Linker Information
General Information of This Linker
| Linker ID |
LIN00011
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
PSMA-I&T Linker
|
|||||
| Structure |
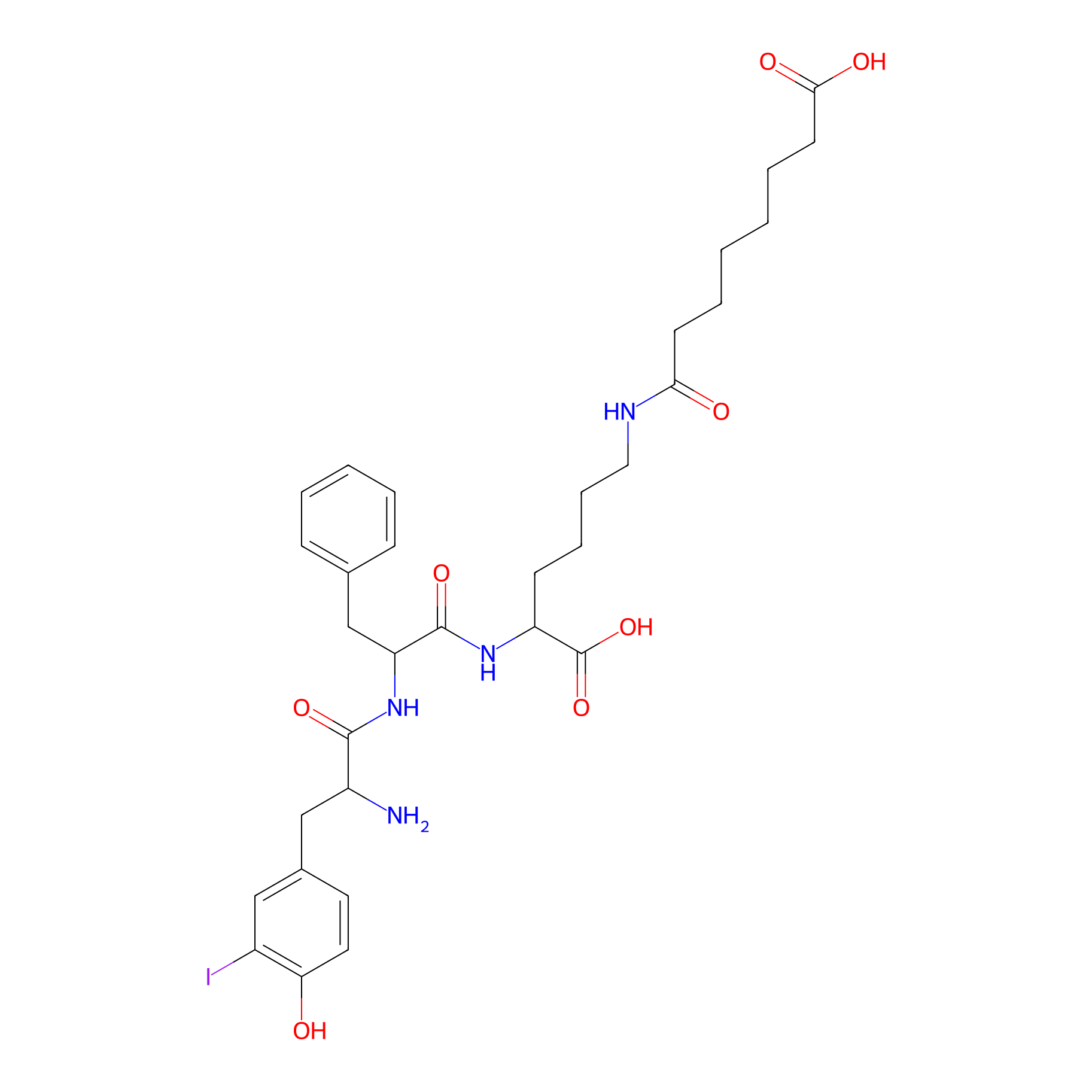
|
|||||
| Formula |
C32H43IN4O8
|
|||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight (mw) | 738.6 | ||||
| Lipid-water partition coefficient (xlogp) | 1.2 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 7 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 9 | |||||
| Rotatable Bond Count (rotbonds) | 21 | |||||
| PubChem CID | ||||||
| Canonical smiles |
C1=CC=C(C=C1)CC(C(=O)NC(CCCCNC(=O)CCCCCCC(=O)O)C(=O)O)NC(=O)C(CC2=CC(=C(C=C2)O)I)N
|
|||||
| InChI |
InChI=1S/C32H43IN4O8/c33-23-18-22(15-16-27(23)38)19-24(34)30(42)37-26(20-21-10-4-3-5-11-21)31(43)36-25(32(44)45)12-8-9-17-35-28(39)13-6-1-2-7-14-29(40)41/h3-5,10-11,15-16,18,24-26,38H,1-2,6-9,12-14,17,19-20,34H2,(H,35,39)(H,36,43)(H,37,42)(H,40,41)(H,44,45)
|
|||||
| InChIKey |
NNCBWVSCMSFHCU-UHFFFAOYSA-N
|
|||||
| IUPAC Name |
8-[[5-[[2-[[2-amino-3-(4-hydroxy-3-iodophenyl)propanoyl]amino]-3-phenylpropanoyl]amino]-5-carboxypentyl]amino]-8-oxooctanoic acid
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
177Lu-PSMA I&T [Phase 2]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Mild xerostomia |
4%
|
|||
| Patients Enrolled |
51 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 1 cycles | ||||
| Administration Dosage | 6.1 ± 1.0 GBq (range, 3.4-7.6 GBq) | ||||
| MOA of PDC |
Both 177Lu-PSMA I&T and 177Lu-PSMA-617 PRLT demonstrated favorable safety in mCRPC patients. The highest absorbed doses among healthy organs were in the lacrimal and parotid glands-not, however, resulting in any significant clinical sequel. 177Lu-PSMA-617 demonstrated a higher absorbed dose to the whole-body and lacrimal glands but a lower renal dose than did 177Lu-PSMA I&T. The mean absorbed tumor doses were comparable for both 177Lu-PSMA I&T and 177Lu-PSMA-617. There was a large interpatient variability in the dosimetry parameters. Therefore, individual patient-based dosimetry seems favorable for personalized PRLT.
