Linker Information
General Information of This Linker
| Linker ID |
LIN00065
|
|||||
|---|---|---|---|---|---|---|
| Linker Name |
Mc-Val-Cit-PAB-OH
|
|||||
| Linker Type |
Enzyme-sensitive linkers
|
|||||
| Structure |
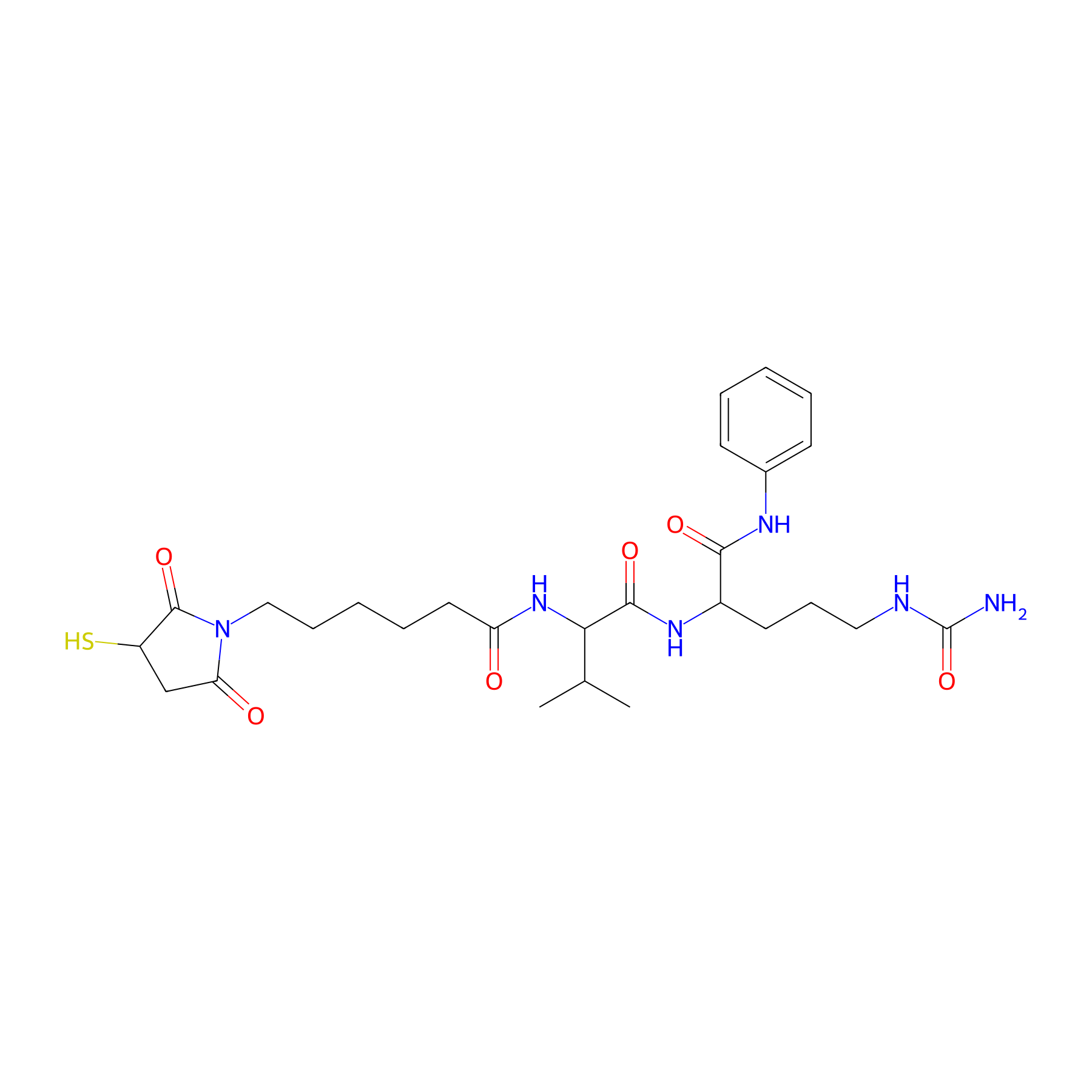
|
|||||
| Formula |
C27H40N6O6S
|
|||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight (mw) | 576.72 | ||||
| Lipid-water partition coefficient (xlogp) | 1.3169 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 6 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 7 | |||||
| Rotatable Bond Count (rotbonds) | 16 | |||||
| Canonical smiles |
CC(C)C(NC(=O)CCCCCN1C(=O)CC(S)C1=O)C(=O)NC(CCCNC(N)=O)C(=O)Nc1ccccc1
|
|||||
| InChI |
InChI=1S/C27H40N6O6S/c1-17(2)23(32-21(34)13-7-4-8-15-33-22(35)16-20(40)26(33)38)25(37)31-19(12-9-14-29-27(28)39)24(36)30-18-10-5-3-6-11-18/h3,5-6,10-11,17,19-20,23,40H,4,7-9,12-16H2,1-2H3,(H,30,36)(H,31,37)(H,32,34)(H3,28,29,39)
|
|||||
| InChIKey |
ZNRCNOKAUOKGAT-UHFFFAOYSA-N
|
|||||
Each Peptide-drug Conjugate Related to This Linker
Full Information of The Activity Data of The PDC(s) Related to This Linker
CBP-1008 [Phase 2/3]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Stable disease (SD) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Stable disease (SD) |
34.50%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Progressive disease (PD) |
17.20%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Progressive disease (PD) |
37.40%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Partial response (PR) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Partial response (PR) |
48.30%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Objective response rate(ORR) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Objective response rate(ORR) |
48.30%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Disease control rate (DCR) |
62.50%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Disease control rate (DCR) |
82.80%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
CBP-1018 [Phase 1]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | WBC decrease |
40%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Stable disease (SD) |
29.40%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Non-progressive disease (Non-PD) |
41.10%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Neutrophil decrease |
50%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Hypokalemia |
