Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00028
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
177Lu-PSMA I&T
|
|||||
| PDC Status |
Phase 2
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
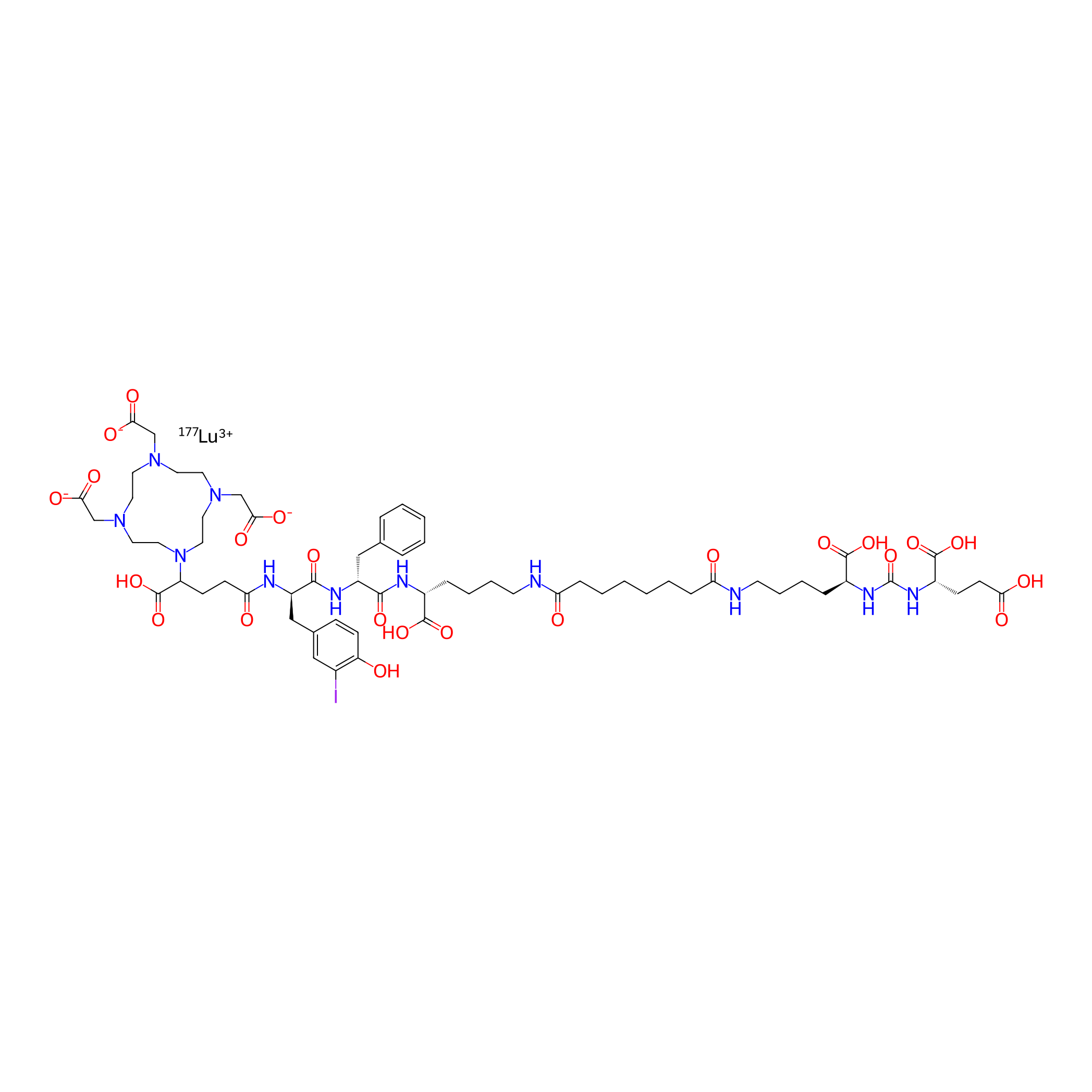
|
|||||
| Peptide Name |
PSMA I&T
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
Lutetium-177
|
Drug Info | ||||
| Therapeutic Target |
Human Deoxyribonucleic acid (hDNA)
|
Target Info | ||||
| Linker Name |
PSMA-I&T Linker
|
Linker Info | ||||
| Absorption |
Based on these findings, we then investigated the tumor accumulation of SN38-HKD and SN38-HKD/RGDR in 4T1 tumor-bearing mice. Live animal fluorescence imaging revealed that SN38-HKD/RGDR was superior to SN38-HKD in tumor accumulation after intravenous injection, and significant amount of the vehicles was observed even at 24 h after the intravenous injection.