Click to Show/Hide
|
||||
| Description |
Five patients (3.6%) reported mild, reversible xerostomia. 2 patients (3.9%) in the 177Lu-PSMA I&T group and 3 (3.4%) in 177Lu-PSMA-617 groupafter 2-6 cycles of treatment and in follow-up. Xerophthalmia was not reported by any patients. No other adverse symptoms were noticed during the entire follow-up period.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Median progression-free survival (mPFS) |
4.1 months
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Prostate-specific antigen decline of ≥50% was achieved in 38 patients, median clinical progression-free survival (cPFS) was 4.1 mouth, and median overall survival (OS) was 12.9 mouth.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Median overall survival (mOS) |
12.9 months
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Prostate-specific antigen decline of ≥50% was achieved in 38 patients, median clinical progression-free survival (cPFS) was 4.1 mouth, and median overall survival (OS) was 12.9 mouth.
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 2 visual salivary gland scintigraphy |
10%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
In the visual grading, no significant changes were found on SGS after PRLT (stage 0 in 16 (40%) patients, stage 1 in 16 (40%) patients, and stage 2 in 8 (20%) patients at the baseline; stage 0 in 16 (40%) patients, stage 1 in 20 (50%) patients, and stage 2 in 4 (10%) patients at the follow-up; p = 0.63). A comparison of the Umax and EF of all SG confirmed no significant changes before and after PRLT.