10%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Hypertriglyceridaemia |
10%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Grade ≥3 treatment-emergent adverse event rate |
75%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
HR97-SunitiGel [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Retinal injury | ||||
| Efficacy Data | RGC survival |
692.2 ± 96.58 RGCs/mm2
|
|||
| Administration Time | 4 week | ||||
| MOA of PDC |
In this work, we hypothesized that conjugation of the engineered multifunctional peptide adaptors to sunitinib for delivery to the posterior segment using the gel-forming eye drop would provide even more prolong therapeutic effects in the posterior tissues. We observed that the HR97-sunitinib conjugate had increased binding capacity to ocular melanin and was cleaved by proteases to release free sunitinib in vitro. Rats were dosed topically with HR97-SunitiGel once daily for seven days, followed by optic nerve head crush at various times after the last dose to assess the duration of RGC protection. We observed that the HR97-SunitiGel showed prolonged neuroprotective effects for up to 2 weeks after the last topical dose, whereas the protective effect of SunitiGel was only observed at 1 week after the last dose. Our observations support the potential for improving and prolonging therapeutic delivery to the posterior segment tissues by addressing multiple barriers to drug delivery and retention in the eye.
Click to Show/Hide
|
||||
| Description |
We next tested the potential duration of neuroprotection after topical dosing of HR97-SunitiGel. Brown Norway rats were dosed with HR97-SunitiGel or SunitiGel daily for 7 days, the optic nerve head crush procedure was performed on day 0, 7, or 21 after the last topical dose, and the RGC survival was characterized 7 days after the injury. The RGC quantification results computed by the cell counting program showed that the neuroprotective effect of HR97-SunitiGel lasted for at least 2 weeks after the last dose (869.2 ± 58.86 RGCs/mm2 compared to sham, 623.7 ± 70.39 RGCs/mm2), with the effect waning 4 weeks after the last dose (692.2 ± 96.58 RGCs/mm2). In contrast, SunitiGel provided significant RGC protection at 1 week (846.4 ± 125.8 RGCs/mm2) compared to the sham group, with protection waning 2 weeks after the last dose (717.3 ± 59.94 RGCs/mm2).