|
|||||
| Distribution |
High performance liquid chromatography (HPLC) was then applied to directly quantify the concentrations of SN38 in the tumors and livers. We found that the concentration of SN38 in the tumors from SN38-HKD/RGDR-treated mice was 1.8- and 1.5-fold of those in tumors from mice receiving SN38-HKD at 12 and 24 h after the treatment. While SN38-HKD and SN38-HKD/RGDR had similar liver accumulation.
|
|||||
| Formula |
C63H89ILuN11O23
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1672.306758 | ||||
| Lipid-water partition coefficient (xlogp) | -3.9903 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 13 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 22 | |||||
| Rotatable Bond Count (rotbonds) | 45 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Anemia |
9.00%
|
|||
| Patients Enrolled |
1192 patients with metastatic castration-resistant prostate cancer.
|
||||
| Description |
Additionally, a recently published meta-analysis of 250 studies with a total of 1192 patients similarly found that while grade 3 and 4 toxicities were uncommon, anemia was the highest reported adverse event for both 177Lu PSMA-617 (0.19 [0.06-0.15]) and 177Lu PSMAI&T (0.09 [0.05-0.16]). Greater than 35% of patients in the treatment group of the VISION trial experienced fatigue, dry mouth, or nausea, though almost entirely grade ≤ 2 AE.
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 1 xerostomia |
14.30%
|
|||
| Patients Enrolled |
91 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 2 cycles | ||||
| Administration Dosage | Median 14.3 GBq, range 9.5-20.2 | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
Grade 1 xerostomia was reported in 13 (14.3%) patients at the baseline and in 22 (24.2%) patients after two cycles of Lu-177 PSMA-I&T/-617 (p < 0.01), with a correlated significant increase of the median sXI-score from 7 (IQR 5.3-9) before to 8 (IQR 6.3-11) after PRLT (p < 0.05). In addition, a moderate correlation of xerostomia symptoms and the sXI-score was found during follow-up (r = 0.43, p < 0.01).
Click to Show/Hide
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 1 xerostomia |
37.50%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
From the baseline to follow-up, xerostomia became more frequent (grade 1 in 2 (5%) patients at baseline, grade 1 in 15 (37.5%) patients, and grade 2 in 1 (2.5%) patient at follow-up; p < 0.001). No grade 3 xerostomia occurred. The data of the sXI questionnaires were available only at the follow-up, showing a moderate but significant correlation to the subjective dryness of mouth (r = 0.41, p < 0.05).
Click to Show/Hide
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 2 xerostomia |
2.50%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
From the baseline to follow-up, xerostomia became more frequent (grade 1 in 2 (5%) patients at baseline, grade 1 in 15 (37.5%) patients, and grade 2 in 1 (2.5%) patient at follow-up; p < 0.001). No grade 3 xerostomia occurred. The data of the sXI questionnaires were available only at the follow-up, showing a moderate but significant correlation to the subjective dryness of mouth (r = 0.41, p < 0.05).
Click to Show/Hide
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 3/4 anemia |
9%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Treatment-emergent hematologic grade 3/4 toxicities were anemia (9%), thrombocytopenia (4%), and neutropenia (6%).
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 3/4 neutropenia |
6%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Treatment-emergent hematologic grade 3/4 toxicities were anemia (9%), thrombocytopenia (4%), and neutropenia (6%).
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grade 3/4 thrombocytopenia |
4%
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Treatment-emergent hematologic grade 3/4 toxicities were anemia (9%), thrombocytopenia (4%), and neutropenia (6%).
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 0 visual salivary gland scintigraphy |
40%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
In the visual grading, no significant changes were found on SGS after PRLT (stage 0 in 16 (40%) patients, stage 1 in 16 (40%) patients, and stage 2 in 8 (20%) patients at the baseline; stage 0 in 16 (40%) patients, stage 1 in 20 (50%) patients, and stage 2 in 4 (10%) patients at the follow-up; p = 0.63). A comparison of the Umax and EF of all SG confirmed no significant changes before and after PRLT.
