Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00142
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Dox-S-S-GFLG-C6-[KTVRTSADE]
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
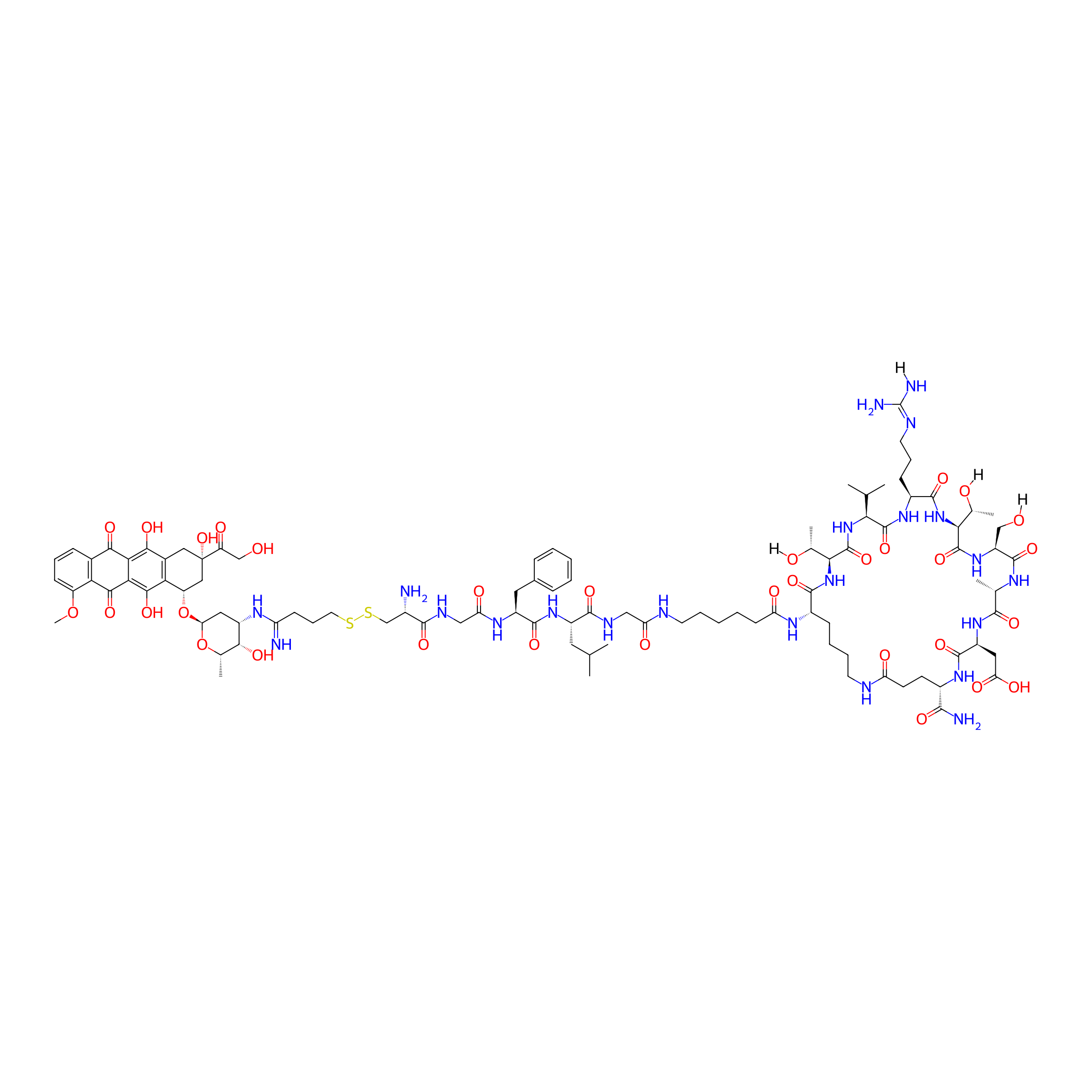
|
|||||
| Peptide Name |
KTVRTSADE
|
Peptide Info | ||||
| Receptor Name |
Fibronectin (FN1)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
S-S-GFLG-C6
|
Linker Info | ||||
| Peptide Modified Type |
Cyclization modification
|
|||||
| Modified Segment |
Head-to-tail cyclization
|
|||||
| Formula |
C99H146N22O32S2
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 2220.513 | ||||
| Lipid-water partition coefficient (xlogp) | -6.84733 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 30 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 36 | |||||
| Rotatable Bond Count (rotbonds) | 47 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
22%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
Dox conjugate 13 was moderately toxic with a reduced cell proliferation to a range of 25-35% as compared to Dox which reduced cell proliferation in the range of 20-34% for all selected four cell lines. However, it was interesting to observe that Doce conjugate 14 was almost nontoxic (cell proliferation within the range of 89-96%) in all the cell lines as compared to Doce alone which reduced the cell proliferation in the range of 54-61% (Figure 9).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP cell | CVCL_0395 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
25%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
Dox conjugate 13 was moderately toxic with a reduced cell proliferation to a range of 25-35% as compared to Dox which reduced cell proliferation in the range of 20-34% for all selected four cell lines. However, it was interesting to observe that Doce conjugate 14 was almost nontoxic (cell proliferation within the range of 89-96%) in all the cell lines as compared to Doce alone which reduced the cell proliferation in the range of 54-61% (Figure 9).