Click to Show/Hide
|
||||
| In Vivo Model | Brown norway rat optic nerve head (ONH) crush model. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Retinal injury | ||||
| Efficacy Data | RGC survival |
869.2 ± 58.86 RGCs/mm2
|
|||
| Administration Time | 2 week | ||||
| MOA of PDC |
In this work, we hypothesized that conjugation of the engineered multifunctional peptide adaptors to sunitinib for delivery to the posterior segment using the gel-forming eye drop would provide even more prolong therapeutic effects in the posterior tissues. We observed that the HR97-sunitinib conjugate had increased binding capacity to ocular melanin and was cleaved by proteases to release free sunitinib in vitro. Rats were dosed topically with HR97-SunitiGel once daily for seven days, followed by optic nerve head crush at various times after the last dose to assess the duration of RGC protection. We observed that the HR97-SunitiGel showed prolonged neuroprotective effects for up to 2 weeks after the last topical dose, whereas the protective effect of SunitiGel was only observed at 1 week after the last dose. Our observations support the potential for improving and prolonging therapeutic delivery to the posterior segment tissues by addressing multiple barriers to drug delivery and retention in the eye.
Click to Show/Hide
|
||||
| Description |
We next tested the potential duration of neuroprotection after topical dosing of HR97-SunitiGel. Brown Norway rats were dosed with HR97-SunitiGel or SunitiGel daily for 7 days, the optic nerve head crush procedure was performed on day 0, 7, or 21 after the last topical dose, and the RGC survival was characterized 7 days after the injury. The RGC quantification results computed by the cell counting program showed that the neuroprotective effect of HR97-SunitiGel lasted for at least 2 weeks after the last dose (869.2 ± 58.86 RGCs/mm2 compared to sham, 623.7 ± 70.39 RGCs/mm2), with the effect waning 4 weeks after the last dose (692.2 ± 96.58 RGCs/mm2). In contrast, SunitiGel provided significant RGC protection at 1 week (846.4 ± 125.8 RGCs/mm2) compared to the sham group, with protection waning 2 weeks after the last dose (717.3 ± 59.94 RGCs/mm2).
Click to Show/Hide
|
||||
| In Vivo Model | Brown norway rat optic nerve head (ONH) crush model. | ||||
HR97-brimonidine [Investigative]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Glaucoma | ||||
| Efficacy Data | Duration of lower intraocular pressure |
18 Days
|
|||
| Evaluation Method | Hand-held rebound tonometer icareTONOVET assay | ||||
| MOA of PDC |
Sustained drug delivery strategies have many potential benefits for treating a range of diseases, particularly chronic diseases that require treatment for years. For many chronic ocular diseases, patient adherence to eye drop dosing regimens and the need for frequent intraocular injections are significant barriers to effective disease management. Here, we utilize peptide engineering to impart melanin binding properties to peptide-drug conjugates to act as a sustained-release depot in the eye. We develop a super learning-based methodology to engineer multifunctional peptides that efficiently enter cells, bind to melanin, and have low cytotoxicity. When the lead multifunctional peptide (HR97) is conjugated to brimonidine, an intraocular pressure lowering drug that is prescribed for three times per day topical dosing, intraocular pressure reduction is observed for up to 18 days after a single intracameral injection in rabbits. Further, the cumulative intraocular pressure lowering effect increases ~17-fold compared to free brimonidine injection. Engineered multifunctional peptide-drug conjugates are a promising approach for providing sustained therapeutic delivery in the eye and beyond.
Click to Show/Hide
|
||||
| Description |
The HR97-brimonidine conjugate provided up to 18 days of IOP lowering with a single ICM injection in normotensive rabbits, which contrasts with the 8 h-effect provided by a brimonidine eye drop.
|
||||
| In Vivo Model | Dutch belted rabbits (2-3 kg) model. | ||||
References
