Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00135
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Dox-GLRKRLRKFRNK
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
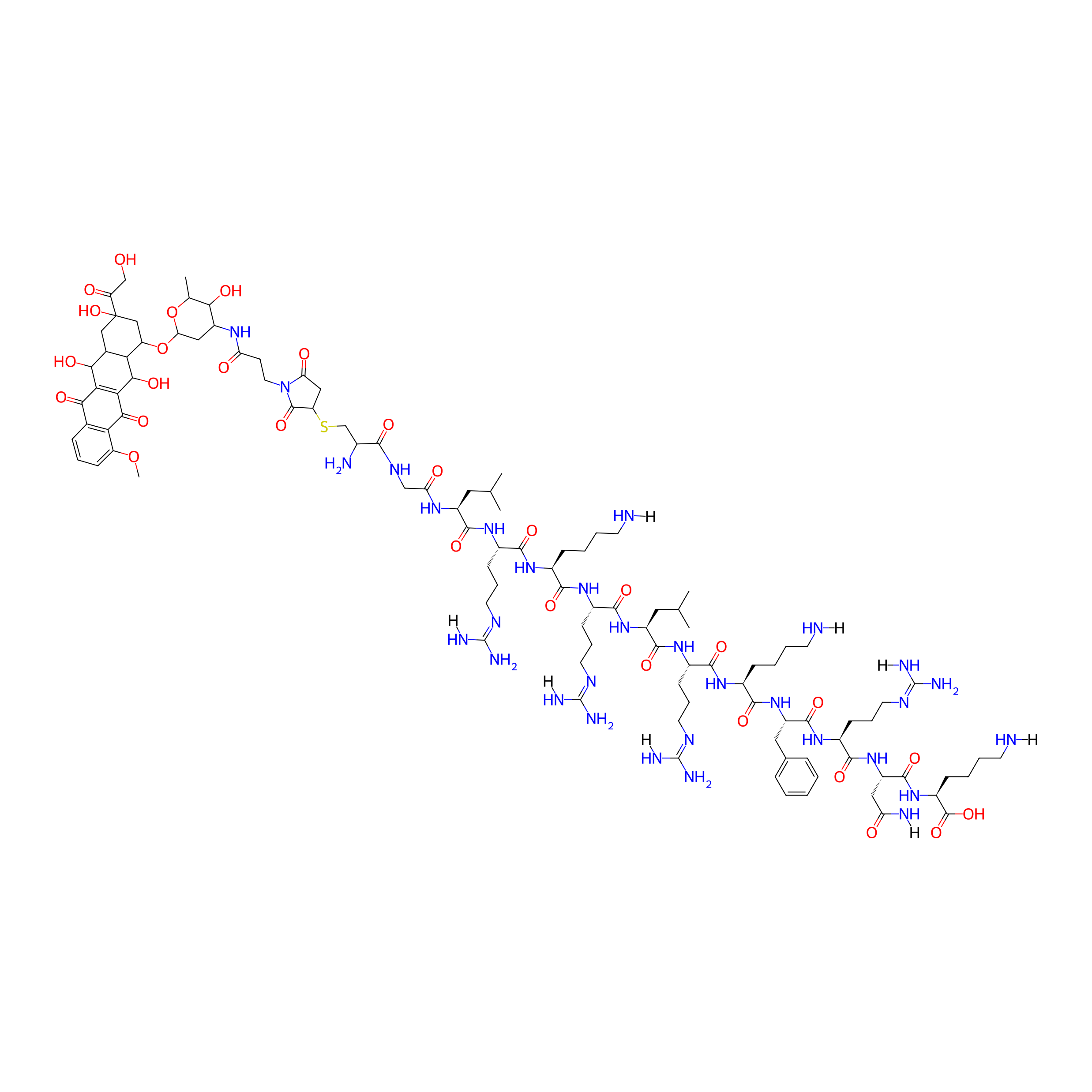
|
|||||
| Peptide Name |
GLRKRLRKFRNK
|
Peptide Info | ||||
| Receptor Name |
Cell membrane
|
|||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
S-(1-(2-carboxyethyl)-2,5-dioxopyrrolidin-3-yl)cysteine
|
Linker Info | ||||
| Formula |
C106H169N31O29S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 2373.773 | ||||
| Lipid-water partition coefficient (xlogp) | -10.2859 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 32 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 37 | |||||
| Rotatable Bond Count (rotbonds) | 76 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Cervical carcinoma | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
27.01 µM
|
|||
| Administration Time | 24 h | ||||
| MOA of PDC |
Herein, the design and synthesis of peptide-drug conjugates (PDCs) including different variants of the cell-penetrating peptide sC18 is presented. We first generated a series of novel sequence mutants of sC18 having either amino acid deletions and/or substitutions, and then tested their biological activity. The effects of histidine substituents were found to be not meaningful for sC18 uptake and cell selectivity. Moreover, building a nearly perfect amphipathic structure within a shortened sC18 derivative provided a peptide that was highly membrane-active, but also too cytotoxic. As a result, the most promising analog was sC18E, which stands out due to its higher uptake efficacy compared to parent sC18. In the last set of experiments, we let the peptides react with the cytotoxic drug doxorubicin by Thiol-Michael addition to form novel PDCs. Our results indicate that sC18E could be a more efficient drug carrier than parent sC18 for biomedical applications. However, cellular uptake using endocytosis and resulting entrapment of cargo inside vesicles is still a major critical step to overcome in CPP-containing peptide-drug development.
Click to Show/Hide
|
||||
| Description |
First, we probed the novel PDCs in non-cancerous human foreskin fibroblasts (HFF-1 cells). After 24 h treatment with different concentrations of PDCs, HFF-1 cells were still viable, while after adding doxorubicin viability was decreased up to 72%. In comparison, when we elucidated PDC activity in HeLa cells and exposed them for 24 h to various concentrations of the conjugates (2.5-70 μM), all of the PDCs, as well as free Dox, exhibited EC50 values in the lower micromolar range (PDC-1: 15.34 μM, PDC-2: 14.47 μM, PDC-3: 27.01 μM, Dox: 6.78 μM data not shown). The higher activity compared to HFF cells might be attributed to the fact that the PDCs were internalized to far less of an extent into the non-cancerous cell line. This observation might be advantageous and could reflect some selectivity of the more basic and positively charged peptides towards cancerous cells. We also noted that the obtained EC50 values somehow agreed with the results of the former assays. For example, sC18E was taken up to a significantly higher extent compared to sC18* and should, therefore, exhibit higher activity, e.g., drug delivery. However, surprisingly, the EC50 values of the PDCs containing sC18 and sC18E were quite similar, although sC18E significantly outcompeted sC18 in internalization activity.
Click to Show/Hide
|
||||
| In Vitro Model | Endocervical adenocarcinoma | HeLa cell | CVCL_0030 | ||
References