Click to Show/Hide
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 1 visual salivary gland scintigraphy |
50%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
In the visual grading, no significant changes were found on SGS after PRLT (stage 0 in 16 (40%) patients, stage 1 in 16 (40%) patients, and stage 2 in 8 (20%) patients at the baseline; stage 0 in 16 (40%) patients, stage 1 in 20 (50%) patients, and stage 2 in 4 (10%) patients at the follow-up; p = 0.63). A comparison of the Umax and EF of all SG confirmed no significant changes before and after PRLT.
Click to Show/Hide
|
||||
| Experiment 10 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Grading stage 2 visual salivary gland scintigraphy |
10%
|
|||
| Patients Enrolled |
40 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | A median of 5.5 (range: 2-9) cycles, median follow up 22.7 months (iqr: 16.4-30.2) | ||||
| Administration Dosage | Median cumulative activity of 35.3 GBq | ||||
| Evaluation Method | Salivary gland scintigraphy assay | ||||
| MOA of PDC |
Salivary gland dysfunction after 177Lu-PSMA-I&T/-617 PRLT has minor clinical relevance, both subjectively and objectively. Even after high cumulative activities, only mild-to-moderate dryness of mouth occurs in a minority of the patients. The prevalence of xerostomia appears to be significantly lower than the historical controls after external radiotherapy, radioiodine therapy, and especially after PRLT with 225Ac-PSMA-617. A validated questionnaire on xerostomia, salivary gland scintigraphy, and PSMA-PET/CT parameters can help to objectify, standardize, and quantify the SG toxicity of PRLT. A decrease of the excretion fraction on SGS and of the metabolic volume on PSMA PET/CT can be early indicators of SG impairment. The co-administration of lower doses of 225Ac in combination with 177Lu-PSMA-617 (Tandem concept) can decrease severe xerostomia after PRLT with alpha-emitters.
Click to Show/Hide
|
||||
| Description |
In the visual grading, no significant changes were found on SGS after PRLT (stage 0 in 16 (40%) patients, stage 1 in 16 (40%) patients, and stage 2 in 8 (20%) patients at the baseline; stage 0 in 16 (40%) patients, stage 1 in 20 (50%) patients, and stage 2 in 4 (10%) patients at the follow-up; p = 0.63). A comparison of the Umax and EF of all SG confirmed no significant changes before and after PRLT.
Click to Show/Hide
|
||||
| Experiment 11 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Median overall survival (mOS) |
12.9 months
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Prostate-specific antigen decline of ≥50% was achieved in 38 patients, median clinical progression-free survival (cPFS) was 4.1 mouth, and median overall survival (OS) was 12.9 mouth.
|
||||
| Experiment 12 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Median progression-free survival (mPFS) |
4.1 months
|
|||
| Patients Enrolled |
Patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 6-8 weekly; 6 cycle | ||||
| Administration Dosage | 7.4 GBq | ||||
| Description |
Prostate-specific antigen decline of ≥50% was achieved in 38 patients, median clinical progression-free survival (cPFS) was 4.1 mouth, and median overall survival (OS) was 12.9 mouth.
|
||||
| Experiment 13 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Metastatic castration-resistant prostate cancer | ||||
| Efficacy Data | Mild xerostomia |
4%
|
|||
| Patients Enrolled |
51 patients with metastatic castration-resistant prostate cancer.
|
||||
| Administration Time | 1 cycles | ||||
| Administration Dosage | 6.1 ± 1.0 GBq (range, 3.4-7.6 GBq) | ||||
| MOA of PDC |
Both 177Lu-PSMA I&T and 177Lu-PSMA-617 PRLT demonstrated favorable safety in mCRPC patients. The highest absorbed doses among healthy organs were in the lacrimal and parotid glands-not, however, resulting in any significant clinical sequel. 177Lu-PSMA-617 demonstrated a higher absorbed dose to the whole-body and lacrimal glands but a lower renal dose than did 177Lu-PSMA I&T. The mean absorbed tumor doses were comparable for both 177Lu-PSMA I&T and 177Lu-PSMA-617. There was a large interpatient variability in the dosimetry parameters. Therefore, individual patient-based dosimetry seems favorable for personalized PRLT.
Click to Show/Hide
|
||||
| Description |
Five patients (3.6%) reported mild, reversible xerostomia. 2 patients (3.9%) in the 177Lu-PSMA I&T group and 3 (3.4%) in 177Lu-PSMA-617 groupafter 2-6 cycles of treatment and in follow-up. Xerophthalmia was not reported by any patients. No other adverse symptoms were noticed during the entire follow-up period.
|
||||
References
