Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00180
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
SOR-C13
|
|||||
| Structure |
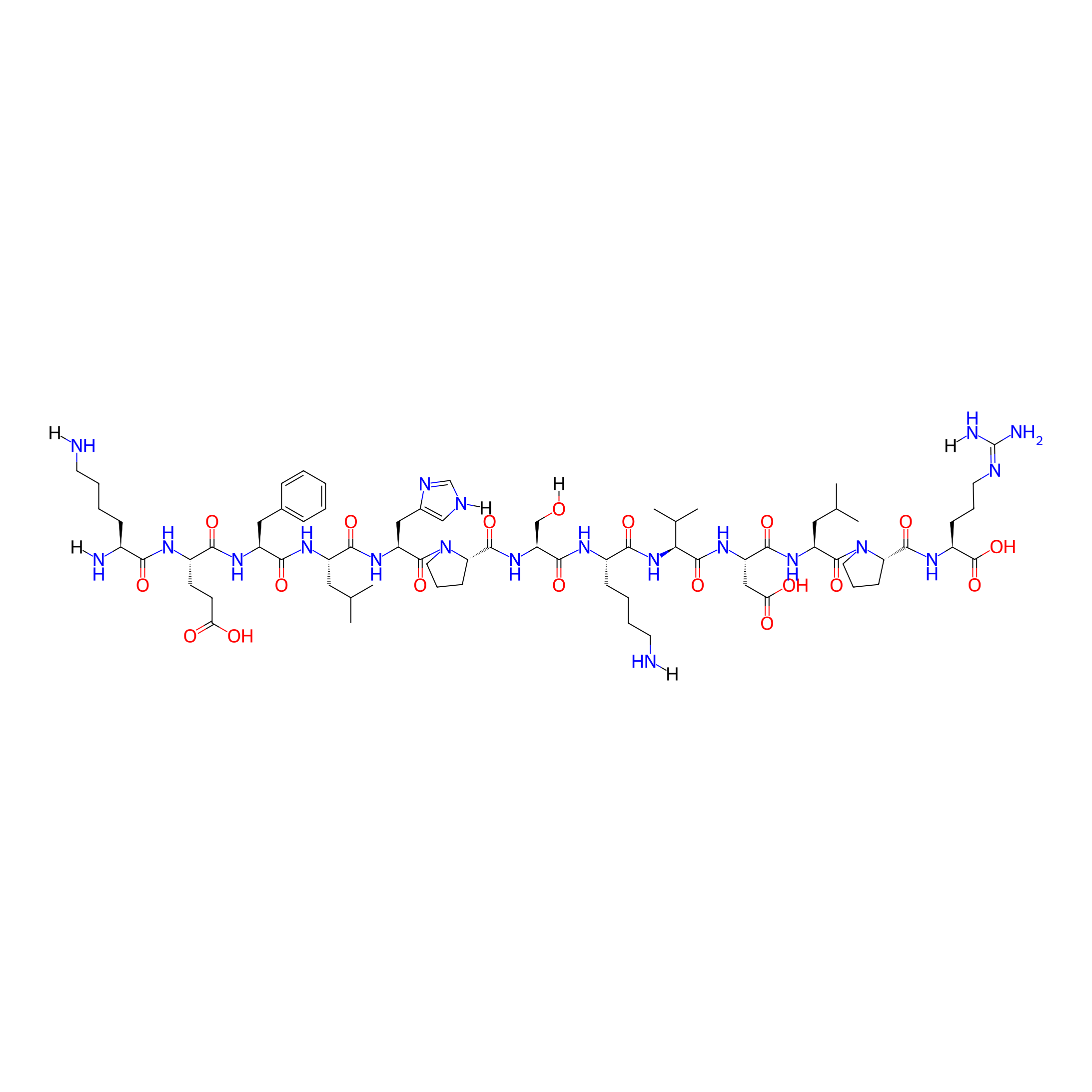
|
|||||
| Sequence |
KEFLHPSKVDLPR
|
|||||
| Peptide Type |
Linear
|
|||||
| Peptide Half Life Period |
<5 min
|
|||||
| Receptor Name |
Transient receptor potential cation channel subfamily V member 6 (TRPV6)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Absorption |
SOR-C13 was detected in plasma at 5 min after the start of infusion and continued to rise during the infusion. There were no marked differences in plasma SOR-C13 levels between 15 and 45 min after the start of infusion, suggesting steady state was achieved by 15 min post-infusion. Plasma SOR-C13 levels generally increased with increasing dose, the only exception to this being the data for 4.13 mg/kg were mean plasma levels were at times lower than those for the 2.75 mg/kg dose. This appeared to be due to large variability in two patients on some dosing days.