Click to Show/Hide
|
||||
| In Vitro Model | Normal | RWPE-1 cell | CVCL_3791 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
28%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
Dox conjugate 13 was moderately toxic with a reduced cell proliferation to a range of 25-35% as compared to Dox which reduced cell proliferation in the range of 20-34% for all selected four cell lines. However, it was interesting to observe that Doce conjugate 14 was almost nontoxic (cell proliferation within the range of 89-96%) in all the cell lines as compared to Doce alone which reduced the cell proliferation in the range of 54-61% (Figure 9).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | DU145 cell | CVCL_0105 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
38%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| Description |
Dox conjugate 13 was moderately toxic with a reduced cell proliferation to a range of 25-35% as compared to Dox which reduced cell proliferation in the range of 20-34% for all selected four cell lines. However, it was interesting to observe that Doce conjugate 14 was almost nontoxic (cell proliferation within the range of 89-96%) in all the cell lines as compared to Doce alone which reduced the cell proliferation in the range of 54-61% (Figure 9).
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
48%
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTS assay | ||||
| Description |
An increase in the cytotoxicity of Dox and Dox/peptide 9 physical mixture as compared to the conjugate 13 over an incubation period of 24 h to 72 h. Conjugates 13 and 14 were found to be less cytotoxic as compared to drug alone in 24-72 h. These cells were not treated with TGF-, so very minimal or no overexpression of EDB-FN. Figure 10b showed the effect of overexpression of EDB-FN in the cell viability. There was no observed effect of TGF- treatment for the cytotoxicity of Dox and physical mixture of Dox/peptide 9 on the cell viability as compared to the TGF- untreated cell lines. However, conjugate 13 showed a decrease in cell viability by 17% after 72 h as compared to untreated cell lines. Similarly, Doce and Doce conjugate 14 showed decrease in cell viability by 16 and 10%, respectively, after 72 h. The physical mixtures of Doce/peptide 9 showed a decrease in cell viability by 16% as compared to untreated cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
70%
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | MTS assay | ||||
| Description |
An increase in the cytotoxicity of Dox and Dox/peptide 9 physical mixture as compared to the conjugate 13 over an incubation period of 24 h to 72 h. Conjugates 13 and 14 were found to be less cytotoxic as compared to drug alone in 24-72 h. These cells were not treated with TGF-, so very minimal or no overexpression of EDB-FN. Figure 10b showed the effect of overexpression of EDB-FN in the cell viability. There was no observed effect of TGF- treatment for the cytotoxicity of Dox and physical mixture of Dox/peptide 9 on the cell viability as compared to the TGF- untreated cell lines. However, conjugate 13 showed a decrease in cell viability by 17% after 72 h as compared to untreated cell lines. Similarly, Doce and Doce conjugate 14 showed decrease in cell viability by 16 and 10%, respectively, after 72 h. The physical mixtures of Doce/peptide 9 showed a decrease in cell viability by 16% as compared to untreated cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
74%
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 100 nM | ||||
| Description |
The conjugate 13, peptide 9, and Dox showed no significant toxicity at or below 10 μM, possibly due to the shorter incubation time of 2 h.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
75%
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 1 µM | ||||
| Description |
The conjugate 13, peptide 9, and Dox showed no significant toxicity at or below 10 μM, possibly due to the shorter incubation time of 2 h.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
77%
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTS assay | ||||
| Description |
An increase in the cytotoxicity of Dox and Dox/peptide 9 physical mixture as compared to the conjugate 13 over an incubation period of 24 h to 72 h. Conjugates 13 and 14 were found to be less cytotoxic as compared to drug alone in 24-72 h. These cells were not treated with TGF-, so very minimal or no overexpression of EDB-FN. Figure 10b showed the effect of overexpression of EDB-FN in the cell viability. There was no observed effect of TGF- treatment for the cytotoxicity of Dox and physical mixture of Dox/peptide 9 on the cell viability as compared to the TGF- untreated cell lines. However, conjugate 13 showed a decrease in cell viability by 17% after 72 h as compared to untreated cell lines. Similarly, Doce and Doce conjugate 14 showed decrease in cell viability by 16 and 10%, respectively, after 72 h. The physical mixtures of Doce/peptide 9 showed a decrease in cell viability by 16% as compared to untreated cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
