molecule, with no modification in target binding, but rather a simple exchange of functional units. Since cancer cells are very different from one another in terms of drug sensibility, not only in different tumors but in different patients and stages of the disease, this approach prefigures the synthesis of a number of constructs conjugated with differently acting chemotherapeutics. The type of linkage between effector unit (drug/imaging agent etc.) and peptide is obviously crucial for this type of approach. The choice must be driven by two issues: 1) The nature of the drug functional groups available for coupling with the peptide; and 2) the mechanism-of-action of the drug. When a prodrug acts without being released from the carrier unit, a strong linker is preferred. However, if a drug has to be released in order to interact with the intracellular target, the linker must be cleavable. In latter, the linker has to be chosen properly-not too labile or leakage will occur during drug distribution, but not too robust or the pharmacological action will be impaired.
Click to Show/Hide
|
||||
| Description |
This clearly indicated that the chemotherapeutic moiety was released into the cell medium and the drug was internalized into the cells, probably by membrane diffusion. This became evident when observing the behavior of the unrelated conjugated peptides.
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | PANC-1 cell | CVCL_0480 | ||
References