|
|||||
| Excretion |
SOR-C13 plasma levels declined rapidly once the infusion was stopped. For the lowest dose of 1.375 mg/kg, plasma SOR-C13 was undetectable after 10 min post-infusion. For the 2.75 and 6.2 mg/kg doses, plasma levels were detectable 10 min post-infusion, but in the majority of cases not at 30 min post-infusion (with the exception of one patient).
|
|||||
| Formula |
C72H116N20O19
|
|||||
| Isosmiles |
[H]NCCCC[C@H](N[H])C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1cn([H])cn1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO[H])C(=O)N[C@@H](CCCCN[H])C(=O)N[C@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC/N=C(/N)N[H])C(=O)O)C(C)C
|
|||||
| InChI |
InChI=1S/C72H116N20O19/c1-39(2)31-48(84-63(102)49(33-42-17-8-7-9-18-42)85-60(99)46(24-25-56(94)95)81-59(98)44(75)19-10-12-26-73)62(101)88-52(34-43-36-78-38-80-43)70(109)92-30-16-23-55(92)67(106)89-53(37-93)65(104)82-45(20-11-13-27-74)61(100)90-58(41(5)6)68(107)86-50(35-57(96)97)64(103)87-51(32-40(3)4)69(108)91-29-15-22-54(91)66(105)83-47(71(110)111)21-14-28-79-72(76)77/h7-9,17-18,36,38-41,44-55,58,93H,10-16,19-35,37,73-75H2,1-6H3,(H,78,80)(H,81,98)(H,82,104)(H,83,105)(H,84,102)(H,85,99)(H,86,107)(H,87,103)(H,88,101)(H,89,106)(H,90,100)(H,94,95)(H,96,97)(H,110,111)(H4,76,77,79)/t44-,45-,46-,47-,48-,49-,50-,51-,52-,53-,54-,55-,58-/m0/s1
|
|||||
| InChIKey |
LGANPTNILMNMES-TVNHODDRSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1565.841
|
Polar area
|
634.89
|
||
|
Complexity
|
1564.872561
|
xlogp Value
|
-4.1716
|
|||
|
Heavy Count
|
111
|
Rot Bonds
|
54
|
|||
|
Hbond acc
|
21
|
Hbond Donor
|
20
|
|||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
CBP-1008 [Phase 2/3]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Stable disease (SD) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Stable disease (SD) |
34.50%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Progressive disease (PD) |
17.20%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Progressive disease (PD) |
37.40%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Partial response (PR) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Partial response (PR) |
48.30%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Objective response rate(ORR) |
31.30%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Objective response rate(ORR) |
48.30%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian clear cell carcinoma | ||||
| Efficacy Data | Disease control rate (DCR) |
62.50%
|
|||
| Patients Enrolled |
16 platinum-resistant ovarian clear cell carcinoma patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
OCCC accounts for 5% to 25% of OC1, and current treatment options have very poor ORR of <10%. The ORR of CBP-1008 (0.15 or 0.17 mg/kg) for OCCC patients was 31.3% (5/16), regardless of FR expression.
|
||||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Ovarian cancer | ||||
| Efficacy Data | Disease control rate (DCR) |
82.80%
|
|||
| Patients Enrolled |
29 platinum-resistant ovarian cancer patients.
|
||||
| Administration Dosage | 0.15 mg/kg | ||||
| MOA of PDC |
Folate receptor α (FRα) and vanilloid subfamily member 6 of transient receptor potential channels (TRPV6) are potential promising therapeutic targets due to their high expression level in many solid tumors including ovarian cancer. CBP-1008 is a first-in-class (FIC) bi-specific ligand drug conjugate targeting FRα and TRPV6 carrying monomethyl auristatin E (MMAE) as payload.
Click to Show/Hide
|
||||
| Description |
Promising efficacy was observed in HGSOC patients (pts) who received prior 1-2L of TIT (if prior 2L, ≥12m of time interval between 2L; and ≥3m of time interval from the last TIT to CBP-1008 first dose). The ORR is 48.3%(14/29) and DCR is 82.8%(24/29), regardless of FR expression.
|
||||
References
