Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00367
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
CBP-1018
|
|||||
| PDC Status |
Phase 1
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
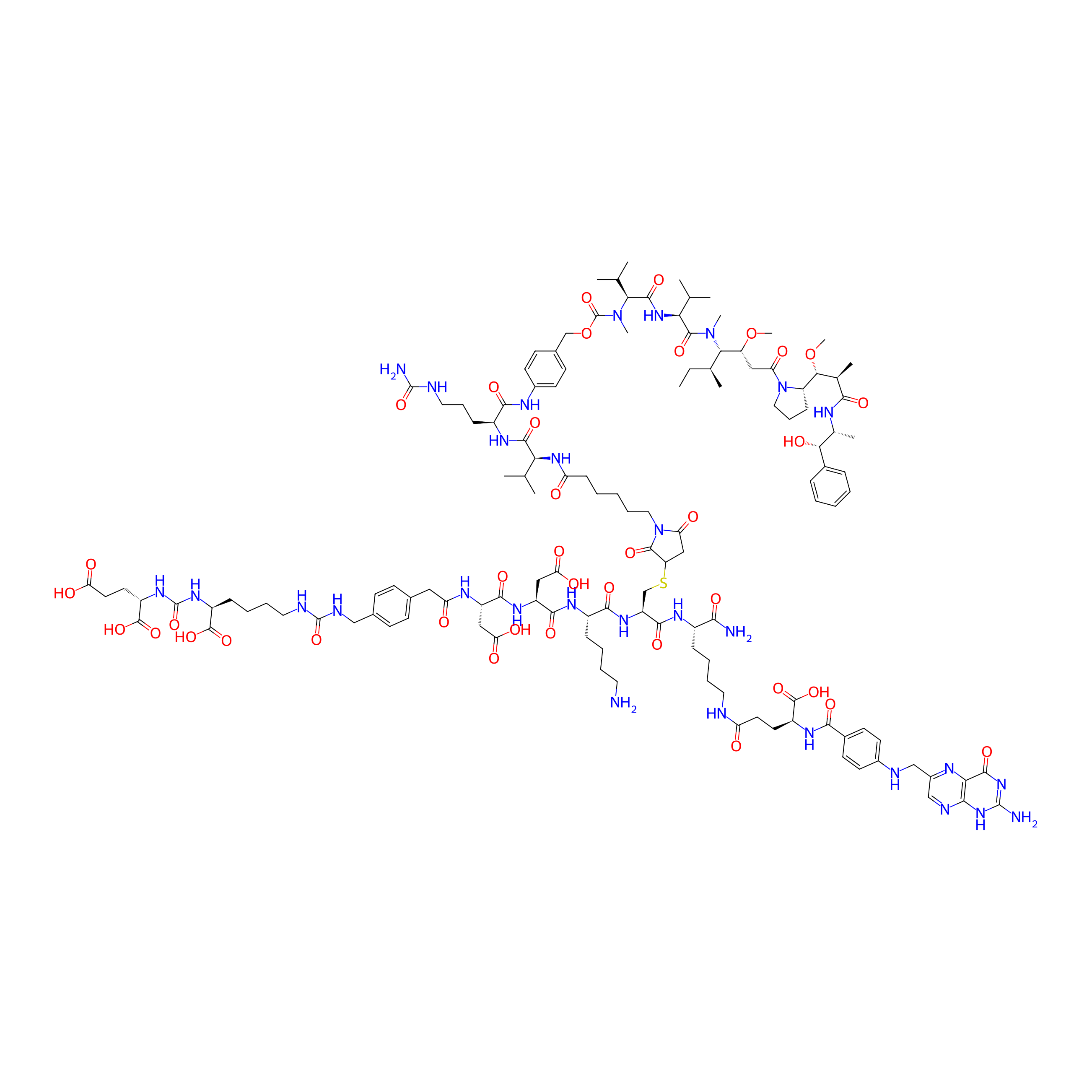
|
|||||
| Peptide Name |
LDC10B
|
Peptide Info | ||||
| Receptor Name |
Glutamate carboxypeptidase 2 (FOLH1)
|
Receptor Info | ||||
| Drug Name |
Monomethyl auristatin E
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Mc-Val-Cit-PAB-OH
|
Linker Info | ||||
| Peptide Modified Type |
The modification of binding with chemical molecules
|
|||||
| Modified Segment |
Urea; 2-(4-(aminomethyl)phenyl)acetic acid
|
|||||
| Formula |
C132H192N30O38S
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 2839.227 | ||||
| Lipid-water partition coefficient (xlogp) | 1.4537 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 30 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 39 | |||||
| Rotatable Bond Count (rotbonds) | 89 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Grade ≥3 treatment-emergent adverse event rate |
75%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Hypertriglyceridaemia |
10%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Hypokalemia |
10%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Neutrophil decrease |
50%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Non-progressive disease (Non-PD) |
41.10%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Stable disease (SD) |
29.40%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | WBC decrease |
40%
|
|||
| Patients Enrolled |
20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma).
|
||||
| Administration Time | 4 week | ||||
| Administration Dosage | 0.03 mg/kg | ||||
| MOA of PDC |
Prostate-specific membrane antigen (PSMA) is highly expressed on prostate cancer and folate receptor α (FRα) overexpressed in various malignant tissues which both related to tumor invasiveness. CBP-1018 is a first-in-class bi-ligand-drug conjugate targeting both PSMA and FRα with monomethyl auristatin E (MMAE) as payload.
|
||||
| Description |
As of 27 April 2023, 20 patients (18 mCRPC, 1 bladder cancer and 1 ureteral carcinoma) were enrolled at 6 dose levels (DLs). No DLTs or drug-related deaths were observed. For 15 patients (75%) experienced treatment-related adverse events (TRAEs) ≥ grade 3, most common were neutrophil decrease (50%), WBC decrease (40%), hypokalemia (10%) and hypertriglyceridaemia (10%). Among 17 evaluable mCRPC patients, 5 SD and 7 Non-PD were observed with 9 patients delayed administration and 6 patients dropped for Covid-19. Prostate-specific antigen (PSA) 50% decrease was detected in 2 patients. The median PFS was 9.2 months (95%CI, 1.7-9.2) in mCRPC patients. For PK profile of CBP-1018 and free MMAE, t1/2z was ranged 0.54-1.15 h and 38.27-57.27 h, respectively, no accumulation of both substances after multiple doses.
Click to Show/Hide
|
||||
| Half life period | 0.54-1.15 h | ||||
References
