Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00212
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
HR97-SunitiGel
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
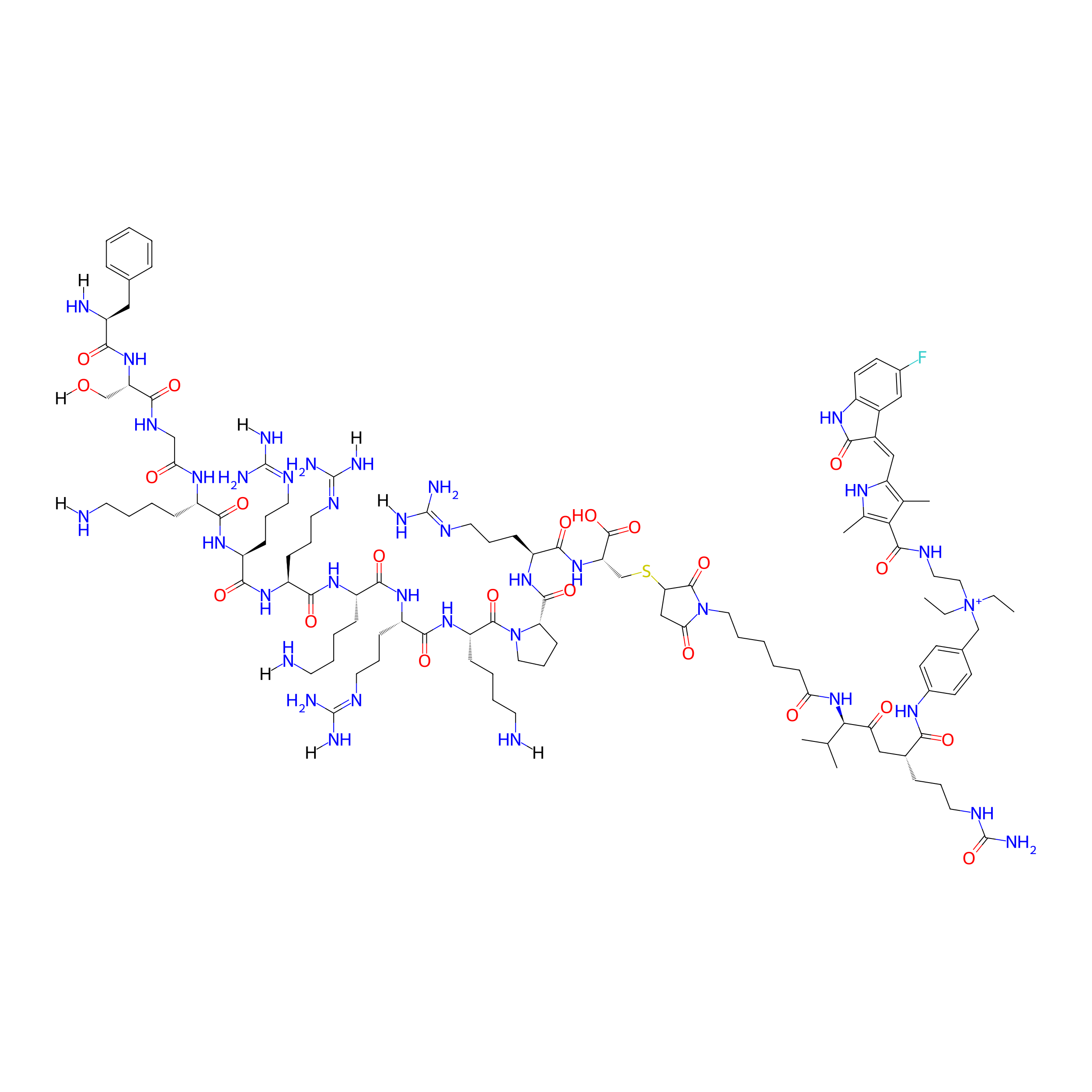
|
|||||
| Peptide Name |
HR97
|
Peptide Info | ||||
| Receptor Name |
Cell membrane
|
|||||
| Drug Name |
Sunitinib
|
Drug Info | ||||
| Therapeutic Target |
Vascular endothelial growth factor receptor 2 (KDR)
|
Target Info | ||||
| Linker Name |
Mc-Val-Cit-PAB-OH
|
Linker Info | ||||
| Formula |
C115H182FN36O22S+
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 2472.016 | ||||
| Lipid-water partition coefficient (xlogp) | -3.70706 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 31 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 30 | |||||
| Rotatable Bond Count (rotbonds) | 87 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Retinal injury | ||||
| Efficacy Data | RGC survival |
692.2 ± 96.58 RGCs/mm2
|
|||
| Administration Time | 4 week | ||||
| MOA of PDC |
In this work, we hypothesized that conjugation of the engineered multifunctional peptide adaptors to sunitinib for delivery to the posterior segment using the gel-forming eye drop would provide even more prolong therapeutic effects in the posterior tissues. We observed that the HR97-sunitinib conjugate had increased binding capacity to ocular melanin and was cleaved by proteases to release free sunitinib in vitro. Rats were dosed topically with HR97-SunitiGel once daily for seven days, followed by optic nerve head crush at various times after the last dose to assess the duration of RGC protection. We observed that the HR97-SunitiGel showed prolonged neuroprotective effects for up to 2 weeks after the last topical dose, whereas the protective effect of SunitiGel was only observed at 1 week after the last dose. Our observations support the potential for improving and prolonging therapeutic delivery to the posterior segment tissues by addressing multiple barriers to drug delivery and retention in the eye.
