Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00366
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
CBP-1008
|
|||||
| PDC Status |
Phase 2/3
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Structure |
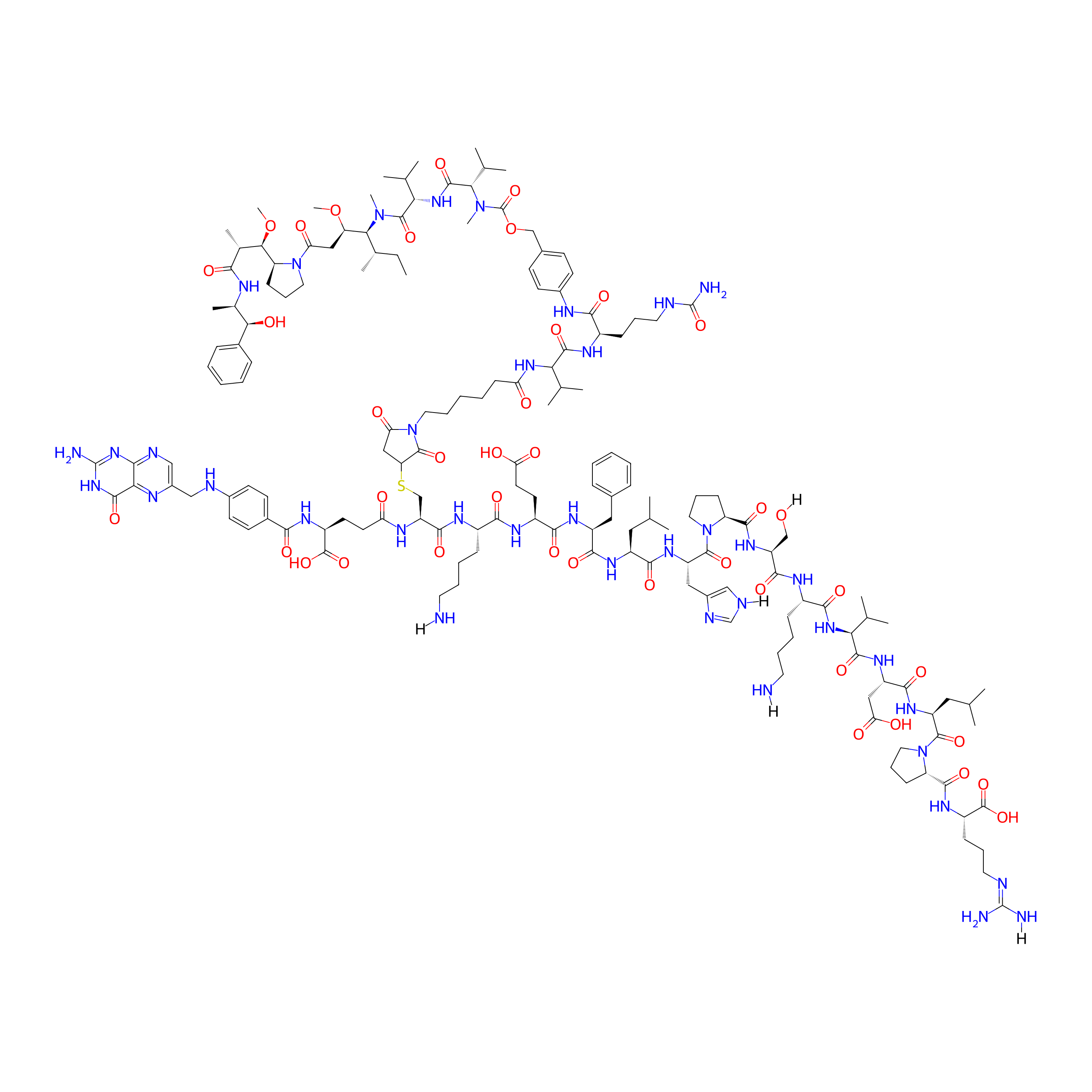
|
|||||
| Peptide Name |
SOR-C13
|
Peptide Info | ||||
| Receptor Name |
Transient receptor potential cation channel subfamily V member 6 (TRPV6)
|
Receptor Info | ||||
| Drug Name |
Monomethyl auristatin E
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Mc-Val-Cit-PAB-OH
|
Linker Info | ||||
| Formula |
C162H243N39O40S
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 3409.026 | ||||
| Lipid-water partition coefficient (xlogp) | 0.3812 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 34 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 46 | |||||
| Rotatable Bond Count (rotbonds) | 106 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Disease control rate (DCR) |
62.50%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Disease control rate (DCR) |
82.80%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Objective response rate(ORR) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Objective response rate(ORR) |
48.30%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Partial response (PR) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Partial response (PR) |
48.30%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Progressive disease (PD) |
17.20%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Progressive disease (PD) |
37.40%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Stable disease (SD) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Stable disease (SD) |
34.50%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
References