Click to Show/Hide
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 1 visual salivary gland scintigraphy |
50%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
In the visual grading, no significant changes were found on SGS after PRLT (stage 0 in 16 (40%) patients, stage 1 in 16 (40%) patients, and stage 2 in 8 (20%) patients at the baseline; stage 0 in 16 (40%) patients, stage 1 in 20 (50%) patients, and stage 2 in 4 (10%) patients at the follow-up; p = 0.63). A comparison of the Umax and EF of all SG confirmed no significant changes before and after PRLT.
Click to Show/Hide
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 0 visual salivary gland scintigraphy |
40%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
In the visual grading, no significant changes were found on SGS after PRLT (stage 0 in 16 (40%) patients, stage 1 in 16 (40%) patients, and stage 2 in 8 (20%) patients at the baseline; stage 0 in 16 (40%) patients, stage 1 in 20 (50%) patients, and stage 2 in 4 (10%) patients at the follow-up; p = 0.63). A comparison of the Umax and EF of all SG confirmed no significant changes before and after PRLT.
Click to Show/Hide
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 3/4 thrombocytopenia |
4%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Treatment-emergent hematologic grade 3/4 toxicities were anemia (9%), thrombocytopenia (4%), and neutropenia (6%).
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 3/4 neutropenia |
6%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Treatment-emergent hematologic grade 3/4 toxicities were anemia (9%), thrombocytopenia (4%), and neutropenia (6%).
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 3/4 anemia |
9%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Treatment-emergent hematologic grade 3/4 toxicities were anemia (9%), thrombocytopenia (4%), and neutropenia (6%).
|
||||
| Experiment 10 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 2 xerostomia |
2.50%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
From the baseline to follow-up, xerostomia became more frequent (grade 1 in 2 (5%) patients at baseline, grade 1 in 15 (37.5%) patients, and grade 2 in 1 (2.5%) patient at follow-up; p < 0.001). No grade 3 xerostomia occurred. The data of the sXI questionnaires were available only at the follow-up, showing a moderate but significant correlation to the subjective dryness of mouth (r = 0.41, p < 0.05).
Click to Show/Hide
|
||||
| Experiment 11 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 1 xerostomia |
14.30%
|
|||
| Patients Enrolled |
91 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 2 cycles | ||||
| Administration Dosage | Median 14.3 GBq, range 9.5-20.2 | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
Grade 1 xerostomia was reported in 13 (14.3%) patients at the baseline and in 22 (24.2%) patients after two cycles of Lu-177 PSMA-I&T/-617 (p < 0.01), with a correlated significant increase of the median sXI-score from 7 (IQR 5.3-9) before to 8 (IQR 6.3-11) after PRLT (p < 0.05). In addition, a moderate correlation of xerostomia symptoms and the sXI-score was found during follow-up (r = 0.43, p < 0.01).
Click to Show/Hide
|
||||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 1 xerostomia |
37.50%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
From the baseline to follow-up, xerostomia became more frequent (grade 1 in 2 (5%) patients at baseline, grade 1 in 15 (37.5%) patients, and grade 2 in 1 (2.5%) patient at follow-up; p < 0.001). No grade 3 xerostomia occurred. The data of the sXI questionnaires were available only at the follow-up, showing a moderate but significant correlation to the subjective dryness of mouth (r = 0.41, p < 0.05).
Click to Show/Hide
|
||||
| Experiment 13 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Anemia |
9.00%
|
|||
| Patients Enrolled |
1192 patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
Additionally, a recently published meta-analysis of 250 studies with a total of 1192 patients similarly found that while grade 3 and 4 toxicities were uncommon, anemia was the highest reported adverse event for both 177Lu PSMA-617 (0.19 [0.06-0.15]) and 177Lu PSMAI&T (0.09 [0.05-0.16]). Greater than 35% of patients in the treatment group of the VISION trial experienced fatigue, dry mouth, or nausea, though almost entirely grade ≤ 2 AE.
Click to Show/Hide
|
||||
References