Click to Show/Hide
|
||||
| Description |
We next tested the potential duration of neuroprotection after topical dosing of HR97-SunitiGel. Brown Norway rats were dosed with HR97-SunitiGel or SunitiGel daily for 7 days, the optic nerve head crush procedure was performed on day 0, 7, or 21 after the last topical dose, and the RGC survival was characterized 7 days after the injury. The RGC quantification results computed by the cell counting program showed that the neuroprotective effect of HR97-SunitiGel lasted for at least 2 weeks after the last dose (869.2 ± 58.86 RGCs/mm2 compared to sham, 623.7 ± 70.39 RGCs/mm2), with the effect waning 4 weeks after the last dose (692.2 ± 96.58 RGCs/mm2). In contrast, SunitiGel provided significant RGC protection at 1 week (846.4 ± 125.8 RGCs/mm2) compared to the sham group, with protection waning 2 weeks after the last dose (717.3 ± 59.94 RGCs/mm2).
Click to Show/Hide
|
||||
| In Vivo Model | Brown norway rat optic nerve head (ONH) crush model. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Retinal injury | ||||
| Efficacy Data | RGC survival |
869.2 ± 58.86 RGCs/mm2
|
|||
| Administration Time | 2 week | ||||
| MOA of PDC |
In this work, we hypothesized that conjugation of the engineered multifunctional peptide adaptors to sunitinib for delivery to the posterior segment using the gel-forming eye drop would provide even more prolong therapeutic effects in the posterior tissues. We observed that the HR97-sunitinib conjugate had increased binding capacity to ocular melanin and was cleaved by proteases to release free sunitinib in vitro. Rats were dosed topically with HR97-SunitiGel once daily for seven days, followed by optic nerve head crush at various times after the last dose to assess the duration of RGC protection. We observed that the HR97-SunitiGel showed prolonged neuroprotective effects for up to 2 weeks after the last topical dose, whereas the protective effect of SunitiGel was only observed at 1 week after the last dose. Our observations support the potential for improving and prolonging therapeutic delivery to the posterior segment tissues by addressing multiple barriers to drug delivery and retention in the eye.
Click to Show/Hide
|
||||
| Description |
We next tested the potential duration of neuroprotection after topical dosing of HR97-SunitiGel. Brown Norway rats were dosed with HR97-SunitiGel or SunitiGel daily for 7 days, the optic nerve head crush procedure was performed on day 0, 7, or 21 after the last topical dose, and the RGC survival was characterized 7 days after the injury. The RGC quantification results computed by the cell counting program showed that the neuroprotective effect of HR97-SunitiGel lasted for at least 2 weeks after the last dose (869.2 ± 58.86 RGCs/mm2 compared to sham, 623.7 ± 70.39 RGCs/mm2), with the effect waning 4 weeks after the last dose (692.2 ± 96.58 RGCs/mm2). In contrast, SunitiGel provided significant RGC protection at 1 week (846.4 ± 125.8 RGCs/mm2) compared to the sham group, with protection waning 2 weeks after the last dose (717.3 ± 59.94 RGCs/mm2).
Click to Show/Hide
|
||||
| In Vivo Model | Brown norway rat optic nerve head (ONH) crush model. | ||||
References