78%
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTS assay | ||||
| Description |
An increase in the cytotoxicity of Dox and Dox/peptide 9 physical mixture as compared to the conjugate 13 over an incubation period of 24 h to 72 h. Conjugates 13 and 14 were found to be less cytotoxic as compared to drug alone in 24-72 h. These cells were not treated with TGF-, so very minimal or no overexpression of EDB-FN. Figure 10b showed the effect of overexpression of EDB-FN in the cell viability. There was no observed effect of TGF- treatment for the cytotoxicity of Dox and physical mixture of Dox/peptide 9 on the cell viability as compared to the TGF- untreated cell lines. However, conjugate 13 showed a decrease in cell viability by 17% after 72 h as compared to untreated cell lines. Similarly, Doce and Doce conjugate 14 showed decrease in cell viability by 16 and 10%, respectively, after 72 h. The physical mixtures of Doce/peptide 9 showed a decrease in cell viability by 16% as compared to untreated cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
| Experiment 11 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
98%
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 100 µM | ||||
| Description |
The conjugate 13, peptide 9, and Dox showed no significant toxicity at or below 10 μM, possibly due to the shorter incubation time of 2 h.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 12 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
102%
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 10 nM | ||||
| Description |
The conjugate 13, peptide 9, and Dox showed no significant toxicity at or below 10 μM, possibly due to the shorter incubation time of 2 h.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 13 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
105%
|
|||
| Administration Time | 2 h | ||||
| Administration Dosage | 10 µM | ||||
| Description |
The conjugate 13, peptide 9, and Dox showed no significant toxicity at or below 10 μM, possibly due to the shorter incubation time of 2 h.
|
||||
| In Vitro Model | Prostate carcinoma | PC-3 cell | CVCL_0035 | ||
| Experiment 14 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
108%
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTS assay | ||||
| Description |
An increase in the cytotoxicity of Dox and Dox/peptide 9 physical mixture as compared to the conjugate 13 over an incubation period of 24 h to 72 h. Conjugates 13 and 14 were found to be less cytotoxic as compared to drug alone in 24-72 h. These cells were not treated with TGF-, so very minimal or no overexpression of EDB-FN. Figure 10b showed the effect of overexpression of EDB-FN in the cell viability. There was no observed effect of TGF- treatment for the cytotoxicity of Dox and physical mixture of Dox/peptide 9 on the cell viability as compared to the TGF- untreated cell lines. However, conjugate 13 showed a decrease in cell viability by 17% after 72 h as compared to untreated cell lines. Similarly, Doce and Doce conjugate 14 showed decrease in cell viability by 16 and 10%, respectively, after 72 h. The physical mixtures of Doce/peptide 9 showed a decrease in cell viability by 16% as compared to untreated cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
| Experiment 15 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Prostate cancer | ||||
| Efficacy Data | Cell viability |
110%
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTS assay | ||||
| Description |
An increase in the cytotoxicity of Dox and Dox/peptide 9 physical mixture as compared to the conjugate 13 over an incubation period of 24 h to 72 h. Conjugates 13 and 14 were found to be less cytotoxic as compared to drug alone in 24-72 h. These cells were not treated with TGF-, so very minimal or no overexpression of EDB-FN. Figure 10b showed the effect of overexpression of EDB-FN in the cell viability. There was no observed effect of TGF- treatment for the cytotoxicity of Dox and physical mixture of Dox/peptide 9 on the cell viability as compared to the TGF- untreated cell lines. However, conjugate 13 showed a decrease in cell viability by 17% after 72 h as compared to untreated cell lines. Similarly, Doce and Doce conjugate 14 showed decrease in cell viability by 16 and 10%, respectively, after 72 h. The physical mixtures of Doce/peptide 9 showed a decrease in cell viability by 16% as compared to untreated cells.
Click to Show/Hide
|
||||
| In Vitro Model | Prostate carcinoma | LNCaP C4-2 cell | CVCL_4782 | ||
References
